Pigs of the Polish native breed Złotnicka Spotted, compared to high-producing breeds, are characterized by a slower growth rate (from 340 to 590 g/day), greater carcass fatness (backfat thickness from 23.7 to 35 mm), good conversion of feed and, above all, very good meat quality. Compared to other breeds used in breeding herds in Poland their meat exhibits high intramuscular fat (from 2.2 to 3.4%) and protein content (from 22.3 to 25.2%). This meat is characterized by a darker color (L* from 46.4 to 50.5), high water holding capacity (from 18.6 to 32.7%) and excellent sensory quality (Szulc et al., 2024). The thickness of the backfat, typical for the breed, and the intramuscular fat content of the meat indicates the special suitability of the meat of the Złotnicka Spotted pigs for the production of traditional, regional, as well as long-matured products. So far, the potential of pigs of this breed in terms of meat quality traits has not been fully exploited. In Poland, the production of cured meats based on raw material from conserved breeds is negligible. This is due to the lack of demand for products containing more fat. Due to the confirmed correlation between the use of fatty products in the human diet, rich in saturated fatty acids, and lifestyle diseases such as obesity and atherosclerosis, consumers are increasingly looking for products with increased nutritional value and/or reduced energy value that fit the definition of functional foods (Baker et al., 2022). When choosing meat products, consumers are primarily guided by the organoleptic properties of the product, and pay little attention to the dietary quality of pork resulting largely from the feeding of fattening pigs. It is known that the components of the feed given to pigs in the final fattening period are a key factor in modulating the fatty acid composition of meat, including the ratio of n-6:n-3 polyunsaturated fatty acids, which can be better adapted to the requirements of a healthy human diet (Lebret and Čandek-Potokar, 2022). Modification of pig diets, in which, in addition to farm feeds, supplements are introduced in the form of, for example, green fodder (which is the case in the extensive management system of native breeds), increases the dietary and health-promoting values of pork meat (Szyndler-Nędza et al., 2021). The dietary value of meat can primarily be improved by adding flax seed to the feed, thereby increasing the level of PUFA n-3 polyunsaturated fatty acids valued by the consumer (Talwar et al., 2025). Found in flax seed, α-linolenic acid (ALA) is highly susceptible to oxidation. After ingestion, it is incorporated into the body’s lipid pool (particularly triglycerides and phospholipid membranes) and then endogenously metabolized through desaturation, elongation and peroxisome oxidation to eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), then beta-oxidized as an energy source or directly metabolized to C18-oxylipin (Takić et al., 2024). However, an excessive amount of PUFA in intramuscular or storage fat adversely affects the technological and qualitative, including sensory, properties of meat (faster rancidity) (Faustman et al., 2010). The addition of flax seeds to animal feed therefore requires the addition of antioxidants, such as vitamin E (tocopherol) (Pettigrew and Esnaola, 2001). Protection of meat fat from oxidation can also be realized by adding dried plants (herbs and fruits) rich in phenolic compounds with a broad spectrum of antioxidant activity to pig feed (Van Hung, 2015; Castrica et al., 2019), such as rosemary, thyme, oregano, sage and mint (Khojasteh et al., 2020).
A number of studies have analyzed the effect of supplementing various bioactive-rich plant additives primarily on fattening traits and health of pigs, to a small extent on the quality of their meat expressed by fatty acid profile and TBARS value (Zeng et al., 2015; Mahfuz et al., 2021; Radzikowski and Milczarek, 2022). In addition, these studies included pigs from genetic groups characterized by high production parameters. Therefore, this type of research should be continued, especially taking into account primitive breeds of pigs that are genetically different from high-producing breeds (Gurgul et al., 2018). Thus, the results of our study add to the knowledge of the effect of plant additives rich in polyphenolic compounds, on the quality of meat of porkers of the native Złotnicka Spotted breed.
The purpose of the study was to determine the effect of a complete feed mixture, containing dried herbs or fruits and flax seeds, used in the final fattening period of the Złotnicka Spotted pigs, on the quality of meat and fat in terms of physicochemical parameters, dietary value and susceptibility to oxidation.
Ethical review and approval was waived for this study, as according to Polish law, ethical approval is not required for services within the scope of the Act of 18 December 2003 on animal treatment facilities, as well as agricultural activities, including the rearing or breeding of animals, carried out in accordance with the provisions on the protection of animals, and activities that, in compliance with the practices of veterinary medicine, do not cause pain, suffering, distress, or permanent damage to the body of animals, to an extent equal to or more intense than a needle prick (Act on the Protection of Animals Used for Scientific and Educational Purposes, Legislative Decree 266/2015). The experiment was conducted according to the guidelines of the Declaration of Helsinki and in compliance with the European Union law (Directive 2010/63/EU, adopted in Poland by Legislative Decree 266/2015) of the European Parliament and of the Council on the protection of animals used for scientific or educational purposes.
The study included 50 fattening pigs (25 barrows, 25 gilts) of the Złotnicka Spotted breed with the known RYR1 genotype. The design of experiment is presented in Table 1.
Distribution of sex and RYR1 genotypes in each experimental group and final average slaughter weight of animals
| A | H | H+ | F | F+ | ||
|---|---|---|---|---|---|---|
| N | 10 | 10 | 10 | 10 | 10 | |
| Sex (no.) | barrows | 5 | 6 | 5 | 5 | 4 |
| gilts | 5 | 4 | 5 | 5 | 6 | |
| RYR (no.) | NN | 6 | 5 | 5 | 4 | 5 |
| Nn | 4 | 5 | 5 | 6 | 3 | |
| nn | – | – | – | – | 2 | |
| Body weight at slaughter (kg) | 136.4 | 139.9 | 136.3 | 138.1 | 139.95 | |
| Weight of right half-carcass (kg) | 52.3 | 54.1 | 52.3 | 53.3 | 54.1 |
During the fattening phase from 30 to 140 kg of body weight, all animals were fed the same grower and finisher diet (in which GM extracted soybean meal was replaced by rapeseed cake and faba bean). The experiment began in the second phase of fattening, i.e. from 90 kg to 140 kg body weight, and the animals were divided into 5 feeding groups of 10 animals each. The control group A was fattened with a complete finisher diet without additives, and the next 4 experimental groups received one of the dried herb mixtures or a mixture of the dried fruits, with or without the addition of extruded flax seeds to the mixed diet. The herbal additive (H) included dried rosemary, thyme, sage, hops and caraway seeds, the fruit additive (F) included dried rose, sea buckthorn, elderberry, rowanberry and caraway seeds. The plant raw materials, before being selected for this research, were analyzed for their nutrient and active ingredient content (Table 2), and their antioxidant properties were also tested (Figure 1) using the DPPH test.
