Sweet cherry (Prunus avium L.) belongs to the Rosaceae family and the Prunus genus (Dirlewanger et al., 2009). This fruit type is favored by consumers worldwide due to its thick flesh, mild flavor and attractive appearance. Along with strawberry, sweet cherry is the first fresh fruit from the open field. World sweet cherry production in 2023 was 2,963,781 t from 462,868 ha harvested area, while European production was 768,592 t from 167,845 ha (FAOSTAT, 2025). Based on data from this source, Serbian sweet cherry production amounted to 15,576 tons from 4,435 ha, which is only 5.26% and 20.27% of the world and European production, respectively.
In Serbia, sweet cherry is considered a “rising star” (Milošević et al., 2025a,b) or a “super fruit” which represents a unique combination of taste and high nutritional and medicinal values (Hu et al., 2021). Sweet cherry fruits are a scarce product on the world markets and, as a rule, have a high price. Due to this, growers are increasingly cultivating them all over the world. On the other hand, due to its attractive appearance, color (light, bi-color and/or red in various shades), taste and aroma, the sweet cherry is considered an elite table fruit. Over 80–85% of the fruits are used for fresh consumption, whereas the rest are processed into juice, marmalades, jams, jelly fruits and alcoholic beverages (Chockchaisawasdee et al., 2016). In Serbia, a traditional product called “slatko” - boiled fruits with sugar - is produced from fruits with light skin (Milošević et al., 2025a). As for the fresh sweet cherry market, consumers are setting higher standards from year to year. For example, consumers in the USA and Canada demand very large fruits - 12 to 15 g in weight, 34 mm in equatorial diameter, 17–19° Brix of soluble solids and less than 0.90% organic acids, dark red in color, slightly flattened in shape and with a medium-long green stem (Kappel et al., 2012).
As is commonly known, fruits of sweet cherry are a rich source of water, organic compounds, also called primary metabolites and minerals, especially K, Ca and Mg which have nutritive values. Apart from these, 26 elements were detected in sweet cherry fruits (Zhang et al., 2025). In the last few decades, consumers have been demanding fruits that, in addition to being nutritious, also have health benefits. From this point of view, sweet cherry fruits are also a rich source of phytochemicals known as secondary metabolites, primarily phenolic compounds (polyphenols), which possess high biological i.e. antioxidant activity, including anti-inflammatory, anticancer, antimutagenic, chemo-preventive and inhibiting-glucosidase activity, UV protection, pathogen resistance, etc. (Milea et al., 2019; Agulló-Chazarra et al., 2020). Tannins as a type of phenolic compounds are important components of many fruits, vegetables, and beverages which contribute to their color and sensory properties such as bitterness and/or astringency (Arnold et al., 1980). The distribution of these compounds differs depending on the plant species, genotype, tissue and harvest time, with many phenolics synthesized from carbohydrates via the shikimate and phenyl propanoid pathways (Lattanzio et al., 2006).
Antioxidant activity and phenolic composition of sweet cherries are generally influenced by climatic factors such as temperature, light intensity, light spectrum, and other environmental factors e.g. biotic and abiotic stresses, soil type, its pH etc. (Milea et al., 2019). Therefore, genotype or cultivar per se is a key factor that determines phenolic composition and antioxidant activity in plant organs and tissues (Boneza and Niemeyer, 2018) and also in fruits, including sweet cherry (Acero et al., 2019; Boskov et al., 2023). Hence, the genetic background is the first parameter with the potential to influence the antioxidant content in a commodity. However, in horticultural practice, the commercial growing of perennial fruit species, including sweet cherries and some small fruits, is unthinkable without the use of rootstocks for grafting of selected cultivars (Ling et al., 2025). Although the mechanism of influence on scion is not sufficiently elucidated, it is known that rootstocks can alter vegetative growth i.e. tree vigor, precocity, yield performance, phenological and physiological traits, leaf nutrient composition, response to pest and disease and fruit external and internal quality, especially fruit size (Milošević et al., 2014, 2025a,b).
In the available literature, data on the influence of sweet cherry rootstocks on the content of phenolic compounds and their antioxidant and inhibitory activity are modest. For these reasons, the main goal of this study was to determine the strength of the impact of two traditional root-stocks - Mazzard seedlings and clonal Colt on the content of bioactive substances in eight cherry cultivars during two consecutive years which grown under Čačak conditions (western Serbia). The obtained results could help find the best root-stock/cultivar combination in terms of the amount of phenolic compounds as an important indicator of fruit quality i.e. their antioxidant potential.
The sweet cherry fruits were collected in two consecutive years (2019 and 2020) from the 11- and 12-year-old private orchard located at Prislonica village (43°33′ N; 16°21′ E) near Čačak city (western Srbia), at 300 m above the sea level. Eight cultivars (‘SHG’, ‘June Early’, ‘Hedelfinger’, ‘New Star’, ‘Summit’, ‘Lapins’, ‘Germersdorfer’ and ‘Sunburst’) were grafted onto invigorating Mazzard seedlings and clonal medium-vigorous Colt rootstocks. The planting distance was 4.0 m × 2.0 m or 1,250 trees per ha. Randomized block design with four replications of five trees per plot of each rootstock–cultivar combination (n = 20) was applied; the training system was Vertical Axis. Standard cultural practices were applied, except irrigation.
Ten fruits of each rootstock/cultivar combination in four replicates (n = 40) were randomly hand picked at commercial harvest (S4 stage proposed by Serrano et al., 2005) by a single person to keep consistency of maturity grade each season. At S4 ripening stage, fruits had dark red color, adequate size and firmness for each cultivar.
The FW (g) was measured using the digital balance VK-Tech Ltd. (Daejeon, South Korea) at the laboratory of the Faculty of Agronomy in Čačak immediately after harvest. After measuring the FW, samples of the same fruits were used for chemical analyses. According to standard laboratory procedures as described by AOAC (2012), samples of whole fruits (flesh + skin) and subsamples were prepared for analytical determinations in three replicates. Spectrophotometric measurements for phenolic compounds and antioxidant activity were performed using Cary 300 UV/Vis spectrophotometer (Agilent Technologies, Santa Clara, CA).
