CPVT is a rare inherited channelopathy which predisposes patients to malignant ventricular arrhythmias during exercise or with emotional stress. One of its hallmark features is sudden cardiac arrest during exertion, which may very well be the inaugural symptom. The exercise ECG test is essential in unmasking PVCs with different morphologies and bidirectional VT and enables diagnosis since the resting ECG is unremarkable. Importantly, the heart is structurally normal. Genetic testing is useful for the diagnosis and prognosis of affected individuals, as well as for family screening. The condition is considered to be highly penetrant, but the clinical manifestations can vary significantly among relatives[1]. Although exceptionally rare, the diagnosis can be made in individuals over 40 years, provided that there is no evidence of coronary artery disease[2].
A 58-year-old man with persistent AF was evaluated in our center for exercise prescription. He was keen on low-intensity skill sports and medium-intensity endurance sports. He reported no significant symptoms and had no relevant family history. He was treated with 2.5mg of bisoprolol. The ECG showed AF at 72 bpm without other abnormalities (see Figure 1).
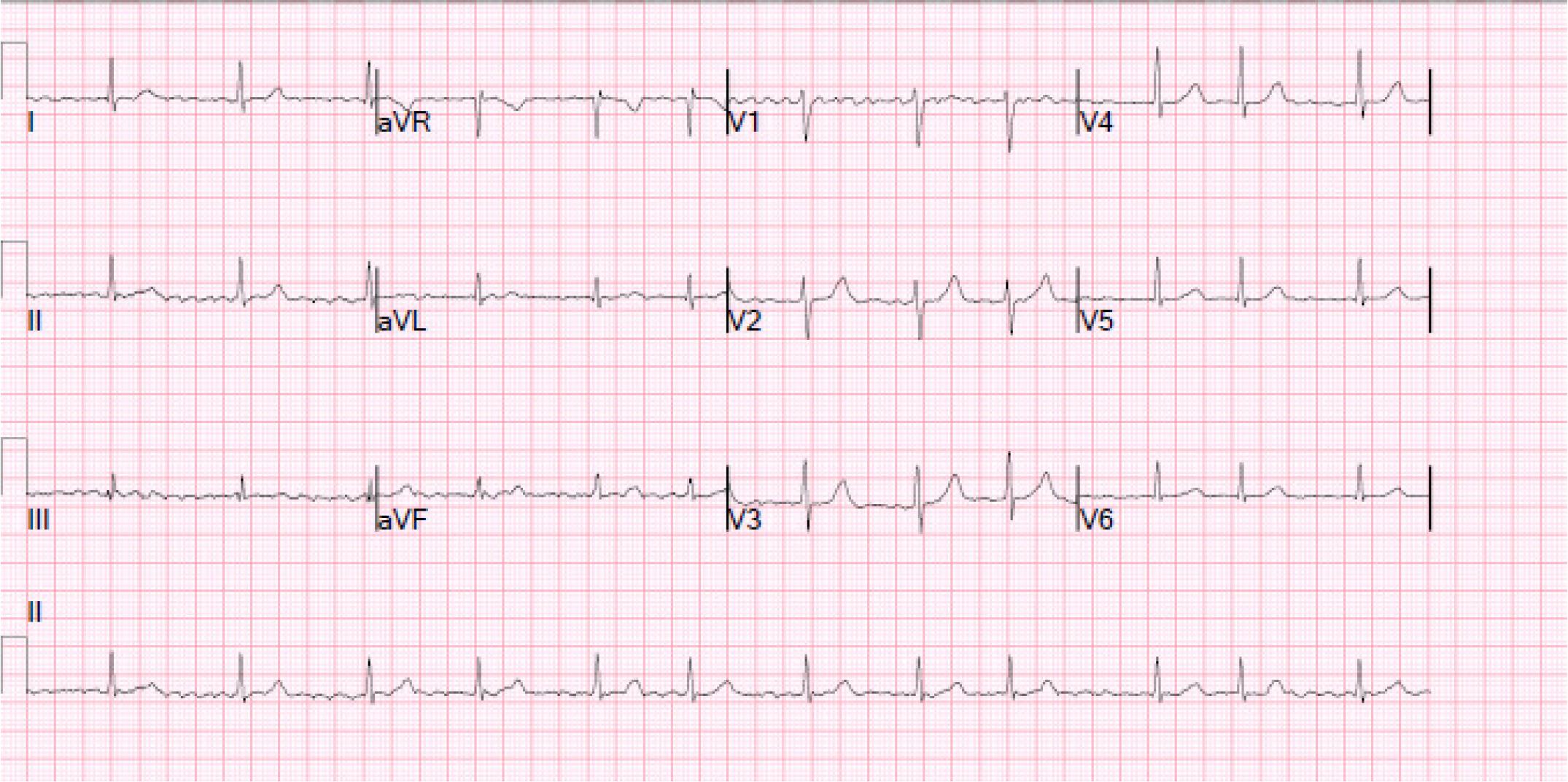
ECG at rest. The ECG trace is showing AF at 72 bpm with normal QRS morphology and no repolarization abnormalities. The QTc interval is 420ms
The TTE was remarkable for severe left atrial dilatation. The exercise test revealed a poorly controlled ventricular rate with effort, as the HR rose to 164 bpm (100% of maximum predicted HR) in just 3 minutes under the Bruce protocol. With peak exertion, several monomorphic PVCs were noted, including couplets and a 4-beat run of NSVT at 150 bpm with a ventricular capture (see Figure 2).
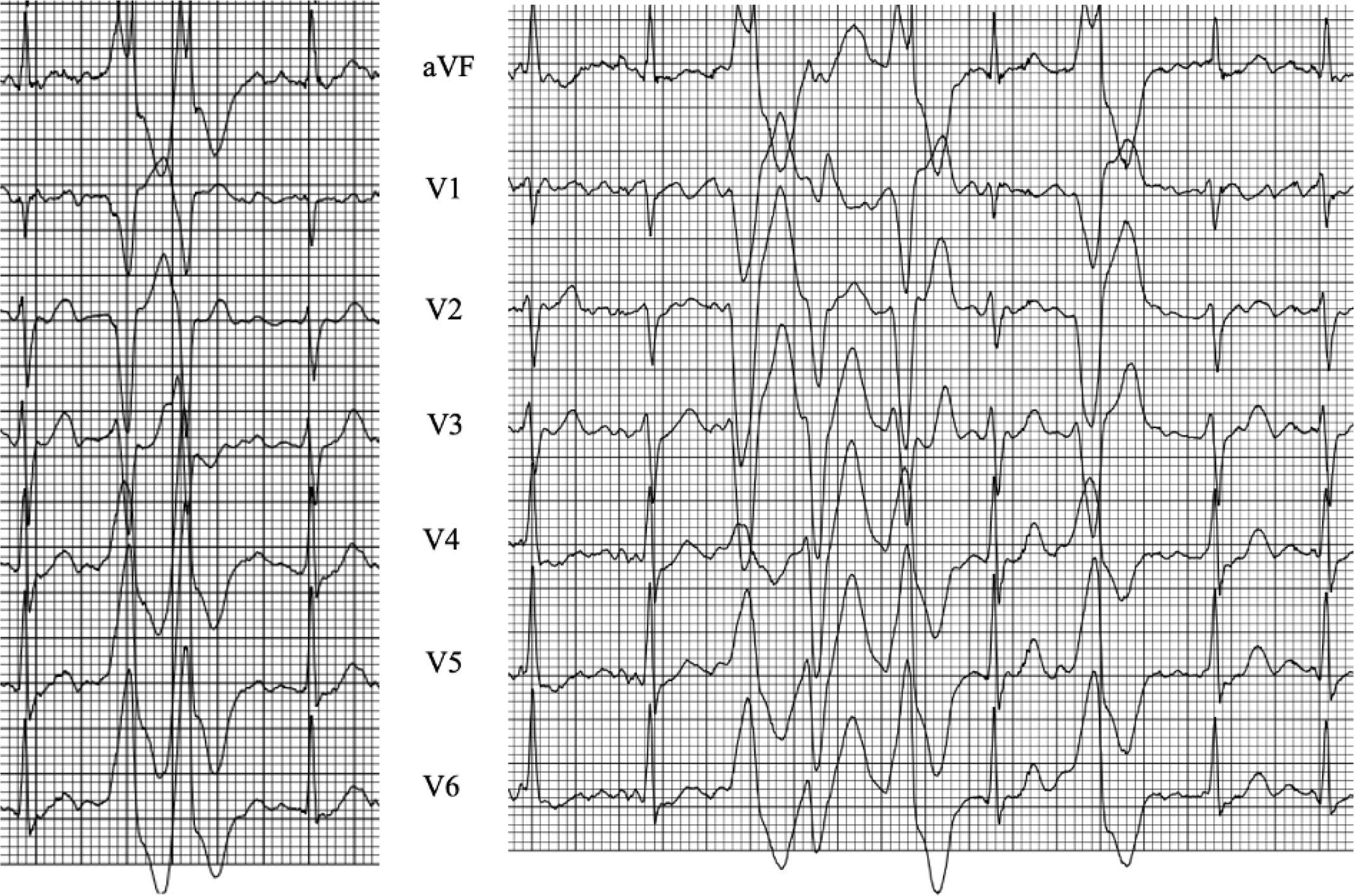
Initial exercise tolerance test. The ECG trace is showing a monomorphic couplet and a polymorphic run of NSVT at 150 bpm with ventricular capture
The ventricular arrhythmias resolved completely in recovery. For completion, a CTCA and CMR were performed. The investigations excluded significant coronary artery disease or an underlying cardiomyopathy. The beta-blocker dose was increased to 5mg od, and the patient was followed up with after one year, having been advised to limit his training to moderate-intensity exercise. Ablation was discussed, although the patient was not keen to proceed further with the procedure. Anticoagulation was not deemed necessary as his CHA2DS2VASC score was 0. In the meantime, the patient’s 25-year-old daughter suffered a sudden cardiac arrest while competing in her first triathlon. She was a recreational endurance athlete, having been engaged in high-intensity endurance exercise for 2 years prior to the event. She did not report any concerning symptoms during her previous training sessions. She was found to harbor a likely pathogenic RYR2 mutation (NM_001035.2, c.7175A>T missense variant, p.Tyr2392Phe) with evidence of biVT during peak exercise.
The same variant was later identified in our patient through predictive genetic testing. Our patient was reassessed in light of this new finding. An exercise test was conducted under bisoprolol where he exercised for 5 minutes under the Bruce protocol. He achieved a maximal HR of 171 bpm (105% of the predicted HR). PVCs were noted beyond 140 bpm, with 2 distinct morphologies. At 170 bpm, a bidirectional couplet was seen, followed shortly by an NSVT (3 beats at 170 bpm) (see Figure 3). The PVCs resolved quickly during recovery.