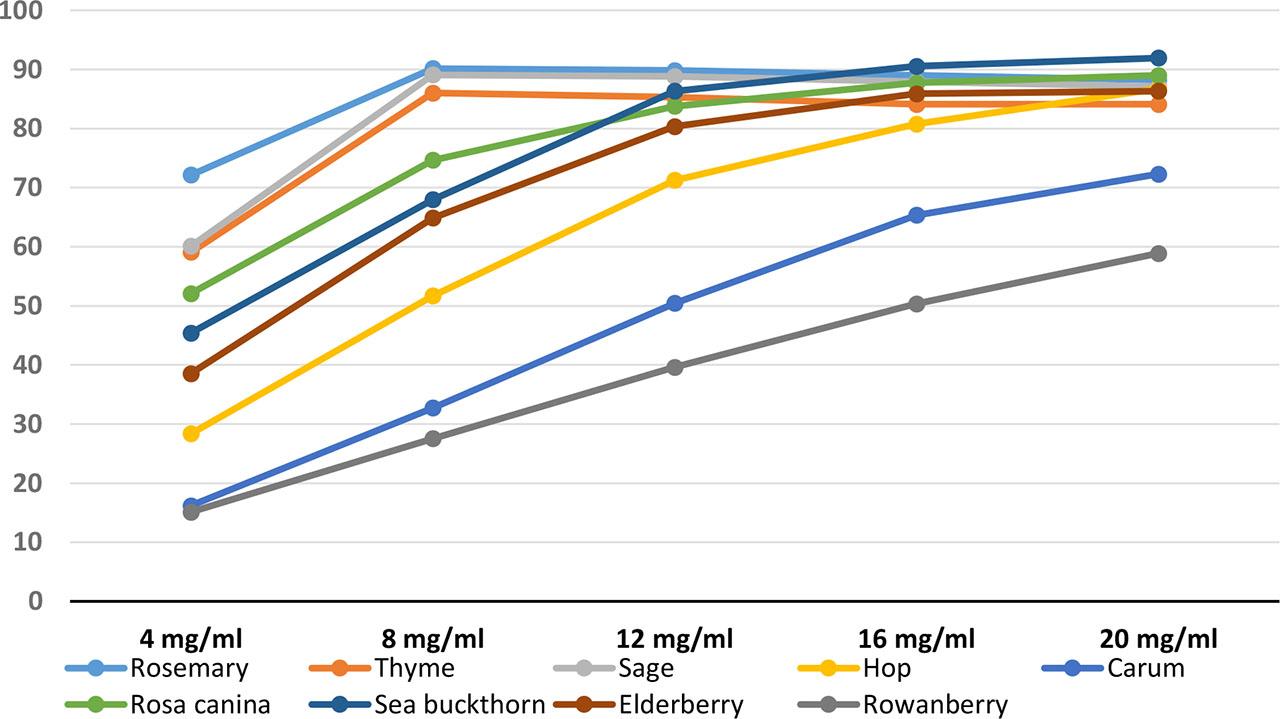
Percentage of reaction inhibition (%), i.e. the ability of the tested herbs and fruits dried raw materials to counteract the oxidation reaction with increasing sample concentration in DPPH test
Basic nutrient and bioactive components in raw dried herb and fruit materials
| Item | Rosemary | Thyme | Sage | Hop | Caraway seeds | Rosa canina | Sea buckthorn | Elderberry | Rowanberry |
|---|---|---|---|---|---|---|---|---|---|
| Nutrient contents | |||||||||
| dry matter (%) | 91.29 | 91.81 | 91.60 | 93.78 | 89.95 | 90.61 | 92.85 | 90.73 | 89.62 |
| crude protein (%) | 6.02 | 15.17 | 15.74 | 22.87 | 21.37 | 4.95 | 11.79 | 10.36 | 4.38 |
| crude fat (%) | 5.39 | 2.52 | 4.23 | 13.82 | 4.27 | 3.86 | 17.18 | 14.00 | 2.39 |
| crude fiber (%) | 19.82 | 15.26 | 20.38 | 18.49 | 28.34 | 36.75 | 9.64 | 18.29 | 7.62 |
| crude ash (%) | 4.93 | 11.87 | 11.03 | 5.73 | 4.75 | 4.18 | 3.37 | 4.40 | 2.91 |
| Polyphenol contents | |||||||||
| content of total hydroxycinnamic acid derivatives expressed as rosmarinic acid (%) | 6.50 | 4.76 | 3.54 | – | 0.86 | – | – | – | – |
| polyphenol content expressed as pyrogallol (%) | – | – | – | 0.65 | – | 2.67 | 0.94 | 0.58 | 0.23 |
| essential oil content (ml/kg) | – | – | – | – | 32.5 | – | – | – | – |
The basic chemical analysis (dry matter, crude protein, crude fat, crude fiber, crude ash) was conducted on dried herbs and fruits, used as feed additives, as well as in the feed mixtures, according to the AOAC (2009) methods. Amino acids were analyzed with an AAA 400 automatic analyzer (INGOS Ltd., Prague, Czech Republic), after hydrolysis in accordance with Commission Regulation (EC) no. 152/2009.
Analysis of content of total hydroxycinnamic acid derivatives expressed as rosmarinic acid (FP, 2008 a) was done using UV-VIS spectrophotometry in the raw materials rosemary, thyme, sage, and caraway seeds. Stock solution: the test samples were ground in a mill, weighed accurately, depending on the sample, from approximately 0.200 g to 0.3500 g, and placed in a 250 ml round-bottom flask. 80.0 ml of 50% (V/V) ethanol was added. This was boiled for 30 minutes in a water bath under a reflux condenser, allowed to cool and filtered into a 100.0 ml volumetric flask. The filter was rinsed with 10.0 ml of 50% ethanol, and then the flask topped up with 50% ethanol. Stock solution: The sample was poured into a 100.0 ml volumetric flask. 1.0 ml of the stock solution was taken, adding 2.0 ml of 0.5 mol/L hydrochloric acid, 2.0 ml of a solution prepared by dissolving 10.0 g of sodium nitrite and 10.0 g of sodium molybdate in 100.0 ml of demineralized water, then 2.0 ml of sodium hydroxide solution (8.5 g/100 ml) was added and topped up with demineralized water. Reference: 1.0 ml of the stock solution was taken in a 10.0 ml volumetric flask and topped up with demineralized water. The absorbance of the test solution was immediately measured against the reference at λ 505 nm. The percentage of total hydroxycinnamic acid derivatives converted to rosmarinic acid was calculated using the following formula:
Determination of total polyphenol and tannin content, expressed as pyrogallol (FP XI) involved using UV-Vis spectrophotometry in dried raw materials: hop, Rosa canina, sea buckthorn, elderberry, rowanberry. The test samples were ground in a mill, weighed accurately, depending on the sample, from approximately 1.0000 g to 5.0000 g, and placed in a 250 ml round-bottom flask. 150 ml of deionized water was added, then heated for 30 minutes in a water bath under a reflux condenser. This was cooled under running water and the contents transferred quantitatively to a 250 ml volumetric flask and topped up with deionized water. The precipitate was allowed to settle and the liquid filtered through a filter paper. The first 50 ml of filtrate was discarded. Total polyphenol content: 5.0 ml of filtrate was diluted to 25 ml with deionized water. Then 2 ml of this solution was transferred to a 25 ml volumetric flask. 1.0 ml of phosphomolybdotungstate reagent (Folin-Ciocalteu’s phenol reagent) and 10.0 ml of deionized water were added and topped up with 290 g/L sodium carbonate solution. After 30 min, the absorbance was measured at λ 760 nm (A1) using deionized water as a reference. Polyphenols not binding to leather powder (tannins): 0.10 g of leather powder was added to 10.0 ml of filtrate and shaken (on a shaker) for 60 min. 5.0 ml of the filtrate was filtered and diluted with demineralized water to 25.0 ml. Then it was proceeded as above (A2). Standard: Immediately before use, 50.0 mg of pyrogallol was dissolved in demineralized water and diluted with water to 100.0 ml. 5.0 ml of this solution was diluted with demineralized water to 100.0 ml, and then it was proceeded as for the test sample (A3). the percentage of polyphenols expressed as pyrogallol was calculated using the following formula:
The percentage of tannins expressed as pyrogallol was calculated using the following formula:
The prepared test solutions and the reference solution were analyzed by UV-Vis spectrometry on a GBC Cintra 20 spectrometer.
Determination of essential oil content in caraway seeds (FP, 2008 b) was carried out using method involving steam distillation in a special apparatus. The test sample was ground in a mill, 10.0 g weighed into a 500 ml round-bottom flask, adding 200.0 ml of deionized water as the distillation fluid and 0.50 ml of xylene in a calibrated tube. This was distilled for 90 minutes at a rate of 2–3 ml/min. The heat was turned off, and after waiting for 10 minutes the volume of liquid collected in the calibration tube was read by subtracting the volume of xylene added earlier. The difference indicated the amount of essential oil per mass of the sample.
Determination of the 2, 2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activity methods of DPPH inhibition developed by Huang et al. (2005), Katalinic et al. (2006) and Qian and Nihorimbere (2004) was adapted to the samples. Activity of the dried plant raw materials toward free radicals was measured at different levels of the extract concentration, which was prepared as a dilution of the sample solution with 40% ethanol. The amounts of 0.5–2.5 ml of the sample solution were filled up with 40% ethanol to the volume of 2.5 ml. The test solution was prepared in a tube with 3.9 ml of DPPH ethanolic solution (6×10−5 M) and 0.1 ml of the sample solution (in different concentrations). The test solution was mixed and kept in the dark. The absorbance of the test solution was measured after 30 min at λ=515 nm by comparison with 40% ethanol.