The TPC and TFC were established using previously described procedures by Brighente et al. (2007) and Singleton and Rossi (1965), respectively. Results were expressed as mg of gallic acid equivalents (mg GAE g‒1) and mg of rutin equivalents (mg RUE g‒1) per g of dry weight (dw), respectively. Vit C content was determined through the 2,6-dichloroindophenol method (Arya et al., 2000). The results were expressed as mg 100 g–1 of fresh weight (fw). The TAc was determined applying the procedure described by Cheng and Breen (1991) using pH single and differential methods. Results were expressed as mg 100 g‒1 of fresh weight (fw). The CT and GT were determined using methods with potassium iodate assay (Vermerris and Nicholson, 2006). Both results were expressed as mg GAE g‒1 dw.
Antioxidant activity of previously prepared extracts was assessed using four different assays: total antioxidant capacity (TAC) (Prieto et al., 1999), hydroxyl radical scavenging activity (HR) (Hinneburg et al., 2006) and DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity (Takao et al., 1994) with slight modification (Kumarasamy et al., 2007). The TAC was expressed as micrograms of ascorbic acid per gram of dw (μg AA g‒1). Results for HR and DPPH as IC50 in μg mL‒1 were presented. The inhibitory activity against lipid peroxidation (LPO) was determined according to the method described by Hsu et al. (2008) and results were expressed as IC50 in μg mL‒1.
All chemicals and reagents were of analytical grade and were purchased from Sigma Chemical Co. (St Louis, USA), Aldrich Chemical Co. (Steinheim, Germany) and Alfa Aesar (Karlsruhe, Germany). All chemicals were of analytical grade and purest quality available.
Data for 2019 and 2020 were pooled and then they were calculated as average values for each property. Difference detection between data was separately done by two-way analysis of variance (ANOVA) using MS-Excel software (Microsoft Corporation, Redmond, WA, USA). Source of variations were rootstock and cultivar. Year alone and its interactions with rootstock and/or cultivar for each property were not significant. The means were compared with the LSD test at the P ≤ 0.05. Pearson’s rank correlation matrix was done using the R corrplot package (Wei and Simko, 2017). The PCA as well as bi-plot graphical display were performed using the XLSTAT software package v. 7.0 (Addinsoft, Paris, France).
The values presented for each measurement are the means of triplicate measures on equidistant points of each compound and expressed as the mean ± standard error (SE).
The FW is the most important property of sweet cherry in terms of external fruit quality and always very important for both, growers and consumers. In the current study FW was significantly affected by rootstock, being 5.33% higher in trees on clonal Colt seedlings compared to those on Mazzard (Fig. 1). It has already been described previously that rootstocks can have a significant positive or negative effect on sweet cherry fruit size (López-Ortega et al., 2016; Milošević et al., 2025a).
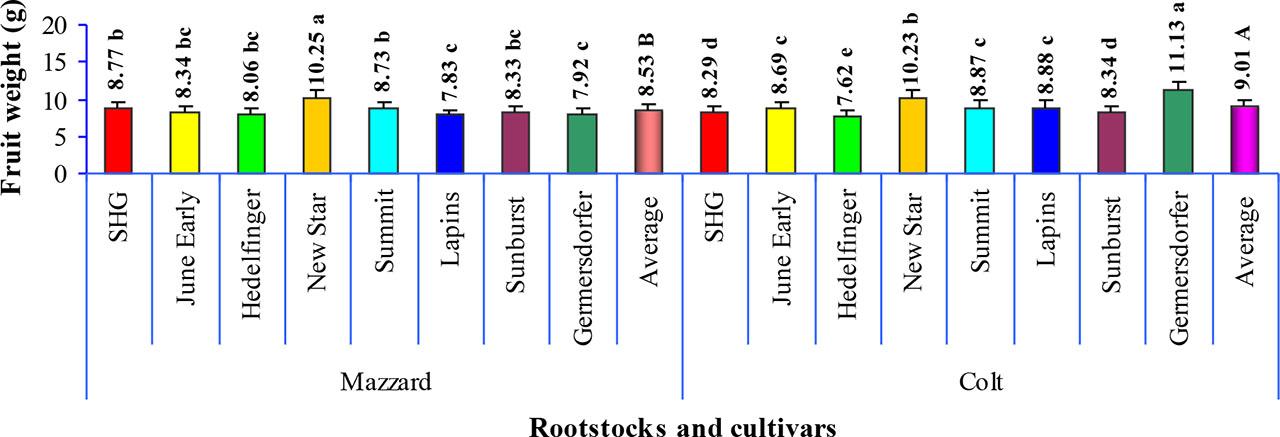
Average fruit weigh of eight sweet cherry cultivars grafted on Mazzard seedlings and clonal Colt rootstocks. Data are mean for 2019 and 2020. Different small letters at the top of the column indicate significant differences between cultivars within rootstocks, while different capital letters indicate significant differences between rootstocks at P ≤ 0.05 by LSD test.
For example, Wociór (2008) noted that there was no significant difference between Mazzard and Colt for FW of ‘Kordia’ sweet cherry which contradicts our results.
Data in Fig. 1 show that the behavior of the cultivars on these two rootstocks was not uniform. On Mazzard, ‘New Star’ had the highest FW whereas the lowest and similar were recorded in ‘Lapins’ and ‘Germersdorfer’. However, the FW of these two cultivars did not significantly differ from that of ‘June Early’, ‘Hedelfinger’ and ‘Sunburst’. On Colt, quite contrary to Mazzard, ‘Germersdorfer’ had the highest FW. Similarly to its performance on Mazzard, ‘New Star’ grafted on Colt also had a FW value exceeding 10 g. The lowest FW on Colt was determined for ‘Hedelfinger’ which also produced smaller fruits on Mazzard.