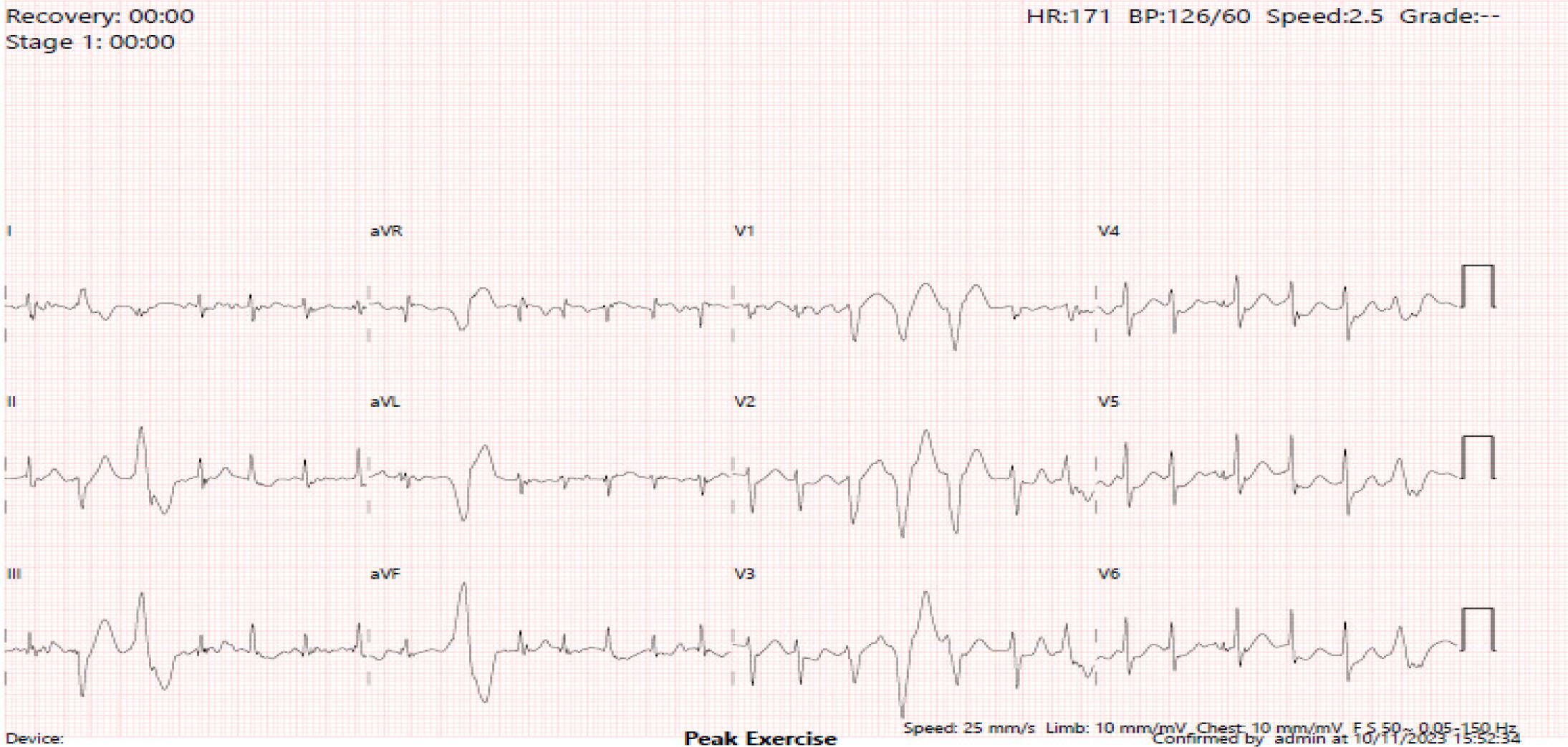
The second exercise tolerance test was performed under bisoprolol. The ECG strip recorded a bidirectional couplet and a 3-beat run at 170 bpm.
Bisoprolol was changed with a non-selective beta-blocker, nadolol 40mg bid. The repeat exercise test under nadolol demonstrated a maximal HR of 130 bpm (80% of predicted HR) with only rare monomorphic isolated PVC. The therapy with nadolol adequately managed the ventricular arrhythmias. Low intensity exercise (3–5 times per week, totaling 150–300 minutes per week) was recommended. Moderate intensity exercise was also discussed as per the patient’s request. Given the patient’s adherence to nadolol, he was advised to limit his HR to 115 bpm (70% of max predicted HR) during exercise and to avoid exercises with sudden bursts of effort.
