Cardiovascular disease (CVD), particularly coronary artery disease (CAD), remains a leading cause of global morbidity and mortality.[1] While percutaneous coronary intervention (PCI) is a mainstay in the treatment of acute and chronic coronary syndromes, long-term success depends on comprehensive secondary prevention strategies, including lifestyle modification, medication adherence, and risk factor control.[2,3]
Cardiac rehabilitation (CR) is a guideline-recommended intervention that improves functional capacity and reduces cardiovascular events, yet global participation remains suboptimal due to barriers such as geographic distance, socioeconomic factors, and limited facility access.[4–6] Seminal studies such as Hambrecht et al.15 demonstrated that structured exercise-based CR can be as effective as PCI in certain populations. In response, telehealth, mobile health (mHealth), and remote monitoring technologies have emerged as promising alternatives to traditional center-based CR, especially during and after the COVID-19 pandemic.[7–9] CR is supported by performance and quality guidelines published by the ACC/AHA.[16]
These digital approaches—including smartphone applications, wearable sensors, remote coaching, and teleconsultations—may enhance patient engagement, support self-care, and deliver comparable clinical outcomes to conventional programs.[10,11] However, implementations vary widely across settings, and standardized outcome data remain limited.
This scoping review aims to map and synthesize recent evidence on telehealth and remote monitoring interventions in post-PCI patients, focusing on implementation strategies, reported clinical outcomes, and patient engagement mechanisms.
We conducted a scoping review according to a predefined protocol based on the following five-stage approach developed by the Joanna Briggs Institute (JBI): Stage 1, identifying the research question; Stage 2, identifying relevant studies; Stage 3, study selections; Stage 4, charting the data; and Stage 5, collating, summarizing, and reporting the results.[12]
To guide study inclusion, we followed the JBI Population–Concept–Context (PCC) framework, adapted to the focus of this scoping review on telehealth and remote monitoring interventions in patients following PCI.
- –
Population: We included studies involving human patients of any age who had undergone PCI for any indication, including acute coronary syndrome (ACS) or chronic coronary syndrome (CCS). Both inpatient and outpatient populations were eligible, provided the study focused on the post-procedural period.
- –
Concept: Eligible studies implemented telehealth, telemedicine, or remote monitoring interventions as part of post-PCI care. This included, but was not limited to, mHealth applications designed to support self-management; wearable devices used to monitor vital signs or physical activity remotely; virtual coaching sessions or teleconsultations with healthcare providers; home-based or hybrid CR programs that combined in-person and digital components; and digital platforms that facilitated patient education, routine follow-up, or medication adherence through remote access.
- –
Context: We considered studies conducted in any healthcare or community setting, including hospitals, rehabilitation centers, outpatient clinics, or home-based environments. To be eligible, studies had to be original research articles—such as randomized controlled trials, cohort studies, feasibility trials, or implementation studies—published in English between January 2020 and April 2025, reflecting the most recent developments in digital and remote care delivery during and after the COVID-19 pandemic.
We excluded studies if they met any of the following criteria:
- –
Studies that did not involve patients who had undergone PCI.
- –
Interventions that did not include any telehealth, telemedicine, or remote monitoring component.
- –
Studies focusing solely on in-hospital monitoring without a post-discharge or follow-up element.
- –
Studies evaluating digital tools unrelated to structured follow-up or CR.
- –
Editorials, commentaries, letters to the editor, or protocols without reported outcome data.
- –
Conference abstracts without access to full-text articles.
- –
Non-English language publications.
- –
Studies involving other cardiac procedures (e.g., coronary artery bypass grafting, valve interventions) without a specific focus on post-PCI care.
A structured literature search was conducted across PubMed and Google Scholar to identify relevant peer-reviewed studies published between January 2020 and April 2025. The search strategy combined Medical Subject Headings (MeSH) and keyword-based terms using Boolean operators to capture studies investigating the use of telehealth, telemedicine, or remote monitoring interventions in post-PCI patients. Google Scholar was searched using aligned combinations of free-text keywords, and results were manually screened. Only English-language, full-text original research articles were considered. All identified citations were imported into Mendeley for organization and duplicate removal. Two reviewers independently screened the titles and abstracts. Full texts of potentially eligible studies were reviewed in detail, and any disagreements regarding inclusion were resolved through discussion. A third reviewer provided methodological oversight.