Variations in FW among different cultivars of the same fruit have been previously investigated. Girard and Kopp (1998) reported a range from 8.8 g to 14.5 g in cherries grown in Summerland, Canada. In their study, ‘Lapins’ and ‘Summit’ had much higher FW compared to our findings. As known, both these cultivars were named and released at Summerland Research Station, British Columbia (Canada) (Lapins, 1974; Lane and Schmid, 1984). Given this origin, they are far better adapted to Summerland environmental conditions than to those in Serbia, which likely explains the drastic differences in FW of both ‘La-pins’ and ‘Summit’. Generally, our range of values was within the specified limits reported for ‘Sun-burst’ on Colt under the conditions of Zaragoza (Spain) by Jiménez et al. (2004). However, Cantín et al. (2010) observed much lower FW values than ours for ‘SHG’ on various rootstocks under Ebro Valley (Spain) conditions, including invigorating ones. Additionally, Radičević et al. (2019) noted that ‘Summit’ under conditions like ours, but grafted on dwarfing Gisela 5 rootstock, had a FW of 9.69 g which was higher than the values recorded on both rootstocks in our study. The discrepancies between our results and those of other authors can be attributed to several factors such as rootstock/cultivar combination i.e. their vigor, leaf/fruit ratio, tree age, training system, cultural practices and pedo-climatic conditions (Jiménez et al., 2004; Whiting et al., 2005; Milošević et al., 2025a, b). Some authors related FW to crop load - when the crop load is high, FW tends to be lower, and vice versa (Whiting and Ophardt, 2005). However, Cantín et al. (2010) reported that there was not significant relationship between these two parameters.
Sweet cherries are a rich natural source of phenolic compounds. The TPC value represents all the phenolic compounds, including flavonoids, phenolic acids and other phenolics (Hu et al., 2021). Flavonoids are a diverse group of compounds found naturally in many fruits and vegetables with a broad spectrum of chemical and biological activities, particularly free radical scavenging and antimicrobial activities (Nunes et al., 2021).
The ANOVA showed significant differences between rootstocks for TPC; however, no significant differences were observed between rootstocks for TFC (Tab. 1). TPC values were generally higher by 6.3% on Colt than on Mazzard. Several authors found significant variations among rootstocks for both TPC and TFC (Spinardi et al., 2005; Milinović et al., 2016; Boskov et al., 2023). There is controversy and disagreement in the available literature regarding the influence of individual rootstocks on these compounds. For example, Balducci et al. (2019) found that P. avium seedlings (Mazzard) induced the lowest TPC value in ‘Sweetheart’ as compared with 9 clonal rootstocks. Contrary to these findings, in our study we determined both, a TPC and TFC, higher on Mazzard than on Colt in ‘Grace Star’ (Milošević et al., 2025a). Probably, the specific physiological relationship of the scion/rootstock combination grown under specific pedo-climatic conditions, training systems and cultural practices is the reason for these contrasting results.
Total phenolics, total flavonoid, vitamin C, condensed tannins and gallotannins contents of eight sweet cherry cultivars grafted on Mazzard and Colt rootstocks. Data are the mean ± SE for 2019 and 2020.
| Rootstock | Cultivar | Total phenolics content (mg GAE g−1) | Total flavonoids content (mg RUE g‒1) | Vitamin C (mg 100 g‒1) | Total anthocyanins content (mg 100 g‒1) | Condensed tannins (mg GAE g‒1) | Gallotannins (mg GAE g‒1) |
|---|---|---|---|---|---|---|---|
| Mazzard | SHG | 1.87 ± 0.06 c | 1.04 ± 0.07 bc | 6.87 ± 0.11 d | 0.72 ± 0.03 b | 7.62 ± 0.15 b | 3.72 ± 0.11 b |
| June Early | 2.03 ± 0.05 b | 1.11 ± 0.04 b | 10.56 ± 0.14 a | 0.92 ± 0.05 a | 7.12 ± 0.23 c | 3.42 ± 0.09 c | |
| Hedelfinger | 2.05 ± 0.07 ab | 0.94 ± 0.04 c | 7.85 ± 0.34 c | 0.85 ± 0.07 a | 7.11 ± 0.20 c | 3.51 ± 0.09 c | |
| New Star | 0.44 ± 0.01 f | 0.32 ± 0.03 f | 9.17 ± 0.30 b | 0.27 ± 0.03 e | 5.21 ± 0.18 e | 2.54 ± 0.08 e | |
| Summit | 2.10 ± 0.06 a | 1.08 ± 0.05 bc | 9.22 ± 0.19 b | 0.42 ± 0.03 d | 8.36 ± 0.20 a | 4.28 ± 0.16 a | |
| Lapins | 2.12 ± 0.07 a | 1.29 ± 0.05 a | 8.32 ± 0.07 c | 0.47 ± 0.02 cd | 6.32 ± 0.21 d | 3.27 ± 0.12 d | |
| Sunburst | 1.05 ± 0.07 e | 0.73 ± 0.03 d | 6.15 ± 0.30 e | 0.51 ± 0.03 c | 7.68 ± 0.17 b | 3.60 ± 0.19 bc | |
| Germersdorfer | 1.45 ± 0.04 d | 0.64 ± 0.03 e | 8.47 ± 0.33 c | 0.45 ± 0.03 cd | 6.58 ± 0.17 d | 3.04 ± 0.13 d | |
| Mean | 1.64 ± 0.05 B | 0.90 ± 0.04 A | 8.32 ± 0.23 A | 0.58 ± 0.04 B | 7.00 ± 0.19 A | 3.42 ± 0.13 A | |
| Colt | SHG | 1.75 ± 0.07 bc | 1.07 ± 0.07 b | 6.95 ± 0.12 e | 0.77 ± 0.04 c | 7.65 ± 0.14 b | 3.75 ± 0.12 b |
| June Early | 2.23 ± 0.06 a | 1.24 ± 0.07 ab | 10.63 ± 0.39 a | 0.86 ± 0.04 b | 7.32 ± 0.19 c | 3.53 ± 0.12 bc | |
| Hedelfinger | 1.94 ± 0.06 b | 0.89 ± 0.05 c | 7.53 ± 0.10 d | 0.93 ± 0.06 a | 7.43 ± 0.11 c | 3.71 ± 0.09 b | |
| New Star | 0.52 ± 0.03 d | 0.34 ± 0.03 d | 9.52 ± 0.15 b | 0.38 ± 0.04 f | 5.60 ± 0.07 f | 2.73 ± 0.11 d | |
| Summit | 2.13 ± 0.08 ab | 1.11 ± 0.04 b | 9.28 ± 0.10 b | 0.44 ± 0.03 ef | 8.39 ± 0.18 a | 4.34 ± 0.15 a | |
| Lapins | 2.19 ± 0.09 a | 1.33 ± 0.10 a | 8.39 ± 0.09 c | 0.53 ± 0.03 de | 6.10 ± 0.20 e | 3.33 ± 0.12 c | |
| Sunburst | 1.67 ± 0.09 bc | 1.00 ± 0.05 bc | 6.22 ± 0.20 f | 0.56 ± 0.02 d | 7.72 ± 0.16 b | 3.69 ± 0.20 b | |
| Germersdorfer | 1.58 ± 0.05 c | 0.66 ± 0.03 c | 8.50 ± 0.10 c | 0.49 ± 0.03 e | 6.61 ± 0.15 d | 3.11 ± 0.13 c | |
| Mean | 1.75 ± 0.07 A | 0.96 ± 0.05 A | 8.38 ± 0.16 A | 0.62 ± 0.04 A | 7.10 ± 0.15 A | 3.52 ± 0.13 A | |
Different small letters in same column indicate significantly different values within each cultivar at P ≤ 0.05 by LSD test.