Fatty acid profile of the finisher diet was determined by gas chromatography using a Shimadzu GC-2010 Plus apparatus (Column Rtx2330, 105 m, 0.32 mm, 0.2 µm) according to P 015 issue 2 dated 01.03.2016. Samples were mixed, and lipids were extracted using a chloroform–methanol solution (2:1) (Folch et al., 1957). After evaporation under a stream of nitrogen and saponification (80°C, 0.5 N NaOH in methanol), the samples were subjected to methylation using a boron trifluoride/methanol reagent (ISO 12966-2:2017). Fatty acid methyl esters (FAMEs) were extracted with hexane and separated using a polar capillary column (105 m × 0.32 mm inner diameter, 0.2 µm film thickness, Rtx-2330; Restek) on a GC-2010 Plus gas chromatograph (Shimadzu). The temperature program was as follows: column temperature from 60 to 120°C at 20°C/min, then from 120 to 240°C at 3°C/min; injector temperatures were set at 250°C. The detector FID was operating in full scan mode. The temperature of the ion source was 250°C. Helium was used as the carrier gas. The analysis was performed using a standard fatty acid solution (Merck) processed in the same way as the samples. Results were expressed as grams per 100 grams of total fatty acids detected. The ingredients composition, nutrients content and fatty acids profile of the diets used in the experiment are presented in Table 3.
Formula, nutrient contents and fatty acids profile (g per 100 g of all estimated fatty acids) of the experimental finisher diets (90–140 kg body weight)
| Item | Experimental group | ||||
|---|---|---|---|---|---|
| A | H | H+ | F | F+ | |
| Components (%) | |||||
| wheat | 13.0 | 13.0 | 13.0 | 13.0 | 13.0 |
| barley | 39.58 | 37.58 | 36.56 | 37.58 | 36.56 |
| triticale | 13.0 | 13.0 | 13.0 | 13.0 | 13.0 |
| rye | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 |
| rapeseed press cake | 8.0 | 8.0 | 6.0 | 8.0 | 6.0 |
| low-tannin faba bean | 14.0 | 14.0 | 14.0 | 14.0 | 14.0 |
| monocalcium phosphate | 0.22 | 0.22 | 0.22 | 0.22 | 0.22 |
| ground limestone | 1.3 | 1.3 | 1.3 | 1.3 | 1.3 |
| salt | 0.22 | 0.22 | 0.22 | 0.22 | 0.22 |
| L-Lysine | 0.18 | 0.18 | 0.2 | 0.18 | 0.2 |
| mineral-vitamin premix | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| dried herbs (H) or fruits (F) | – | 2.0 | 2.0 | 2.0 | 2.0 |
| flax seeds | – | – | 3.0 | – | 3.0 |
| In 1 kg of feed mixture | |||||
| dry matter (%) | 880 | 881 | 880 | 881 | 881 |
| crude protein (%) | 147 | 147 | 147 | 146 | 146 |
| crude fat (%) | 26 | 27 | 37 | 27 | 38 |
| crude fiber (%) | 48 | 51 | 51 | 52 | 53 |
| crude ash (%) | 49 | 50 | 49 | 49 | 49 |
| metabolizable energy (MJ)* | 12.9 | 12.8 | 13.0 | 12.8 | 13.0 |
| Lys (g) | 8.30 | 8.35 | 8.40 | 8.31 | 8.39 |
| Met + Cys (g) | 5.13 | 5.12 | 5.10 | 5.11 | 5.09 |
| Tre (g) | 4.73 | 4.77 | 4.70 | 4.73 | 4.69 |
| Trp (g) | 1.98 | 1.99 | 1.99 | 1.98 | 1.98 |
| Ca (g) | 6.25 | 6.24 | 6.20 | 6.23 | 6.21 |
| P digestible (g) | 2.04 | 2.01 | 2.00 | 2.01 | 2.02 |
| Fatty acids profile | |||||
| C16:0 palmitic | 22.62 | 21.23 | 13.43 | 20.13 | 13.50 |
| C16:1 n-7 palmitoleic | 0.40 | 0.39 | 0.24 | 0.77 | 0.39 |
| C18:0 stearic | 1.98 | 2.10 | 3.07 | 2.23 | 3.11 |
| C18:1 n-9 oleic | 24.27 | 25.95 | 21.76 | 26.99 | 21.56 |
| C18:2 n-6 linoleic | 41.63 | 41.21 | 27.51 | 36.74 | 26.00 |
| C18:3 n-3 alpha-linolenic | 7.93 | 7.81 | 33.33 | 12.02 | 34.82 |
| C20:5 n-3 EPA | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| C22:6 n-3 DHA | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| SFA | 25.40 | 24.20 | 17.07 | 23.15 | 17.15 |
| UFA | 74.60 | 75.80 | 82.93 | 76.85 | 82.85 |
| MUFA | 24.82 | 26.49 | 22.07 | 27.88 | 22.02 |
| PUFA | 49.78 | 49.31 | 60.86 | 48.97 | 60.83 |
| PUFA n-6 | 41.84 | 41.49 | 27.53 | 36.95 | 26.01 |
| PUFA n-3 | 7.93 | 7.82 | 33.33 | 12.02 | 34.82 |
Metabolizable energy was calculated using equation from Hoffmann and Schiemann (1980).
All animals were slaughtered within 2 slaughter cycles (2 days). Slaughter occurred after the animals reached a body weight of 140 kg ± 5 kg. The same people always performed the slaughtering process and carcass dissection (the human factor had a statistically insignificant effect and was not used in the statistical model). The animals were subjected to the same procedure before slaughter. The day before slaughter, they were weighed and subjected to a 1-day fast. On the day of slaughter, they were transported about 100 m on transport carts to the slaughterhouse, which is part of the inspection station complex. Slaughter was performed using electric tongs (Koma, STZ-6i) with parameters consistent with applicable standards.
On the day of slaughter, measurements of the pH of the loin muscle and ham muscle were taken on the right half-carcasses. These measurements were made in the longissimus dorsi muscle between the last thoracic vertebra and the first lumbar vertebra, and in the middle part of the ham muscle.
After the carcasses had been refrigerated at 4°C for 24 hours, 150 g of longissimus lumborum (LLM) muscle samples were taken from the right half-carcasses from the section of the 3 thoracic vertebrae for meat color evaluation. Then, 300 g samples of semimembranosus muscle (SM) and of longissimus lumborum muscle (LLM) from the section behind the last rib were taken and frozen at −18°C and stored frozen for 2 months for chemical and meat texture analysis. A raw matured longissimus lumborum muscle (RMLLM) cured meat was made from the remainder of the muscle. This cured meat was made in the traditional way without the use of chemical enhancers, the production time was about 31 days. Dry curing of the meat with a curing and spice mixture (without aromatic spices) was performed. The first stage of meat maturation was carried out at 10–12°C and a relative humidity of about 75%. This was followed by a two-stage smoking in cold smoke and a second maturation at 10–12°C and a relative humidity of about 75% until a final yield of about 75–80% was achieved. Finished cured meat samples were vacuum-packed in foil and marked with the animal number and the symbol of the experimental group.
Quality parameters of meat (m. longissimuss lumborum; m. semimembranosus) were assessed:
Meat acidity was measured with a Matthäus pH Star pH-meter twice: 45 min after slaughter (pH45) and after 24 h of cooling at +4°C (pH24). Each time on the day of measurement, the device was calibrated using buffers with pH = 4.01 and pH = 7.0. The measuring device had automatic temperature compensation.
Color parameters (L, a, b) – meat color was assessed only on m. longissimus lumborum using the L*, a*, b* system with a Minolta CR-310 colorimeter (Japan). Source of light C, standard 100, aperture 50 mm, blooming time 1 sec. Lightness (L*), redness (a*), and yellowness (b*) were measured in fresh samples (24 h after slaughter).
Cooking loss (%). Frozen LLM and SM samples were thawed at room temperature for 17 h. Thawed LLM and SM muscle slices (weighing about 300 g) were cleaned of perimuscular membranes. Samples weighing 250 g were placed in a polyethylene bag, cooked in a water bath until the temperature inside the sample reached 80°C and cooled for 45 minutes at room temperature, then at 4°C for 24 hours. The samples were then weighed to determine cooking loss (%). Cooking loss (%) of LLM and SM meat was estimated from the difference between the weight of the sample before and after cooking and cooling.