Our initial search across PubMed and Google Scholar identified a total of 191 articles published between January 2020 and April 2025. Of the 191 records initially identified, 18 were excluded as review articles, and five were excluded for being non-English. A large number of studies (n = 97) were screened in the initial pass based on titles or abstracts that mentioned cardiovascular rehabilitation or telehealth in cardiac care but were later excluded upon full-text review because they did not specifically include patients who had undergone PCI. This reflects the broad nature of the initial search strategy, which was intentionally designed to be inclusive and capture all potentially relevant studies involving telehealth in post-cardiac care, in line with the JBI scoping review methodology.[12] The remaining 71 articles were assessed in full text, of which 59 were excluded for not incorporating any telehealth, telemedicine, or remote monitoring component. Ultimately, 12 studies met all inclusion criteria and were included in the final scoping review.
The 12 studies included in this scoping review implemented a variety of telehealth and remote monitoring interventions aimed at improving outcomes in patients following PCI. To enhance clarity and highlight patterns across studies, we categorized the interventions into four thematic groups based on modality and delivery model:
mHealth applications and smartphone-based coaching
hybrid and home-based CR programs
wearable devices and remote monitoring tools
communication platforms and digital disease management systems
Several studies employed mobile applications to deliver interactive education, real-time feedback, and personalized coaching. Yamashita et al.[2] evaluated a smartphone app that incorporated live coaching from health professionals. The intervention significantly improved medication adherence and quality of life metrics compared to standard care. Similarly, Cruz-Cobo et al.[9] studied the “eMOTIVA” app, which featured gamified education modules and self-monitoring tools. This app demonstrated enhanced adherence to dietary and lifestyle recommendations, improved cardiovascular knowledge, and higher user satisfaction scores.
Four studies examined hybrid or entirely home-based CR programs combining telehealth with traditional rehabilitation components. Mattioli et al.[4] compared a hybrid CR model (center-based sessions plus synchronous telerehabilitation) to standard CR and found noninferior outcomes in exercise capacity, muscle strength, and functional assessments. The role of home-based CR has been recognized in AHA/ACVPR scientific statements.[17] In a separate study, Pini[11] reported that a teleCR program achieved similar improvements in LDL cholesterol and exercise tolerance compared to center-based CR, with greater acceptance among younger, tech-savvy patients. These findings suggest that remote rehabilitation models can maintain clinical efficacy while improving accessibility.
Wearable technologies were used in multiple studies to monitor physical activity, vital signs, and exercise adherence. Sato et al.[13] utilized accelerometer-equipped wearables to deliver remote feedback and monitor physical activity in ACS patients. The intervention preserved renal function (eGFR) and increased step counts. The SWEDEHEART study by Lindman et al.[10] demonstrated the feasibility and safety of exercise-based cardiac telerehabilitation with remote monitoring in a multicenter real-world setting. These studies highlight the utility of wearables in extending rehabilitation monitoring beyond clinic visits.
Other studies leveraged digital communication tools and intelligent platforms to deliver structured follow-up and psychosocial support. Li et al.[8] used a WeChat-based intervention to improve quality of life and blood pressure control in post-PCI patients. Wang et al.[3] tested a “hospital-community-family” trinity model, integrating digital follow-up, community clinic support, and family engagement, which improved CR adherence and cardiac function. Chen et al.[14] applied a chronic disease intelligent information platform that enhanced self-care behaviors and health literacy. Zhang et al.[7] also showed that Internet-based telerehabilitation improved left ventricular function and reduced anxiety and depression compared to standard care.