Different capital letters in same column indicate significantly different values within each rootstock at P ≤ 0.05 by LSD test.
Regarding cultivars, ‘Lapins’ and ‘Summit’ on Mazzard and ‘June Early’ and ‘Lapins’ on Colt show the tendency to accumulate similar and highest TPC levels. However, these values were not significantly different from those of ‘Germersdorfer’ on Mazzard and ‘Summit’ on Colt. The lowest TPC was observed in ‘New Star’ on both rootstocks. ‘Lapins’ and ‘New Star’ had the highest and the lowest TFC, respectively. Significant inter-cultivar variation in the TPC and TFC has been documented in other studies on sweet cherry (Prvulović et al., 2012; Boskov et al., 2023; Kazazic et al., 2024). Hu et al. (2021) also found that ‘La-pins’ had the highest TPC and TFC of four commercial sweet cherries, whereas, similarly to our results, Papapetros et al. (2019) reported that ‘La-pins’ and ‘Hedelfinger’ had higher TPC than other cultivars. In a study of Prvulović et al. (2019), ‘Hedelfinger’ had higher TFC than other cultivars such as ‘Germersdorfer’, ‘New Star’, ‘Summit’ and ‘Sunburst’ and respectable TPC which generally agreed with our results. In addition, apart from climatic and agronomic factors, the biosynthesis of phenolic compounds is triggered by exposure to stress conditions, as a natural defense system, generating considerable variability (Kumar et al., 2023).
Vit C is known for its strong antioxidant properties, as it, among other things, protects the human body from oxidative stress, facilitates collagen synthesis, supports skin health, improves immune function and wound healing, and promotes iron absorption in the intestines. (Alberts et al., 2025). Fruits rich in this vitamin are considered beneficial for human health. Data in Tab. 1 showed that the rootstocks did not significantly influence Vit C content. This result is not in agreement with data of other authors (Spinardi et al., 2005; Prichko and Sivoplyasov, 2021) according to which the root-stocks significantly change the amount of Vit C in sweet cherries. Probably, the rootstock type and their genetic background play an important role in the possibility of influencing this trait (Prichko and Sivoplyasov, 2021).
The behaviour of the cultivars in terms of Vit C content on both Mazzard and Colt rootstocks was consistent. ‘Early June’, as a early ripening cultivar, had the highest content, followed by ‘New Star’ and ‘Summit’, whereas the late ripening cultivar ‘Sunburst’ had the lowest. There were approximately 1.7-fold differences between the highest and the lowest contents. The content of Vit C depends on the cultivar and significantly varied among them as previously reported (Bieniek et al., 2011). In a study of these authors, content of this compound in 7 sweet cherry cultivars on Mazzard varied from 5.50 mg 100 g‒1 to 8.15 mg 100 g‒1, whereas Prichko and Sivoplyasov (2021) noted that the ranges of this compound were from 6.8 mg 100 g‒1 to 9.8 mg 100 g‒1. In general, contents of Vit C in sweet cherries are low and varied from 5 to 10 mg 100 g‒1 (Sitarek, 2004, cit. by Bieniek et al., 2011) which is confirmed by our results.
Anthocyanins as water-soluble natural pigments are responsible for most of the red, blue and purple colors of fruits, grapes, vegetables, flowers and other plant tissues or products (Khoo et al., 2017). These compounds are the most abundant phenolics in sweet cherry fruits, especially in skin (Agulló-Chazarra et al., 2020) of dark-red colored genotypes (Mozetič et al., 2002).
In the present study (Tab. 1), Mazzard induced slightly but significantly higher TAc than Colt, in agreement with results of other authors (Milinović et al., 2016; Balducci et al., 2019). Boskov et al. (2023) reported that Colt promoted the highest value of TAc in comparison with other clonal root-stocks. In our recent study (Milošević et al., 2025a), we found that Mazzard and Colt had moderate to high TAc values compared to 11 clonal dwarf to semi-dwarf rootstocks with different genetic origin. In this study, we also found that Colt induced significantly higher TAc content as compared to Mazzard. Similarly, Balducci et al. (2019) noted that the worst result of TAc was registered for fruits from trees on P. avium rootstock when compared to 9 clonal rootstocks.
