Figure 1
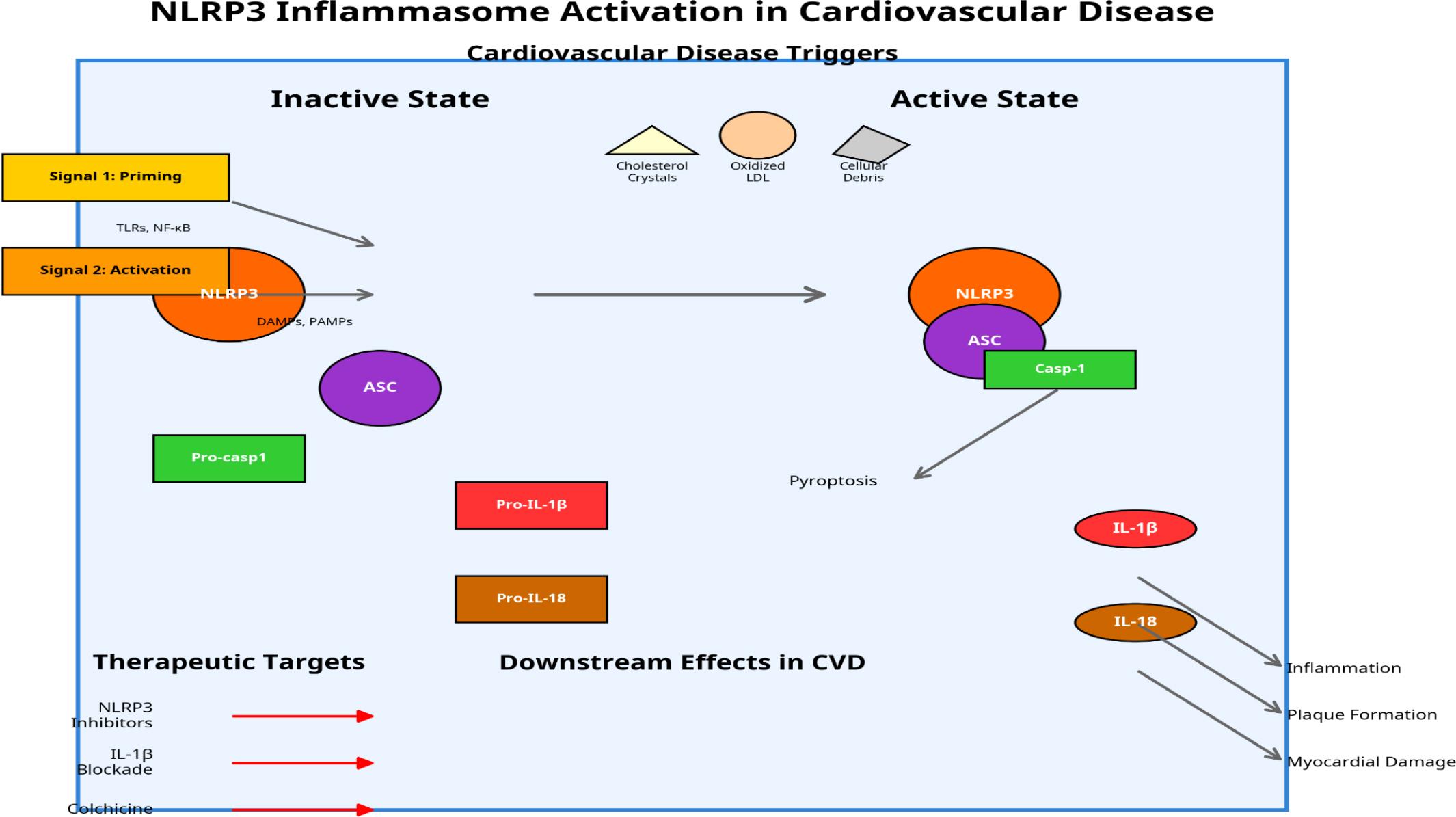
Figure 2
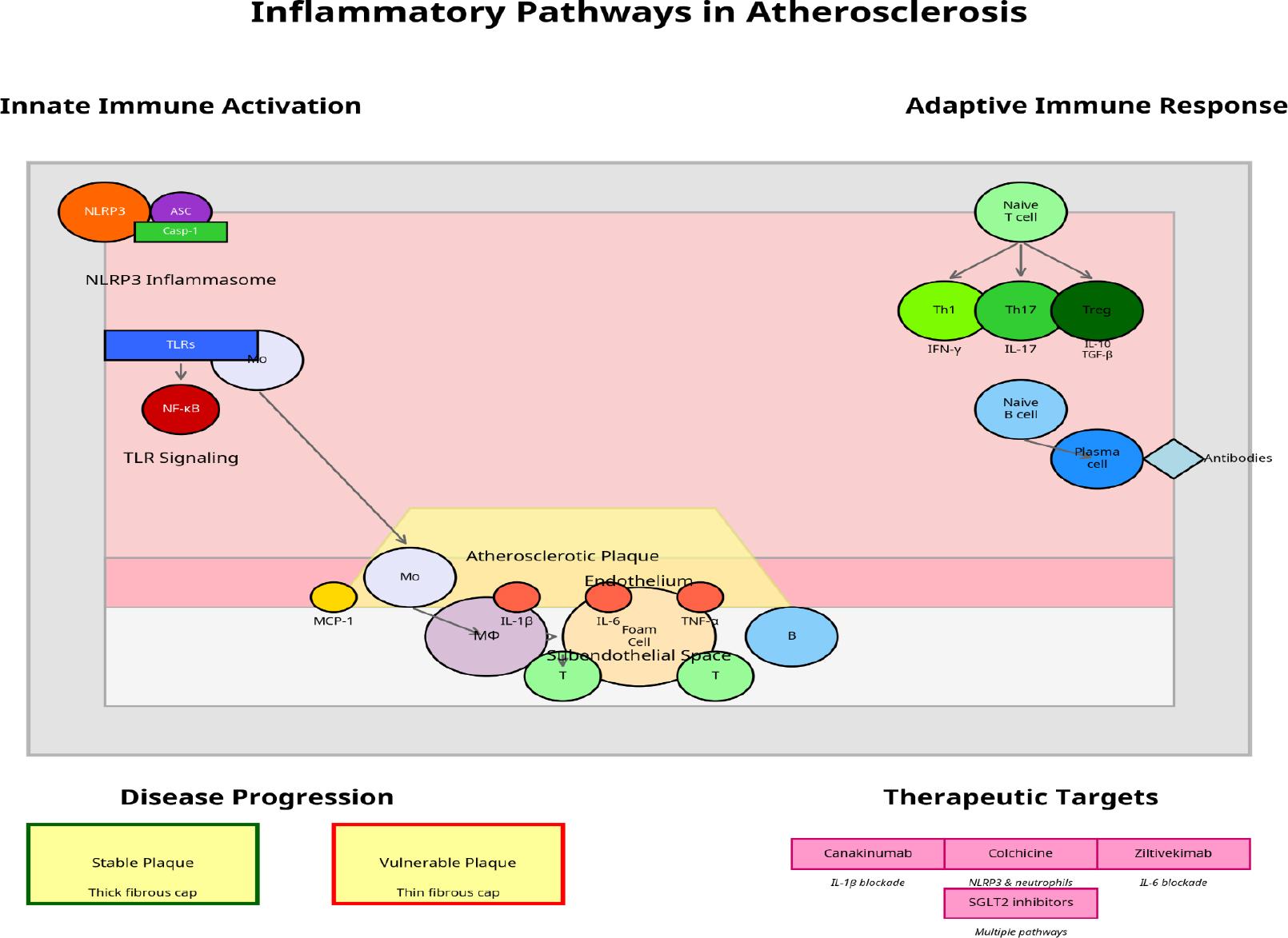
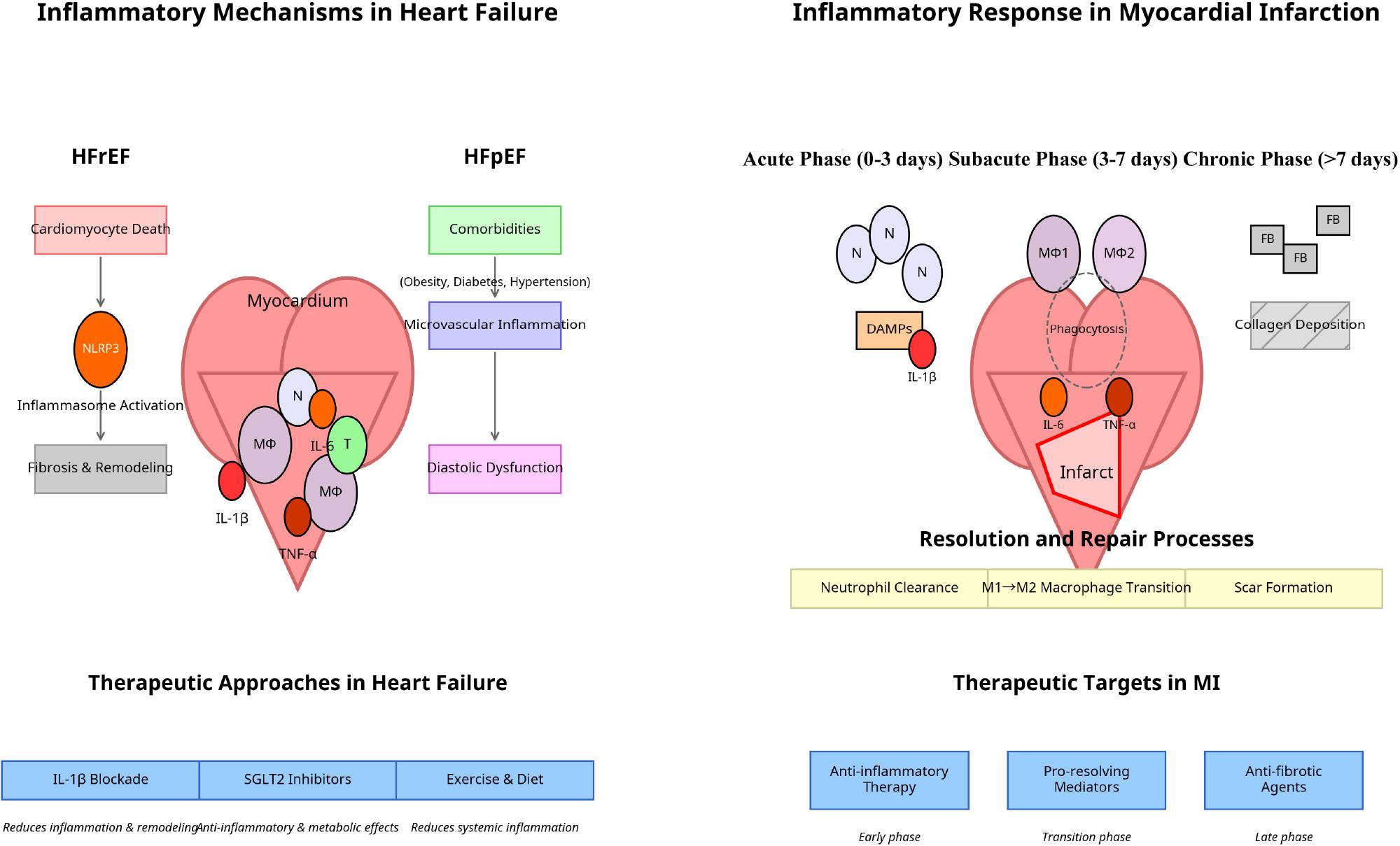
Figure 3
Clinical Trials Summary: CANTOS, COLCOT, and LoDoCo2 with Statistical Results_
| Trial Name | Target/Intervention | Study Population | Primary Endpoint | Key Findings | Statistical Results |
|---|---|---|---|---|---|
| CANTOS | Canakinumab (IL-lß inhibitor) | Post-MI patients with elevated hs-CRP (>2 mg/L) | MACE (CV death, MI, stroke) | Reduced MACE independent of lipid levels; no LDL-C change; ↑ infection risk | HR = 0.85; 95% CI: 0.74-0.98; p = 0.021 |
| COLCOT | Low-dose Colchicine (0.5 mg/day) | Patients within 30 days post-MI | Composite CV death, cardiac arrest, MI, stroke, angina, hospitalization | 23% relative risk reduction in the primary endpoint | HR = 0.77; 95% CI: 0.61-0.96; p = 0.02 |
| LoDoCo2 | Low-dose Colchicine (0.5 mg/day) | Stable coronary artery disease patients | Composite CV death, MI, stroke, or ischemia-driven revascularization | 31% relative risk reduction in the primary endpoint | HR = 0.69; 95% CI: 0.57-0.83; p < 0.001 |