CR is an essential component of secondary prevention after PCI, yet participation remains low due to access barriers, time constraints, and socioeconomic disparities.[4–6] In recent years, the integration of telehealth and digital technologies has offered new pathways for delivering CR remotely. Comprehensive secondary prevention through CV rehabilitation is emphasized in recent European guidelines.[18] The COVID-19 pandemic further accelerated interest in remote care delivery, prompting broader exploration of mobile applications, wearable technologies, and hybrid rehabilitation models as scalable alternatives to conventional programs.[7–9]
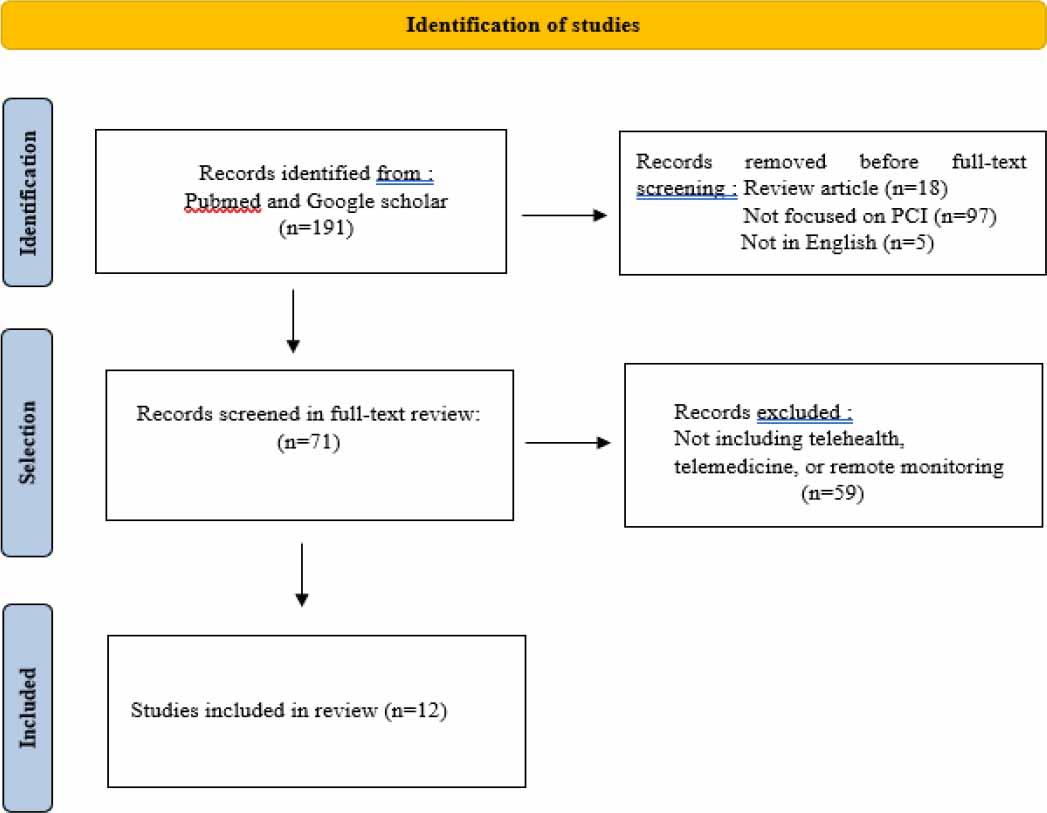
PRISMA flow diagram summarizing the study selection process.
The 12 studies included in this scoping review provide growing evidence that telehealth-based interventions can effectively support post-PCI recovery. Across diverse platforms—smartphone apps,[2,9] hybrid CR programs,[4,11] wearable devices[10,13] and digital followup tools[3,8]—these interventions improved medication adherence, physical activity, lifestyle modification, patient satisfaction, and psychological health. Although most studies focused on soft endpoints, several also reported improvements in functional capacity, lipid control, and cardiac function.
Successful interventions typically integrate one or more of the following elements:
- –
Exercise training delivered through hybrid CR or wearable-assisted telerehabilitation[4,10,11,13]
- –
Risk factor management, such as LDL control and medication adherence[2,11]
- –
Education and counseling via structured coaching, gamified apps, or remote health education[2,9]
- –
Remote monitoring using wearables or intelligent digital platforms for follow-up and symptom tracking[10,13,14]
These components reflect a shift toward personalized, selfdirected care with remote support.