Texture analysis of LLM and SM meat was carried out after thawing the samples at room temperature. Texture analysis was performed using texture analyzer TA-XTplus (Stable Micro Systems, Godalming, UK). Warner-Bratzler shear force (WBS; shear force and shear energy) analyses were performed on cooked meat. Texture analysis of the RMLLM samples was carried out after they were removed from the cooler and vacuum packaging. Seven cores (1.5 cm diameter cylinders) were taken from the muscle section parallel to the direction of the muscle fibers. Two cylinders, 1.5 cm in diameter and 3.5 cm in length, were cut using a triangular WB blade (4.5 mm/s) on a texture analyzer TA-XTplus. Peak shear force (N) and shear energy (N/sec), which is an indication of strength, were recorded. Five cylinders 1.5 cm in diameter and 2 cm in length were used to determine the texture profile (TPA). The cylinders were compressed twice by a cylinder probe (SMS P/25, base diameter 50 mm) to 70% of their height at a rate of 2 mm/sec with a 3-second interval between compression recordings. Texture parameters (TPA) such as hardness, cohesiveness, chewiness and springiness were calculated from force–deformation curves. Hardness (N) was defined as the maximum force, applied to the specimens during the first compression cycle, necessary to achieve a given deformation. Cohesiveness, or the strength of the internal bonds that form the product’s framework, was calculated as the ratio of the area under the second curve to the area under the first curve. Springiness (mm), or the rate of return from the deformed state to the original state, was determined as the ratio of the contact time with the sample during the second compression to the first compression. Resilience, or the ability of the product to return to its initial form after the first compression, was defined as the ratio of the negative input force to the positive input force during the first compression, while chewiness, or the energy required to grind (chew) a solid product, was obtained by multiplying hardness, cohesiveness and springiness (Meullenet et al., 1998). The resulting data were collected and calculated by Texture Expert software version 1.20.
Chemical analyses. Collected samples of raw and raw matured meat were stored for 2 months: raw meat (LLM, SM) at −18°C, and raw matured loins (RMLLM) at 4°C. Frozen meat samples were then thawed at room temperature for 17 h. A series of chemical analyses were carried out in the thawed LLM raw meat samples and the RMLLM loin. The content of crude protein and crude fat was determined according to the Official Method of Analysis of AOAC (2005) (18th edition; Method 981.10 Crude Protein in Meat; Method 950.46 Moisture in Meat; Method 991.36 Fat (Crude) in Meat and Meat Products). Meat lipid oxidation (TBARS) was determined by method P025 Issue 2, 18/12/2019 based on Pikul et al. (1989). The fatty acid profile of the meat and products was estimated using the same method as described above for the feed, except that the samples stored at −20°C were mixed, and the quadrupole electron ionization (70eV) mass spectrometer was operating in full scan mode. The determined fatty acids are expressed in g per 100 g of all determined acids. Crude protein, crude fat, TBARS and fatty acid profile were also determined in SM meat according to the methods described above.
Ratio calculations. Based on the fatty acid profile, the following indices were calculated (Σg/100 g):
AI (atherogenic index) = [C12:0+(4 × C14:0) + C16:0 + C18:0] / [n-6 PUFA + n-3 PUFA + MUFA] (Ulbricht and Southgate, 1991),
TI (thrombogenic index) = [C12:0 + C14:0 + C16:0 + C18:0] / [(0.5 × MUFA) + (0.5 × n-6 PUFA) + (3 × n-3 PUFA) + n-3 / n-6 PUFA] (Ulbricht and Southgate, 1991),
h/H (the hypocholesterolemic/hypercholesterolemic ratio) = (C18:1 + C18:2 + C18:3 + C20:4 + C20:5 + C22:6) / (C14:0 + C16:0) (Fernández et al., 2007),
OFA (odd fatty acids) = C15:0 + C17:0 + C17:1 (Díaz et al., 2002),
DFA (hypocholesterolemic fatty acids) = (UFA + C18:0).
An analysis of the effect of the experimental factor on the color, pH, texture, chemical composition and fatty acid profile and oxidative stability of meat and products was performed. Calculations were made based on analysis of variance using the GLM model according to the formula:
Normality of distribution was assessed using the Kolmogorov-Smirnov test, and Levene’s test of homogeneity of variance was used to test for equality of variance. Statistically significant differences between group means were determined at P≤0.05 using the LSD test. The software used for the analyses was STATISTICA version 12 (StatSoft, Inc., 2014, www.statsoft.com).
Analyzing the texture profile parameters (TPA) in LLM and SM meat, there was no effect of the tested feed additives on hardness, cohesiveness, chewiness and resilience (Table 4). Only in the case of LLM springiness was it shown that compared to the control group, the feed additive in all experimental groups increased the value of this parameter (P≤0.05).
Results of cooking loss. Warner-Bratzler shear force and texture profile analysis (TPA) of longissimus lumborum muscle, semimembranosus muscle and raw matured loin (longissimus lumborum muscle)
| Experimental group | |||||
|---|---|---|---|---|---|
| A | H | H+ | F | F+ | |
| Longissimus lumborum muscle (LLM) | |||||
| cooking loss (%) | 34.09±2.76 | 33.21±3.54 | 32.99±3.93 | 35.74±3.33 | 33.51±2.29 |
| shear force (N) | 52.72±14.69 | 53.18±11.84 | 50.72±9.72 | 56.00±13.83 | 49.67±12.32 |
| shear energy (N/sec) | 133.01±34.82 | 136.27±32.93 | 132.18±27.74 | 146.67±35.24 | 119.86±34.34 |
| texture profile TPA | |||||
| hardness (N) | 9.55±3.11 | 9.80±3.79 | 9.46±1.83 | 11.55±3.22 | 8.94±2.63 |
| springiness | 0.70±0.04 a | 0.74±0.03 b | 0.74±0.02 b | 0.73±0.03 b | 0.73±0.02 b |
| cohesiveness | 0.68±0.04 | 0.68±0.04 | 0.69±0.02 | 0.69±0.02 | 0.68±0.02 |
| chewiness | 4.62±1.26 | 5.11±2.50 | 4.91±0.97 | 5.95±1.61 | 4.50±1.38 |
| resilience | 0.31±0.02 | 0.31±0.03 | 0.31±0.02 | 0.32±0.02 | 0.31±0.01 |
| Semimembranosus muscle (SM) | |||||
| cooking loss (%) | 31.93±3.92 | 33.59±3.14 | 32.78±3.65 | 33.28±3.54 | 33.56±2.63 |
| shear force (N) | 53.21±17.67 | 56.42±18.55 | 50.98±15.95 b | 51.92±21.61 b | 73.11±31.98 a |
| shear energy (N/sec) | 138.84±35.68 | 145.83±38.46 | 134.16±44.95 | 143.33±40.74 | 167.85±70.92 |
| texture profile TPA | |||||
| hardness (N) | 6.64±3.32 | 7.28±2.08 | 6.91±3.30 | 6.98±2.70 | 6.76±2.14 |
| springiness | 0.73±0.03 | 0.76±0.03 | 0.75±0.03 | 0.74±0.06 | 0.73±0.06 |
| cohesiveness | 0.63±0.05 | 0.66±0.03 | 0.66±0.05 | 0.65±0.04 | 0.63±0.05 |
| chewiness | 3.28±1.77 | 3.72±1.20 | 3.44±1.64 | 3.37±1.21 | 3.08±0.82 |
| resilience | 0.28±0.04 | 0.29±0.03 | 0.29±0.04 | 0.28±0.02 | 0.27±0.02 |
| Raw matured longissimus lumborum muscle (RMLLM) | |||||
| shear force (N) | 56.89±26.36 | 56.34±20.82 | 51.65±18.86 | 56.76±15.43 | 44.14±17.61 |
| shear energy (N/sec) | 137.68±69.07 | 140.20±53.42 | 133.55±54.74 | 139.44±45.76 | 109.87±49.41 |
– means in rows with different letters differ significantly at P≤0.05.
No statistically significant differences were found in the cooking loss of LLM and SM meat between the analyzed experimental groups (Table 5). When analyzing the shear force and shear energy values of LLM and RMLLM meat, there were also no statistically significant differences between the analyzed experimental groups. Only in the case of SM was it shown that this product from fatteners in the H+ and F groups had statistically significantly lower shear force (P≤0.05), but only compared to SM from fatteners in the F+ group.