The TAc was also influenced by cultivar and a relatively consistent pattern was observed across rootstocks. ‘June Early’ and ‘Hedelfinger’ had similar TAc on Mazzard whereas the lowest was observed in ‘New Star’. On Colt, the highest value was also found with ‘Hedelfinger’ and the lowest with ‘New Star’ which was no different from ‘Summit’. Mozetič et al. (2002) reported that in fruits of 5 dark colored cultivars, TAc varied from 97.38 to 196.98 mg GAE g‒1 which is much higher than the values found in our study. On the other hand, Prvulović et al. (2019) reported a behavior that was - in terms of TAc content - diametrically different to the results in our study observed with ‘Germersdorfer’, ‘Hedelfinger’, ‘New Star’, ‘Summit and ‘Sunburst’ when grown under northern Serbian conditions. In their study, for example, ‘New Star’ had the highest TAc content and ‘Hedelfinger’ the lowest. These discrepancies can be attributed to the fact that apart from the root-stock, and mostly the cultivar, the level of Tac is also influenced by climatic factors such as temperature, light intensity, light spectrum, maturity stage and other environmental factors and cultural practices (Agulló-Chazarra et al., 2020). Additionally, exposure to stressful conditions of biotic and abiotic origin also contributes to greater accumulation of anthocyanins in tissues (Kumar et al., 2023).
Condensed tannins are abundant in fruits, fruit products and beverages and associated with the astringency, bitterness and color (Arnold et al., 1980). Gallotannins are a type of tannin, which are naturally occurring polyphenols. They are found in various plants, fruits and berries and are known for their astringent taste. According to data in Tab. 1, the rootstocks did not significantly influence CT and GT contents which is in contrast with our findings in pear, where rootstocks significantly affected both compounds (Milošević et al., 2020). It is likely that the rootstock type, its origin and cultivar type play an important role in the accumulation of these compounds in the fruits.
Unlike among rootstocks, the variability of the content of these phytochemicals among cultivars was pronounced and statistically significant. The behavior of the cultivars was consistent on both rootstocks. ‘Summit’ contained the highest both CT and GT, whereas the lowest was determined in fruits of ‘New Star’. Following ‘Summit’, high contents of these phytochemicals were observed in ‘SHG’, ‘Sunburst’ and partially in ‘June Early’. Hu et al. (2021) reported that ‘Lapins’ had much higher tannins content than ‘Merchant’ and ‘Bing’, respectively. Interestingly, in a study of above authors, tannins were not detected in ‘Ron’s’ sweet cherry. In contrast, ‘Lapins’ in our experiment had low values in comparison to other cultivars. On this occasion we state that fruits of ‘Sunburst’ have pronounced astrigent taste under Čačak conditions, probably due to high content of both tannins (see Tab. 1), which discourages consumers in markets (Milošević et al., 2014). However, in the experiment of Prvulović et al. (2019) ‘Sunburst’ contained lower tannins content in comparison to more cultivars, especially ‘Germersdorfer’, ‘Hedelfinger’, ‘New Star’ and ‘Summit’ which were all included in our trial. A wide variation in tannins content among cultivars was also noted by Prvulović et al. (2012), which is in accordance with our results.
Data from relevant literature indicated that sweet cherry fruits contain less tannin than sour cherry but much more than apricot and plum cv. ‘Green Gage’ (Çevik et al., 2013). All previous similarities and discrepancies can be attributed to the influence of the fruit species, genotype or cultivar per se, local weather conditions during fruit development, the course of the tannin synthesis process in the tannosomes (organelles found in plant cells) and fruit maturity stage.
It has long been known that the use of a single method for determining the antioxidant capacity of foodstuff of different origins and food extracts is insufficient (Schlesier et al., 2002). Given this fact, a large number of authors recommend at least two validated methods, and some suggest more than two in order to better determine results authenticity (Milošević et al., 2020).
Results in Tab. 2 showed that no significant differences between rootstocks for TAC, LPO, HR and DPPH scavenging activity were found. These findings are inconsistent with those of Kazazic et al. (2024) and Milošević et al. (2025a) who reported that rootstocks significantly changed these parameters. For instance, Balducci et al. (2019) compared effect of several rootstocks and determined that P. avium seedlings as rootstock induced the lowest TAC value of ‘Sweetheart’ sweet cherry in relation to others.
Total antioxidant capacity, inhibitory activity against lipid peroxidation, hydroxyl radical scavenging activity and DPPH scavenging activity of eight sweet cherry cultivars grafted on Mazzard and Colt rootstocks. Data are the mean ± SE for 2019 and 2020.
| Rootstock | Cultivar | Total antioxidant capacity (μg AA g−1) | Inhibitory activity against lipid peroxidation aIC50 (µg mL‒1) | Hydroxyl radical scavenging activity aIC50 (µg mL‒1) | DPPH scavenging activity aIC50 (µg mL‒1) |
|---|---|---|---|---|---|
| Mazzard | SHG | 2.09 ± 0.05 b | 33.24 ± 0.43 ab | 34.44 ± 0.45 b | 47.34 ± 0.38 g |
| June Early | 1.62 ± 0.06 c | 32.12 ± 0.37 b | 37.89 ± 0.34 ab | 71.12 ± 0.67 b | |
| Hedelfinger | 2.24 ± 0.07 a | 33.54 ± 0.40 ab | 39.54 ± 0.40 a | 86.00 ± 0.59 a | |
| New Star | 0.94 ± 0.03 f | 33.79 ± 0.50 ab | 39.85 ± 0.54 a | 47.31 ± 0.63 g | |
| Summit | 1.65 ± 0.04 c | 32.57 ± 1.03 ab | 35.47 ± 1.35 b | 57.02 ± 0.54 e | |
| Lapins | 1.29 ± 0.04 d | 31.91 ± 0.39 b | 30.86 ± 1.45 c | 59.23 ± 0.49 d | |
| Sunburst | 1.17 ± 0.05 e | 31.65 ± 0.48 b | 32.02 ± 1.36 bc | 68.10 ± 0.53 c | |
| Germersdorfer | 1.30 ± 0.05 d | 34.96 ± 1.00 a | 29.96 ± 2.04 c | 54.62 ± 0.42 f | |
| Mean | 1.54 ± 0.05 A | 32.97 ± 0.58 A | 35.00 ± 0.99 A | 61.44 ± 0.53 A | |
| Colt | SHG | 2.12 ± 0.05 a | 33.47 ± 0.43 ab | 34.98 ± 0.97 b | 47.54 ± 0.30 h |
| June Early | 1.71 ± 0.05 b | 31.61 ± 0.74 b | 38.61 ± 0.37 b | 71.27 ± 0.62 b | |
| Hedelfinger | 2.11 ± 0.06 a | 34.11 ± 0.44 a | 40.42 ± 1.17 a | 86.94 ± 0.57 a | |
| New Star | 1.07 ± 0.03 d | 34.65 ± 0.34 a | 40.07 ± 0.72 a | 49.27 ± 0.73 g | |
| Summit | 1.67 ± 0.08 b | 32.63 ± 0.34 b | 35.65 ± 0.64 ab | 57.14 ± 0.40 e | |
| Lapins | 1.32 ± 0.04 c | 32.10 ± 0.92 b | 30.75 ± 1.26 c | 60.04 ± 0.58 d | |
| Sunburst | 1.19 ± 0.06 d | 31.70 ± 0.30 b | 31.36 ± 1.36 c | 68.13 ± 0.44 c | |
| Germersdorfer | 1.36 ± 0.05 c | 34.99 ± 1.32 a | 29.99 ± 2.26 c | 54.68 ± 0.38 f | |
| Mean | 1.57 ± 0.05 A | 33.16 ± 0.60 A | 35.23 ± 1.09 A | 61.88 ± 0.50 A | |
Different small letters in same column indicate significantly different values within each cultivar at P ≤ 0.05 by LSD test.