Summary of included studies: telehealth and remote monitoring in post-PCI patients.
| Study | Technology | Intervention | Application | Type | Outcome Summary |
|---|---|---|---|---|---|
| Zhang et al. (2023) | Remote rehab app | Internet-based telerehab | CR adherence, LV function | Telehealth | Improved LV function, reduced anxiety |
| Wang et al. (2023) | Trinity rehab model | Hospital-community-family integration | Cardiac rehab participation | Telehealth + community | Improved LVEF and adherence |
| Mattioli et al. (2022) | Hybrid CR sessions | Hybrid cardiac rehab | Functional capacity, strength | Hybrid | Noninferior to center-based CR |
| Sato et al. (2025) | Wearable device | Remote PA feedback | Renal function, activity level | Wearable + web-based | Preserved eGFR, increased steps |
| Cruz-Cobo et al. (2023) | eMOTIVA app | Gamified mHealth app | Lifestyle, knowledge, satisfaction | mHealth | Improved diet, PA, satisfaction |
| Yamashita et al. (2022) | Smartphone app | Live health coaching | Medication adherence, QoL | mHealth + Coaching | Improved adherence, QoL |
| Pini (2024) | TeleCR program | Home-based rehab vs center | LDL, exercise capacity | TeleCR | Equivalent to center-based CR |
| Lindman et al. (2024) | Exercise tracking | Multicenter telerehab | Feasibility, engagement | Wearable/Tracking | Feasible, well-received |
| Li et al. (2022) | WeChat app | Structured health management | QoL, BP control | Messaging-based | Improved QoL, BP |
| Chen et al. (2023) | Chronic disease platform | Digital disease management | Self-care behavior | Web-based platform | Improved self-care literacy |
| Pini (2024) | TeleCR | Remote hybrid delivery | Risk factor control | Hybrid model | Preferred by younger patients |
| Chen et al. (2023) | Chronic disease platform | Digital disease management | Self-care behavior | Web-based platform | Improved self-care literacy |
Telehealth-based CR offers several advantages over traditional programs:
- –
Improved accessibility: reduces geographical and transportation barriers[3,11]
- –
Convenience and flexibility: supports patient engagement on their own schedule[9,11]
- –
Cost-effectiveness potential: reduces hospital visits and infrastructure needs
- –
Enhanced adherence: real-time feedback and coaching can improve long-term compliance[2,9,14]
Despite promising results, current evidence is limited by heterogeneity in study design, small sample sizes, and short follow-up durations. Most studies focused on behavioral or intermediate outcomes (e.g., adherence, anxiety) rather than hard clinical endpoints such as mortality, repeat revascularization, or hospital readmission. Data from large-scale randomized trials (e.g., TELE-ACS) remain sparse and are needed to strengthen the clinical evidence base. Additional challenges include technological literacy, patient supervision, lack of standardized protocols, and reimbursement uncertainties.
Future research should prioritize:
- –
larger, multicenter randomized controlled trials with long-term follow-up;
- –
standardized reporting of outcomes, including major adverse cardiovascular events (MACE);
- –
economic evaluations to assess cost-effectiveness and scalability;
- –
integration of artificial intelligence and predictive analytics to personalize care;
- –
health system-level strategies to support widespread implementation.
This scoping review has several limitations that should be acknowledged. First, although a structured and comprehensive search was conducted across PubMed and Google Scholar, the inclusion was limited to English-language articles, which may have introduced language bias and excluded relevant studies published in other languages.
Second, the heterogeneity of study designs, interventions, outcome measures, and populations limited our ability to perform comparative analysis or metasynthesis. The included studies varied widely in terms of intervention type (e.g., mHealth apps, wearables, hybrid CR models), follow-up duration, and reported outcomes, making it difficult to generalize findings across settings.
Third, many of the included studies were small-scale, singlecenter trials or feasibility studies with short-term follow-up. Longitudinal outcomes, such as sustained behavior change, MACE, and cost-effectiveness, were seldom addressed. This limits the ability to draw conclusions about the long-term clinical impact and scalability of these interventions. Last, although patient-reported outcomes such as satisfaction, engagement, and quality of life were commonly reported, few studies used standardized instruments or blinded assessments, which may affect the reliability of those results.
This scoping review highlights the expanding role of telehealth, mobile health applications, wearable devices, and hybrid rehabilitation models in the post-PCI setting. The 12 included studies demonstrate that such interventions can enhance CR participation, improve medication adherence, promote self-care, and positively impact both physical and psychological outcomes. These digital approaches show promise in overcoming long-standing barriers to secondary prevention, including geographical constraints, limited access to facilities, and patient disengagement. While the evidence supports their short-term feasibility and effectiveness, further research is needed to establish long-term clinical impact, cost-effectiveness, and integration into routine care. As healthcare systems increasingly adopt digital tools, telehealth has the potential to become a cornerstone of personalized, accessible, and sustainable cardiovascular care for PCI patients.