Acidity and nutritional value and susceptibility to oxidation of raw loin (longissimus lumborum muscle), ham (semimembranosus muscle) and raw matured loin (raw matured longissimus lumborum muscle) meat from Złotnicka Spotted pigs
| Experimental group | |||||
|---|---|---|---|---|---|
| A | H | H+ | F | F+ | |
| Longissimus lumborum muscle (LLM) | |||||
| L* | 48.09±4.85 | 47.8±4.98 | 50.16±5.09 | 49.9±6.23 | 48.36±2.60 |
| a* | 19.53±0.89 | 19.77±1.81 | 19.88±1.98 | 20.08±2.20 | 19.89±1.25 |
| b* | 1.88±1.20 | 2.28±1.48 | 3.33±1.81 | 2.94±2.03 | 2.1±1.71 |
| pH45 | 6.48±0.03 | 6.53±0.07 | 6.52±0.10 | 6.54±0.07 | 6.51±0.07 |
| pH24 | 5.46±0.05 b | 5.49±0.06 | 5.53±0.09 a | 5.51±0.07 | 5.46±0.04 b |
| protein (%) | 23.33±1.18 | 23.04±0.77 | 23.27±0.55 | 23.70±0.66 | 23.44±0.72 |
| fat (%) | 3.54± 1.71 a | 3.33±1.34 a | 2.76±0.85 | 2.30±0.45 b | 2.28±0.77 b |
| TBARS (mg/kg) | 0.364±0.09 | 0.354±0.12 | 0.374±0.10 | 0.319±0.13 | 0.379±0.13 |
| Semimembranosus muscle (SM) | |||||
| pH45 | 6.36±0.04 b | 6.40±0.05 | 6.40±0.05 | 6.41±0.04 a | 6.40±0.02 |
| pH24 | 5.59±0.04 | 5.42±0.05 | 5.41±0.03 | 5.41±0.03 | 5.40±0.03 |
| protein (%) | 22.50±0.87 | 22.71±0.36 | 22.44±0.71 | 22.63±0.72 | 22.81±0.72 |
| fat (%) | 2.61±0.85 a | 1.76±0.38 b | 2.34±1.37 a | 1.65±0.26 b | 1.58±0.21 b |
| TBARS (mg/kg) | 0.350±0.09 | 0.322±0.13 | 0.333±0.07 | 0.284±0.12 | 0.338±0.08 |
| Raw matured longissimus lumborum muscle (RRLLM) | |||||
| protein (%) | 31.58±1.24 | 31.88±1.76 | 31.58±2.27 | 32.27±2.12 | 32.48±2.91 |
| fat (%) | 4.17±2.10 | 5.39±2.24 a | 3.03±0.87 b | 4.47±2.29 | 3.23±1.07 b |
| TBARS (mg/kg) | 0.944±0.37 b | 1.043±0.34 | 1.109±0.32 | 1.138±0.46 a | 1.192±0.28 a |
– means in rows with different letters differ significantly at P≤0.05.
There was no effect of the tested feed additives on the color (L*, a*, b*) of raw LLM (Table 5). However, it was shown that the SM of the fattening pigs in the F experimental group, compared to the control group, had higher pH45 values. On the other hand, the LLM of fatteners in the H+ group showed higher pH24 values (P≤0.05) compared to both the control and experimental F+ groups.
Analyzing the nutrients of raw and raw matured meat, it was found that there was no effect of feed additives on protein content (P>0.05) in each of the products analyzed (Table 5). In the case of fat content (Table 5), it was shown that the use of additives in finisher diet contributed to a statistically significant reduction in fat content primarily in SM in the H, F and F+ experimental groups (A>H, F, F+; P≤0.05). In the case of LLM, the reduction in fat content, compared to the control and experimental group H, was influenced by dried fruits and flax seeds used in the F and F+ groups (F, F+<A, H; P≤0.05). Feed additives had no effect on the meat fat content of long-matured RMLLM loin (relative to the control group), while the highest fat content was in RMLLM from fattening pigs in the H group, which was statistically significantly higher only compared to the H+ and F+ groups (P≤0.05).
In raw LLM and SM meat stored frozen for 2 months, there was no significant effect of feed additives on the color reaction of 2-thiobarbituric acid with malondialdehyde formed during fat oxidation (TBARS). In contrast, a significant effect of dried fruits in the F and F+ groups was demonstrated in the case of loin subjected to maturation. RMLLMs from fattening pigs fed with dried fruits (F) and dried fruits and flax seeds (F+) had the highest TBARS, which was significantly higher compared to control group A (A<F, F+; P≤0.05).
Table 6 shows the effect of feed additives on the fatty acid profile in LLM, SM and RMLLM. Most differences in fatty acid profile were found in SM. Compared to the control group, feeding diet with dried fruits (F and F+) and dried herbs (H) had a significant (P≤0.05) effect on reducing the content of MUFAs and increasing the content of PUFAs, including PUFA n-6 in SM. The lower MUFA content was due to a lower content of C16:1 n-7 and mainly C18:1 n-9 in the groups fed with dried fruit (F, F+) diet (by 4.9 and 4.4 g/100 g, respectively) and in the group receiving dried herbs (H) by 3.3 g/100 g. PUFA n-6 content was increased by increasing primarily C18:2 n-6 in the groups receiving dried fruits (by 4.3 and 3.3 g/100 g, respectively) and in the herbal group (H) by 3.08 g/100 g. It was also shown that feeding diet with flax seeds (F+, H+) significantly increased the content of PUFA n-3 acids in SM. At the same time, from among these groups, the highest content of PUFA n-3 was established in SM of fattening pigs from the F+ group (F+>H+>F> H, A; P<0.05). The higher content of these acids in the F+ and H+ groups was due to a significantly higher content of C18:3 n-3 and C20:5 n-3 (EPA) acids. In contrast, the higher content of PUFA n-3 in the F group resulted from a significantly higher content of C22:6 n-3 (DHA) and C20:5 n-3 (EPA).