Different capital letters in same column indicate significantly different values within each rootstock at P ≤ 0.05 by LSD test.
The disagreement between our results and those of other authors may be related to the fact that the two rootstocks used in our study have a similar genetic origin, since one of the parents of the Colt rootstock, which was created by crossing, is P. avium i.e. wild cherry. Indeed, rootstocks of different vigor and different origin produced fruits with very different chemical properties, indicating that the rootstock effect is more complex than just the influence on vigor and other commonly known properties (Jakobek et al., 2009; Milošević et al., 2025a).
In contrast to the rootstocks, the cultivars showed a significant impact on the antioxidant potential. With regard to TAC, ‘Hedelfinger’ alone on Mazzard and alongside with ‘SHG’ on Colt had the highest values, whereas ‘New Star’ had the lowest on both rootstocks. In addition, ‘Sunburst’ on Colt had a statistically similar TAC value as ‘New Star’.
The TAC values induced by cultivars are highly variable in literature, demonstrating a great variability of this parameter (Mehić et al., 2023; Kazazic et al., 2024). In general, our values were similar to those found by Usenik et al. (2008).
On average, a higher value of LPO was recorded in ‘Germersdorfer’ trees grafted on Mazzard, without being significantly different from ‘New Star’, ‘Hedelfinger’, ‘SHG’ and ‘Summit’, respectively (Tab. 2). The lower and statistically similar values were observed in ‘June Early’, ‘Lapins’ and ‘Sun-burst’. On Colt, ‘Germersdorfer’, ‘New Star’ and ‘Hedelfinger’ showed a tendency to induce similar and higher LPO than the rest of the cultivars, although no differences were observed with ‘SHG’. For HR values, similar behavior of the cultivars was observed on both rootstocks. In general, ‘New Star’, ‘Hedelfinger’ had the highest values, whereas the lowest and most similar ones were recorded in ‘Lapins’, ‘Sunburst’ and ‘Germersdorfer’. ‘June Early’ on Mazzard and ‘Summit’ on Colt had similar values with cultivars which had the highest HR levels. Cultivars with lower values were the most active against LPO and HR. Mulabagal et al. (2009) reported that values of above assays in sweet cherry fruits were highly cultivar-dependent which is supported by our data.
The antioxidant activity using DPPH assay in sweet cherries is shown in Tab. 2. The IC50 DPPH values significantly varied in a wide range among cultivars whose behavior on both Mazzard and Colt rootstocks was consistent. ‘Hedelfinger’ had the highest IC50 DPPH values on both rootstocks i.e. the lowest capacity to neutralize DPPH radicals. The lowest IC50 DPPH values i.e. the highest anti-oxidant potential was observed in ‘SHG’ on both rootstocks and in ‘New Star’ on Mazzard with no significant differences between them. There was a 1.8-fold difference between the highest and the lowest IC50 DPPH values i.e. between cultivars, which is generally in agreement with results of Prvulović et al. (2012). However, in another study of Prvulović et al. (2019), there was a 5.4-fold difference between the lowest and the highest IC50 DPPH values of 15 cultivars which is much higher than our ratio. DPPH values and ratios between them for the same cultivars included in a study of Prvulović et al. (2019) and our trial, such as ‘Germersdorfer’, ‘Hedelfinger’, ‘New Star’, ‘Summit’ and ‘Sunburst’ were different. Hu et al. (2021) reported that ‘Lapins’ had the highest DPPH values of four sweet cherries. Several authors also reported high differences among cultivars with regard to DPPH values (Faniadis et al., 2010; Zhao et al., 2019; Li et al., 2020). It can be said that a comparison of the IC50 DPPH values obtained in our work with those of other studies suggests similar trends although differences in the units reported, methods used, way calculations and spectophoto-metric standards employed make a direct comparison difficult. Also, discrepancies between our data and results of other authors, demonstrate a great variability of this parameter depending on the scion genotype, pedo-climatic conditions and also cultural practices used, as previously reported by Milošević et al. (2020).
Pearson’s correlation analysis among the evaluated parameters revealed several noteworthy relationships (Fig. 2). The FW, TAc, TPC, TFC and DPPH scavenging activity have been directly correlated to each other; in fact, the FW was inversely correlated with TAc, TPC, TFC and DPPH values and vice versa, indicating that cultivars with smaller sized fruits contained higher phenolic compounds and better antioxidant activity than larger fruits, which is in agreement with results of Milošević et al. (2025a) who reported a similar relationship in sweet cherry. The TAc positively and highly correlated with TAC, whereas TPC very strongly and positively correlated with TFC. Similar relationships were found by Prvulović et al. (2019). Namely, fruits with high contents of total phenols and anthocyanins, both high antioxidants, exhibited a high antioxidant potential through the TAC value (Balducci et al. 2019). The correlation of antioxidant activity with total phenolics content and amount of anthocyanins was cultivar dependent (Usenik et al., 2008; Prvulović et al., 2019). A strong positive correlation of CT versus GT was found. This result showed that sweet cherry fruits rich in CT are also a rich source of GT, as previously reported (Çevik et al., 2013; Cosme et al., 2025). Other correlations were observed but lacked statistical significance.
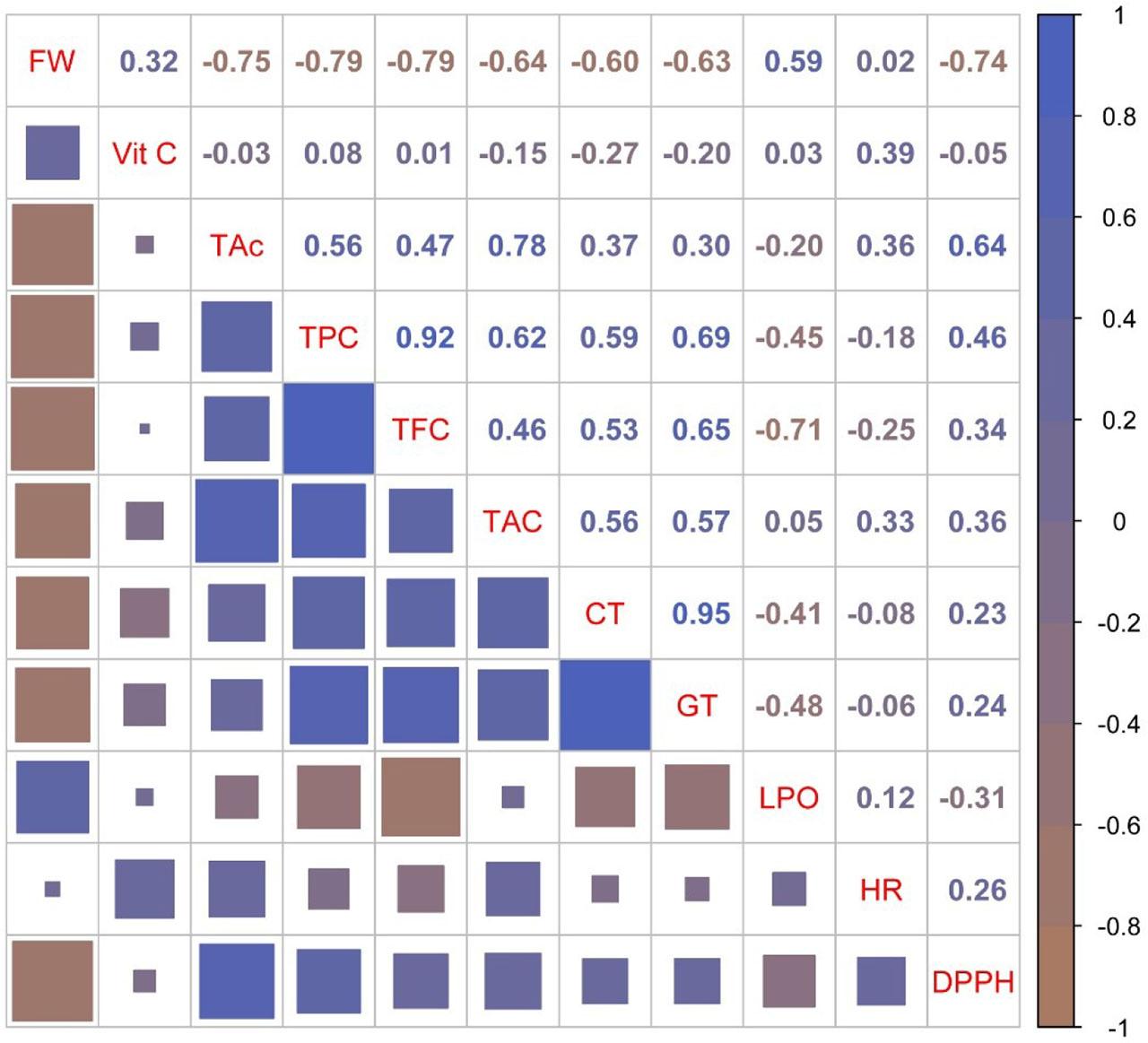
Pearson’s correlation coefficients for 11 studied fruit quality properties of 8 commercial sweet cherry cultivars. The size of the square for each correlation and the color reflect the statistical significance and magnitude of the correlation coefficient (r). Brown squares indicate negative correlations, whereas blue stands for positive correlations.
The PCA was conducted to evaluate variation in fruit weight and antioxidant properties among eight sweet cherry cultivars. In the present study, the first three principal components explained a cumulative 78.24% of the total variability, with PC1 accounting for 49.40%, PC2 for 17.19% and PC3 for 11.65%.
PC1 was positively associated only with FW, while negative loadings were observed for total TPC, TFC, CT and GT. PC2 was defined by positive loadings for TAc, HR and DPPH, whereas no parameters were associated with negative PC2 values (Fig. 3). PC3 was linked positively by Vit C, while TAC and LPO were associated with its negative side.