Profile of selected fatty acids in longissimus lumborum muscle, semimembranosus muscle and raw matured longissimus lumborum muscle (g per 100 g of all estimated fatty acids)
| Experimental group | |||||
|---|---|---|---|---|---|
| A | H | H+ | F | F+ | |
| Longissimus lumborum muscle (LLM) | |||||
| C16:0 palmitic | 25.519±1.24 | 25.423±0.88 | 25.195±0.75 | 25.125±0.66 | 24.761±0.87 |
| C16:1 n-7 palmitoleic | 3.000±0.59 a | 3.142±0.55 a | 2.688±0.30 | 2.711±0.29 | 2.536±0.32 b |
| C18:0 stearic | 15.463±1.76 | 14.662±1.14 b | 15.434±0.75 | 15.302±0.67 | 15.557±0.88 a |
| C18:1 n-9 oleic | 44.981±2.94 | 45.922±1.91 a | 43.991±2.48 | 44.115±2.11 | 43.519±3.46 b |
| C18:2 n-6 linoleic | 6.497±2.36 | 6.440±1.38 | 7.726±2.11 | 7.921±1.55 | 8.298±2.11 |
| C18:3 n-3 alpha-linolenic | 0.204±0.04 b | 0.258±0.15 b | 0.655±0.17 c | 0.333±0.09 b | 0.852±0.24 a |
| C20:5 n-3 EPA | 0.049±0.04 c | 0.057±0.04 c | 0.140±0.08 b | 0.086±0.04 c | 0.199±0.08 a |
| C22:6 n-3 DHA | 0.030±0.02 | 0.027±0.01 a | 0.037±0.02 | 0.046±0.03 b | 0.046±0.02 b |
| SFA | 42.513±2.34 | 41.523±1.45 | 42.033±1.19 | 41.806±1.17 | 41.689±1.65 |
| UFA | 57.487±2.34 | 58.477±1.45 | 57.967±1.19 | 58.194±1.17 | 58.311±1.65 |
| MUFA | 48.869±3.11 | 49.962±2.28 a | 47.506±2.45 b | 47.646±2.18 b | 46.844±3.50 b |
| PUFA | 8.618±3.53 b | 8.515±2.03 b | 10.461±3.03 | 10.549±2.17 | 11.467±3.08 a |
| PUFA n-6 | 8.299±3.44 | 8.129±1.98 | 9.527±2.86 | 10.025±2.09 | 10.250±2.77 |
| PUFA n-3 | 0.318±0.09 c | 0.385±0.20 c | 0.934±0.26 b | 0.524±0.15 c | 1.217±0.35 a |
| Semimembranosus muscle (SM) | |||||
| C16:0 palmitic | 24.167±0.91 a | 23.452±0.70 | 23.836±0.89 | 23.403±0.66 b | 23.258±0.94 b |
| C16:1 n-7 palmitoleic | 2.593±0.38 a | 2.407±0.50 a | 2.286±0.24 | 2.029±0.25 b | 2.050±0.33 b |
| C18:0 stearic | 14.201±1.12 b | 14.211±1.14 b | 14.762±0.64 | 14.771±0.36 | 15.049±0.89 a |
| C18:1 n-9 oleic | 42.446±3.87 a | 39.169±3.82 b | 40.164±3.16 | 37.512±2.29 b | 38.004±3.02 b |
| C18:2 n-6 linoleic | 10.862±2.90 b | 13.942±3.14 ac | 12.459±3.08 bc | 15.175±1.65 a | 14.197±1.86 ac |
| C18:3 n-3 alpha-linolenic | 0.348±0.07 c | 0.453±0.27 c | 1.001±0.23 b | 0.570±0.14 c | 1.326±0.36 a |
| C20:5 n-3 EPA | 0.079±0.04 c | 0.140±0.10 cd | 0.240±0.09 b | 0.189±0.08 bd | 0.385±0.09 a |
| C22:6 n-3 DHA | 0.040±0.02 b | 0.065±0.02 cd | 0.058±0.03 bd | 0.090±0.04 a | 0.083±0.03 ac |
| SFA | 39.782±1.63 | 38.939±1.39 | 39.894±1.34 | 39.409±0.84 | 39.530±1.57 |
| UFA | 60.218±1.63 | 61.061±1.39 | 60.106±1.34 | 60.591±0.84 | 60.470±1.57 |
| MUFA | 45.951±3.91 a | 42.432±4.24 bc | 43.303±3.27 ab | 40.332±2.42 c | 40.834±3.20 bc |
| PUFA | 14.267±4.06 c | 18.629±4.11 ab | 16.803±4.20 bc | 20.259±2.13 a | 19.636±2.73 ab |
| PUFA n-6 | 13.746±3.95 c | 17.909±3.95 ab | 15.363±4.07 bc | 19.323±2.06 a | 17.671±2.51ab |
| PUFA n-3 | 0.521±0.12 d | 0.720±0.41 cd | 1.440±0.32 b | 0.936±0.23 c | 1.965±0.43 a |
| Raw matured longissimus lumborum muscle (RMLLM) | |||||
| C16:0 palmitic | 25.290±0.98 | 24.973±0.76 | 24.875±0.51 | 24.815±0.60 | 24.917±0.99 |
| C16:1 n-7 palmitoleic | 2.980±0.50 a | 3.056±0.44 a | 2.738±0.29 | 2.629±0.33 b | 2.519±0.31 b |
| C18:0 stearic | 15.472±1.40 | 14.982±1.12 | 15.056±0.79 | 15.529±0.84 | 15.784±0.89 |
| C18:1 n-9 oleic | 45.177±2.13 | 45.979±1.69 a | 45.154±2.12 | 43.926±2.94 b | 44.300±2.38 |
| C18:2 n-6 linoleic | 6.596±2.25 b | 6.681±1.14 b | 7.413±1.34 | 8.193±2.31 a | 7.572±1.58 |
| C18:3 n-3 alpha-linolenic | 0.201±0.03 d | 0.287±0.15 cd | 0.616±0.14 b | 0.417±0.15 c | 0.774±0.24 a |
| C20:5 n-3 EPA | 0.048±0.03 c | 0.050±0.03 c | 0.129±0.05 a | 0.091±0.03 b | 0.158±0.06 a |
| C22:6 n-3 DHA | 0.031±0.02 bc | 0.026±0.01 c | 0.040±0.01 ab | 0.042±0.02 a | 0.039±0.02 ab |
| SFA | 42.224±2.00 | 41.359±1.41 | 41.244±0.81 | 41.706±1.14 | 42.008±1.74 |
| UFA | 57.776±2.00 | 58.641±1.41 | 58.756±0.81 | 58.294±1.14 | 57.992±1.74 |
| MUFA | 49.049±2.26 | 49.976±2.06 a | 48.716±2.13 | 47.401±3.06 b | 47.670±2.40 b |
| PUFA | 8.727±3.22 b | 8.666±1.73 b | 10.040±1.87 | 10.893±2.95 a | 10.321±2.40 |
| PUFA n-6 | 8.411±3.14 | 8.255±1.69 b | 9.167±1.73 | 10.277±2.95 a | 9.242±2.06 |
| PUFA n-3 | 0.316±0.08 c | 0.411±0.18 bc | 0.873±0.20 a | 0.616±0.18 b | 1.079±0.33 a |
– means in rows with different letters differ significantly at P≤0.05.
Analyzing the fatty acid profile of LLM, it was found that, compared to the control group, feeding fatteners with diet supplemented with F+, significantly increased the content of PUFA fatty acids in this product primarily (by 2.85 g/100 g), including PUFA n-3 acids (by 0.9 g/100 g). A statistically significantly higher (by 0.6 g/100 g) content of PUFA n-3 fatty acids in LLM, compared to the control group, was also noted when fatteners were fed H+ diet. The high content of PUFA n-3 acids in the F+ and H+ groups was mainly due to the high content of C18:3 n-3 and C20:5 n-3 acid (EPA), which was statistically significantly higher compared to the other experimental groups. Feeding the other feed additives (H, F) had no statistical effect on the change in the fatty acid profile of LLM with respect to the control group. In addition, it was found that the LLM of the fattening pigs fed the dried herbs supplement (H) was characterized by the highest content of MUFA fatty acids. However, this value was statistically significantly higher only compared to the other experimental groups (H>H+, F, F+; P≤0.05). The high content of MUFA fatty acids in LLM from the H group was mainly due to the higher content of C16:1 n-7 and C18:1 n-9 monounsaturated fatty acids, which were statistically significantly higher compared to the F+ experimental group (P≤0.05).
As in LLM so in RMLLM, it was noticed that, compared to the control group, RMLLM from pigs fed flax-seed-supplemented diet had a statistically significantly higher content of PUFA n-3 acids (H+, F+>A, H, F; P≤0.05). In addition, it was demonstrated that feeding with the addition of F also affected, although to a lesser extent, the increase in the content of PUFA n-3 fatty acids, but this difference was significant only compared to the control group (F>A, P≤0.05). The high content of PUFA n-3 acids in RMLLM from the F+, H+ and F groups was mainly due to the higher content of C18:3 n-3 and C20:5 n-3 acid (EPA) (P≤0.05). It was also shown that RMLLM from fattening pigs fed dried fruits (F) was characterized by a statistically significantly higher content of total PUFA fatty acids (F>A, H; P≤0.05), including a statistically significantly higher content of C18:2 n-6 and C22:6 n-6 polyunsaturated fatty acids (DHA) (F>A, H; P≤0.05).
For the values of dietary indices, the most statistically significant differences between the analyzed groups were found in raw meat (Table 7). Ham meat (SM) from fattening pigs fed H, F and F+, compared to the control group, had a statistically significantly higher PUFA/SFA ratio (P≤0.05) and higher DFA content (P≤0.05). Both SM and LLM from pigs receiving feed with flax seeds (H+ and F+) were characterized by the lowest PUFA n-6/n-3 ratio (H+, F+<F<A, H; P≤0.05) and lower TI value (F+, H+<A; P≤0.05). The addition of dried fruits with flax seeds further increased the h/H ratio in SM (F+>A; P≤0.05). In the case of DFA content and PUFA/SFA ratio, it was shown that LLM from fatteners in the F+ group had a higher value of these indices compared to the control group (P≤0.05). There was also a significant effect of H+, F+ and F feed additives on the reduction of PUFA n-6/n-3 fatty acid ratio and TI values in RMLLM (H+, F+, F<A; P≤0.05). For the other values of dietary indices in RMLLM, there was no statistically significant effect of the factor of the tested feed additives.
Dietetic value of meat of longissimus lumborum muscle, semimembranosus muscle and raw matured longissimus lumborum muscle
| Experimental group | |||||
|---|---|---|---|---|---|
| A | H | H+ | F | F+ | |
| Longissimus lumborum muscle (LLM) | |||||
| PUFA/SFA | 0.205±0.09 b | 0.206±0.05 b | 0.250±0.08 | 0.253±0.06 | 0.275±0.07 a |
| PUFA n-6/n-3 | 25.388±4.29 a | 23.623±6.73 ab | 10.726±3.94 c | 19.761±3.46 b | 8.512±1.13 c |
| index AI | 0.786±0.08 | 0.752±0.05 | 0.764±0.04 | 0.756±0.04 | 0.752±0.05 |
| index TI | 1.425±0.14 a | 1.358±0.08 a | 1.323±0.07 b | 1.357±0.07 a | 1.276±0.09 b |
| ratio h/H | 2.016±0.19 a | 2.055±0.12 | 2.073±0.11 | 2.088±0.10 | 2.132±0.14 b |
| DFA | 72.949±1.41 a | 73.139±0.98 | 73.401±0.85 | 73.496±0.71 | 73.868±0.97 b |
| OFA | 0.216±0.05 | 0.224±0.03 | 0.216±0.03 | 0.216±0.03 | 0.203±0.02 |
| Semimembranosus muscle (SM) | |||||
| PUFA/SFA | 0.360±0.11 c | 0.479±0.11 ab | 0.424±0.12 bc | 0.514±0.05 a | 0.497±0.07 ab |
| PUFA n-6/n-3 | 26.186±3.27 ab | 28.252±7.54 a | 11.129±4.54 c | 21.351±3.61 b | 9.232±1.72 c |
| index AI | 0.695±0.05 | 0.664±0.04 | 0.694±0.04 | 0.675±0.02 | 0.680±0.05 |
| index TI | 1.249±0.09 a | 1.187±0.07 ab | 1.168±0.07 b | 1.189±0.04 ab | 1.107±0.07 c |
| ratio h/H | 2.248±0.15 b | 2.366±0.12 | 2.292±0.14 | 2.367±0.10 | 2.381±0.16 a |
| DFA | 74.420±1.00 b | 75.272±0.75 a | 74.868±0.97 | 75.362±0.71 a | 75.519±1.01 a |
| OFA | 0.308±0.06 | 0.341±0.07 a | 0.298±0.05 | 0.307±0.07 | 0.282±0.04 b |
| Raw matured longissimus lumborum muscle (RMLLM) | |||||
| PUFA/SFA | 0.209±0.08 | 0.210±0.04 | 0.243±0.04 | 0.261±0.07 | 0.247±0.06 |
| PUFA n-6/n-3 | 25.910±3.92 a | 21.777±5.16 ac | 10.932±3.09 b | 17.840±5.91 c | 9.380±3.98 b |
| index AI | 0.774±0.07 | 0.746±0.04 | 0.738±0.02 | 0.752±0.04 | 0.762±0.06 |
| index TI | 1.408±0.12 a | 1.346±0.07 | 1.289±0.05 b | 1.341±0.06 b | 1.308±0.09 b |
| ratio h/H | 2.047±1.17 | 2.098±0.10 | 2.131±0.7 | 2.119±0.10 | 2.109±0.15 |
| DFA | 73.247±1.13 | 73.623±0.80 | 73.812±0.55 | 73.823±0.65 | 73.775±1.09 |
| OFA | 0.210±0.06 | 0.231±0.02 a | 0.204±0.02 | 0.216±0.03 | 0.194±0.02 b |
– means in rows with different letters differ significantly at P≤0.05.
The color and aroma of raw meat are the parameters on which consumers first form their opinion of the quality of this product. The attractive visual appearance of pork is often associated with freshness, flavor and overall good quality, while defects in meat color are often perceived by consumers as signs of spoilage and unhealthiness. The color of raw meat is influenced by many factors (ranging from the genotype of the pigs, the housing and feeding system, to the handling of the animals before slaughter, to post-slaughter and storage conditions of the meat), which can determine the final color of the meat and its perception by consumers (Gagaouai et al., 2023). In our study, there was no effect of the feed additives used on the change in meat color (L*, a*, b*) determined 24 hours after slaughter. Similar results were obtained in studies where feed additives were used in the form of a mixture of herbs, oil (rapeseed and flaxseed) with the addition of grapes or pepper (Hanczakowska et al., 2015, 2017; Cheng et al., 2017; Bertol et al., 2017; Sampath et al., 2020). In contrast, Zhou et al. (2013) showed that the addition of Chinese herbs did not change the meat’s lightness (L*) and redness (a*), but only reduced yellowness (b*). Zhang and Kim (2022) showed that the ration of the feed additive used was significant in changing the redness of meat, as it was observed that as the amount of milk thistle fed increased, the a* value of meat color increased linearly.
Cooking loss and texture of cooked meat are other important parameters for the consumer. Too much cooking loss and meat that is too tough will discourage the consumer from making another purchase of this type of product. The water holding capacity of meat and cooking loss after cooking are closely related to the acidity level of raw meat (pH). This indicator is the basis for identifying good quality meat (RFN – red, firm, normal) and its PSE (pale, soft, exudative) or DFD (dark, firm, dry) defects (Lebret and Čandek-Potokar, 2022). A PSE defect occurs when the pH of the meat after slaughter has dropped too low (below 5.5) and too quickly, a DFD defect occurs when the pH of the meat remains above 6.0. These defects in the consumer’s perception will be identified by the color, drip loss and texture of the raw meat. The feed additives used in our study, containing dried herbs or fruits and flax seeds, had no effect on the cooking loss values of meat, nor on the Warner-Bratzler shear force parameters of all products analyzed (LLM, SM, RMLLM). The lack of effect of feed additives on meat cooking loss values and meat toughness is consistent with the results of other authors who added oregano (Cheng et al., 2017), pepper (Sampath et al., 2020) or Chinese herbs to the feed (Zhou et al., 2013). On the other hand, Zhang and Kim (2022) demonstrated that cooking loss of meat decreased with increasing the proportion of milk thistle in the feed. As for the meat texture profile (TPA), our study showed a significant increase only in the springiness of cooked LLM in all experimental groups. In a study by other authors, the addition of oregano oil to the feed increased tenderness of the meat (Cheng et al., 2017), while the addition of a mixture of herbs had no statistically significant effect on this parameter (Hanczakowska et al., 2015). In the experiment in question, 45 minutes after slaughter, a higher pH was found in the SM ham meat of fatteners fed with dried herb diet (F), while 24 hours after slaughter in the LLM loin meat of fatteners fed with dried herb diet and flax seeds (H+). However, with respect to the control group, these differences were small (about 1%). All the obtained pH values of loin and ham meat, determined 45 minutes and 24 hours after slaughter, indicate that the products from the pigs included in the study met the standards of good quality meat (RFN). The results of studies by other authors indicate that the pH of meat was not significantly affected by the feed additives used (Cheng et al., 2017; Fang et al., 2016; Hanczakowska et al., 2015; Sampath et al., 2020; Zhang and Kim, 2022). Some authors have also reported that it is rather the amount of additive used in the mixed feed that may be more important in shaping this parameter (Hanczakowska et al., 2017; Zhou et al., 2013).