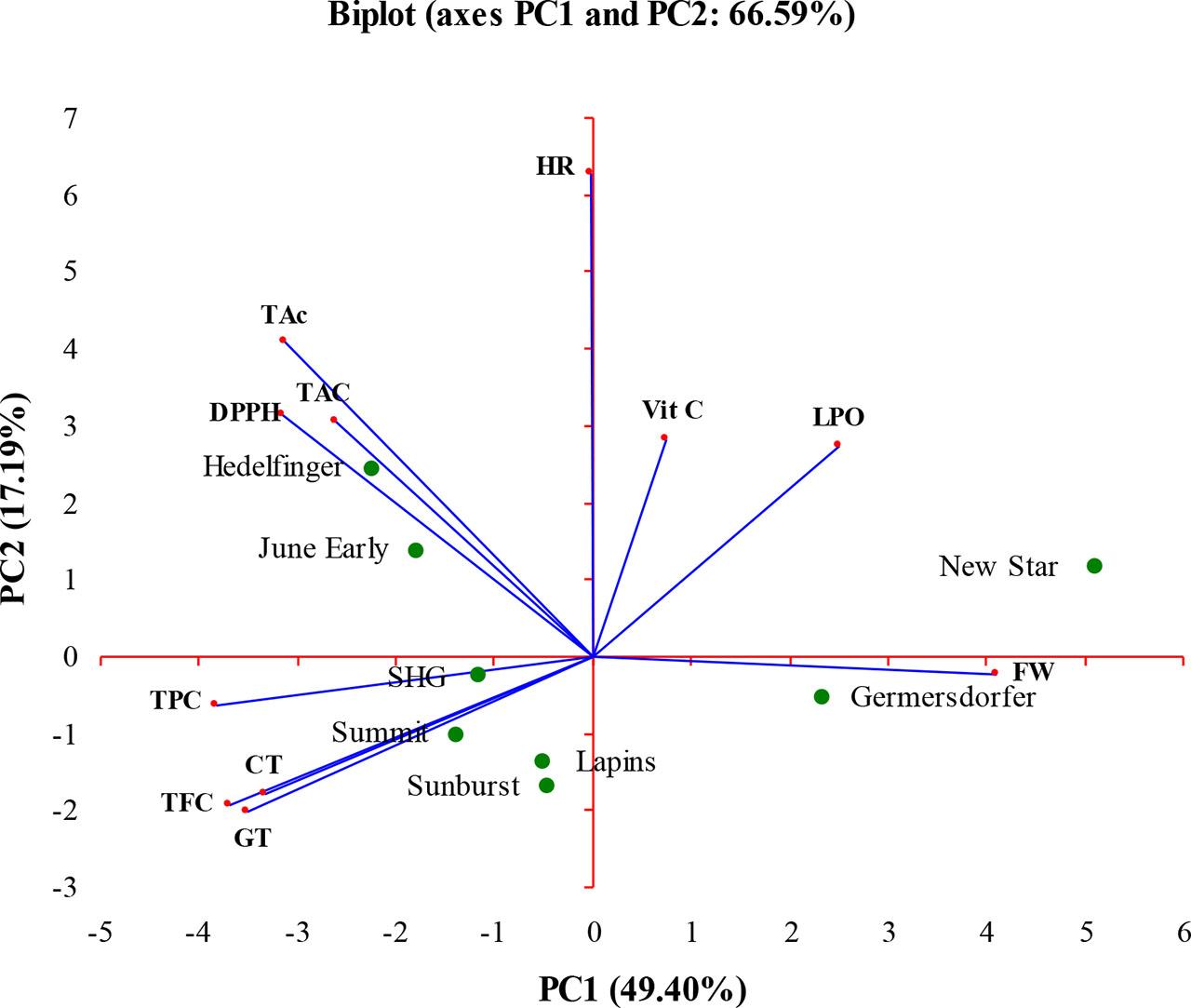
Segregation of 8 sweet cherry cultivars according to their fruit weight, phenolic compounds content and antioxidant activity determined by principal component analysis (PCA).
Regarding cultivar positioning, ‘New Star’ and ‘Germersdorfer’ were associated with positive PC1 values, indicating higher fruit weight, whereas ‘Summit’ was positioned on the negative side of PC1, suggesting higher levels of phenolic and tannin compounds but lower fruit weight. ‘Hedelfinger’ was linked to positive PC2 values, corresponding to elevated antioxidant activity (TAc, HR, DPPH). ‘Sunburst’ was positioned on the negative side of PC2, implying lower values for these anti-oxidant characteristics. Nevertheless, due to its high content of phenolic and tannin compounds (TPC, TFC, CT, GT), traits associated with the negative side of PC1, ‘Sunburst’ may represent a cultivar with strong phenolic accumulation but limited radical scavenging capacity. On the PC3 axis, ‘Lapins’ and ‘June Early’ were associated with positive values, indicating higher Vit C content, while ‘SHG’ was reflecting higher TAC and LPO levels.
In summary, based on PCA results, ‘Hedelfinger’, ‘New Star’ and ‘Lapins’ may be considered superior due to their favorable combination of FW and antioxidant properties. Conversely, ‘Summit’, ‘SHG’ and ‘Sunburst’ were less favorably positioned and may be considered less promising in the context of the studied traits.
The traditional invigorating Mazzard seedlings and medium-vigorous clonal Colt rootstocks tested in our study demonstrated limited influence on fruit weight, phenolic compounds content and antioxidant activity of eight commercial sweet cherries. Of the 11 evaluated traits, only three (27.3%) were induced by the rootstocks, i.e. Colt induced higher values of fruit weight, total anthocyanins content and total phenolics content, while other differences were not significant. The absence of root-stock effects on the remaining secondary metabolites content and antioxidant capacity is likely due to their similar genetic origin, since one of the parents of the Colt rootstock, created by crossing, was Mazzard, i.e. P. avium L. On the other hand, phenolic compounds content and antioxidant activity were more strongly influenced by cultivar than rootstock. In general, ‘Hedelfinger’ as a late ripening and smaller fruited cultivar can be categorized as the richest in total phenolic compounds and total anthocyanins, as well as the one with the highest antioxidant potential. In contrast, ‘New Star’ characterized by the largest fruits, had the lowest levels of total phenols, total flavonoids, tannins and total antioxidant capacity but showed the highest inhibition of lipid peroxidation and hydroxyl radical scavenging activity. Correlation and principal component analyses confirmed that the antioxidant potential of sweet cherries is related to fruit size, phenolics, flavonoids or anthocyanins content. Finally, both Mazzard and Colt seem to be well adapted to the environmental conditions in this trial. On the other hand, producers should seriously consider adequate cultivar selection in order to achieve optimal accumulation of secondary metabolites and the best antioxidant potential of their fruits. However, the final recommendation should also consider additional factors not assessed in this study, such as tree vigor, precocity, yield, content of primary metabolites and minerals, resistance to diseases and pests, and market value of the fruit.