A diet rich in polyunsaturated fatty acids (e.g., flax seeds) can be reflected in a higher unsaturation index of fat in the body’s tissues, which can lead to reduced oxidative stability and cause flavor defects, both in fat and in meat. The magnitude of the flavor change depends on the polyunsaturated fatty acid composition, iron content and pH of these tissues (Amaral et al., 2018). Lipid oxidation is one of the main factors responsible for the deterioration of meat and meat products. When lipids are oxidized, a number of unpleasant tastes and odors develop, as well as changes in color and texture. This is a rather complex process involving unsaturated fatty acids that react with molecular oxygen to form peroxides, which produce aldehydes, ketones and acids, many of which are responsible for the unpleasant, rancid odor of oxidized fats (Byrne et al., 2000; Estevez and Cava, 2004). It is difficult to unequivocally determine the limit above which fat oxidation will cause the consumer to reject meat. This difficulty is due to the variability in the perception of different consumers. It is assumed that the limit value of TBARS (thiobarbituric acid reactive substances), expressed in mg malondialdehyde (MDA) per kg of meat and at the same time the limit of positive sensory evaluation of meat is 2 mg MDA/kg (Campo et al., 2006). The feed additives used in our study, in the form of 2% dried herbs (H) or 2% dried fruits (F), did not significantly reduce the oxidation of fats in meat, either during frozen storage or during 31-day maturation. On the other hand, the addition of 3% flax seeds (H+, F+) to the feed with the mentioned additives (H and F) did not increase the susceptibility of raw meat to oxidation. In fact, it was found, compared to the control group, that there were no significant differences in TBARS values in all analyzed samples of LLM raw meat, including those from groups fed simultaneously dried herbs and flax seeds (H+ and F+). The TBARS values obtained for raw meat were comparable to those obtained in raw pork stored at 0°C for 3 days (Faustman et al., 2010). In the case of RMLLM, there were no significant differences in TBARS values in the products of the groups fed dried herbs (A=H and H+; P>0.05), indicating an antioxidant effect of the herbs, and a significantly higher TBARS value in the groups fed dried fruits (A<F and F+; P≤0.05). The difference between the experimental groups is likely due to the lower content of polyphenols in the dried fruits, compared to the dried herbs. It should be noted that the observed increase in TBARS values in RMLLM did not exceed the limit of meat sensory acceptability. The lesser usefulness of fruit additives in preventing fat oxidation processes during storage of raw meat at 0°C was also demonstrated by Bertol et al. (2017). In their study, they found that the addition of grape pomace to pig feed enriched with flaxseed oil had no effect on lowering the TBARS value of meat. This is in contrast to the plant additives oregano and milk thistle (Cheng et al., 2017; Zhang and Kim, 2022), where positive meat oxidative stability results were demonstrated. In the case of pork culinary treatment, it was shown that the simultaneous use of an additive of grape peel extract and oregano in a mixed feed containing extruded flax seeds reduced the amount of lipid oxidation products (hydroperoxides and TBARS) in grilled pork, as well as in in vitro digested meat (Martini et al., 2020).
The nutritional value of pork refers to its composition including protein with essential amino acids (AA), fat with essential fatty acids (especially polyunsaturated), and health-promoting compounds, among others vitamins, minerals and antioxidants (Lebret and Čandek-Potokar, 2022). In the experiment presented here, the pig feed additives used did not affect the protein content of LLM, SM, or RMLLM raw meat, which is in line with the results of Cheng et al. (2017), Fang et al. (2016), Zhang and Kim (2022). The protein content of LLM was typical of the Złotnicka Spotted pigs (Szulc et al., 2024). However, the addition of dried fruits (F, F+) was noticed to reduce the fat content of LLM and SM raw meat, as well as the addition of dried herbs (H) to reduce the fat content of SM. In the absence of any effect of herbal additive (H) on fat content in LLM, our results are in line with Cheng et al. (2017) and Zhang and Kim (2022). In contrast, Tognocchi et al. (2023), analyzing the effect of supplementing Large White pig feed with flax seed showed that this additive in the amount of 8% had an effect on reducing the amount of fat, but only in the lean part of the ham and bacon, while in the other tissues analyzed (fat, Boston shoulder, fatty part of the ham) they found no such effect
The diet of pigs has a significant effect primarily on the fatty acid profile of the fat (Lebret, 2008; Wood et al., 2008). The most pronounced changes in the fatty acid profile produced by nutrition can be seen in the ham muscle, as this muscle contains a higher amount of phospholipids derived from red fibers, which are more susceptible to dietary factors (Gandemer, 2002; Wood et al., 2003). In our study, the use of both 2% herb (H) and 2% dried fruit (F) additives in the diet of Złotnicka Spotted pigs primarily increased the content of PUFAs (including n-6 PUFAs) and reduced the MUFA content of raw ham meat. No such effect of herb addition was observed in LLM and RMLLM meat. Only an increase in PUFA content (including C18:2 n-6) was observed in RMLLM from pigs fed dried fruit supplement. Fang et al. (2016), analyzing the effect of adding fresh apple pomace to pig feed mixtures on the fatty acid profile of backfat, showed, similarly to our study, an increase in PUFA content (including C18:2 n-6). When a Chinese herb blend extract was applied at both 0.5 g/kg and 1 g/kg of the blend, Zhou et al. (2013) demonstrated, contrary to our results, a reduction in SFA content in LLM. Increasing the amount of herbal extract to 1 g/kg further increased the UFA in the LLM of these pigs. Also in a study by Hanczakowska et al. (2017), the application of hops in the pig feed at 500 mg or 1000 mg/kg of the mixture decreased SFA and increased UFA, including an increase in PUFA n-3 in LLM meat. In the experiment presented here, a greater effect of feeding in the last phase of fattening on the fatty acid profile of fattening pig meat was found when 3% flax seed was added to the mixed feeds. Meat of SM, LLM and RMLLM from fatteners fed a mixture with the addition of dried herbs and flax seeds (H+) was characterized by increased PUFA n-3 content, while the addition of dried fruits and flax seeds (F+) increased PUFA content (including PUFA n-3) in SM and LLM and PUFA n-3 in RMLLM. The increase in PUFA n-3 content in all analyzed products was mainly due to an increase in the amount of C18:3. This result is as expected, because, as shown in other studies, the use of extruded flax seed, which is a very good source of PUFA n-3 acids (Mourot, 2009) in feed mixtures affects the deposition of these acids, both in meat and in subcutaneous fat (Wood et al., 2008; Bertol et al., 2017; Tognocchi et al., 2023). Associated with the above change in fatty acid profile are changes, more favorable from the consumer’s point of view, in the values of the dietary indices of SM, LLM and RMLLM meat. All of these products, from the H+ and F+ groups, were characterized, compared to the control group, by a significantly lower PUFA n-6/n-3 ratio and a lower thrombogenic index (TI) value. It was also shown that feeding pigs with feed containing dried fruits and flax seeds (F+) had a beneficial effect on increasing the content of hypocholesterolemic acids (DFA), and thus increasing the ratio of hypocholesterolemic to hypercholesterolemic acids (h/H) in the meat of LLM fatteners. Also, Tognocchi et al. (2023) showed that meat from pigs fed flax seed was characterized by a significant reduction in the PUFA n-6/n-3 ratio, which was particularly evident in fat-rich tissues (backfat, bacon and the fatty part of ham). Bertol et al. (2017), using the addition of a mixture of rapeseed and flaxseed oils to pig feed, showed their significant effect on reducing the content of SFA and increasing PUFA n-6 and n-3, as well as decreasing the PUFA n-6/n-3 ratio in subcutaneous fat. The authors of this study further showed that the addition of grape pomace to feed containing a mixture of oils did not change the fatty acid profile in this tissue. Klimiuk et al. (2023), using the addition of 3% thyme to a mixture enriched with flaxseed oil, showed its positive effect on lowering SFA content, increasing PUFA (including PUFA n-3) and lowering the PUFA n-6/n-3 ratio and TI index in loin meat. The positive effect of using dried bergamot pulp in pig feeding on lowering the PUFA n-6/n-3 ratio in loin meat and salami was shown by Scerra et al. (2022). They also found that the additive affected the reduction of TI and AI indices in salami.
Summarizing the results obtained, it should be said that the addition of 2% dried herbs or fruits to the feed for the Złotnicka Spotted pigs had no effect on the texture profile (TPA, shear force), degree of oxidation (TBARS), physicochemical traits (color, pH) of the meat, and had little effect on improving the dietary value of the pork. The greatest effect on improving the dietary value of pork was found when an additional 3% of extruded flax seed was used. Raw ham and loin meat and raw maturing loins from fatteners fed with mixed feeds containing flax seeds and dried herbs or fruits were characterized by the highest content of PUFA n-3 acids. These additives, by lowering the PUFA n-6/n-3 ratio and the thrombogenic index (TI) in raw meat SM and LLM and RMLLM, increased the dietary and health-promoting value of these products. In addition, the use of an additive containing flax seeds and dried herbs in the mixture reduced the fat’s susceptibility to oxidation, as expressed by the TBARS value, primarily during loin maturation. The results suggest that a feed additive in the form of dried herbs and flax seeds may be particularly useful in feeding slow-growing pigs whose meat is intended for traditional long-maturing products.