CPVT bears a dire prognosis in the absence of treatment and appropriate lifestyle recommendations. Approximately 30% of affected individuals may experience a cardiac arrest and up to 80% may suffer one or more syncopal episodes. The genes linked to CPVT are RYR2, CALM1-2-3, and KCNJ2, all of which follow an autosomal dominant inheritance, whereas CASQ2, TECRL, and TRDN are predominantly autosomal recessive[3].
RYR2 is the largest known ion channel in the human genome, playing a key role in calcium regulation in cardiomyocytes[4]. More than 200 gain-of-function RYR2 mutations have been documented. In contrast, loss-of-function mutations in RYR2, although rare in CPVT, have been linked to short-coupled torsades de pointes and idiopathic ventricular fibrillation. Although the myocardium is normal in CPVT, large deletions in the RYR2 gene may be associated with structural heart disease [5]. Symptom onset varies by mutation, but most presentations occur before the age of ten, followed by adults in their 20s and 30s[2]. Disease expression may span from severe early-onset events to milder, late-onset presentations, which are rare. Consequently, our patient would have remained undiagnosed had it not been for the sudden cardiac arrest in his daughter. Importantly, she did not report any concerning symptoms with her training prior to the event, adding evidence to the well-known fact that sudden cardiac arrest may be the inaugural symptom in these patients. This was her first triathlon, which exposed her to elevated and sustained levels of catecholamines, triggering the malignant arrhythmia. Quite concerningly, such cases might be missed even in professional or elite athletes, who undergo screening with an ECG and questionnaire. The resting 12-lead ECG is unremarkable in CPVT. In the absence of symptoms or relevant family history, no further investigations would be performed. However, some countries, such as Italy, include exercise tolerance tests as part of screening in professional athletes.
The estimated penetrance rate of RYR2-related disease is high, estimated at 75-80%, which would indicate that asymptomatic individuals are a minority[6]. The disease penetrance reported in the literature ranges from 65 to 83%. As the disease expresses itself during exercise and stress, the exercise tolerance test protocols employed by different teams are important to consider, possibly explaining the differences in disease penetrance reported by studies[7–10]. Burst protocols could be of better value in unmasking the characteristic phenotype as they reproduce the sudden catecholamine surge. However, the evidence is rather limited[11]. The first exercise tolerance test failed to show the characteristic bidirectional ventricular arrhythmias as the patient was in AF, being able to exercise for only 3 minutes because of the uncontrolled ventricular rate. Hence, a significant or sustained catecholamine surge, which would have triggered the characteristic ventricular arrhythmias, was not achieved. As demonstrated by a study looking into families with RYR2 mutations, the severity of the phenotype can vary significantly, at least when comparing probands with their RYR2 genotype-positive relatives. Importantly, while only half of the mutation carriers showed evidence of the disease, 25% presented NSVT. Cardiac arrest may be the first symptom in otherwise silent carriers of pathogenic mutations, emphasizing the role of beta-blocker therapy in all individuals known to carry the mutation. A study by Hayashi et al. found that 13% of mutation carriers with negative stress tests experienced cardiac arrest during follow-up without therapy[12]. Hence, a homogenous clinical approach seems reasonable, with treatment being recommended in all carriers. Disease severity correlated significantly with mutation location; carriers with mutations in the C-terminal channel-forming domain exhibited more severe phenotypes. Meanwhile, mutations in the central domain were significantly associated with supraventricular disease manifestations[1]. The mutation identified in our patient affects the central domain.
A systematic approach is warranted to accurately evaluate the substrate of the bidirectional ventricular arrhythmias. The differential diagnosis includes conditions like myocardial ischemia, which is prevalent in our patient’s age group, or other rare hereditary cardiac conditions, such as ATS or arrhythmogenic cardiomyopathy. Notably, a truncating PKP2 variant may present with malignant polymorphic ventricular arrhythmias in the absence of any structural abnormalities[13]. Exercise testing can help distinguish between ATS and CPVT, as ventricular arrhythmias are typically suppressed at peak exercise in ATS but provoked in CPVT. Other findings should also be sought in the differential diagnosis, such as the presence of characteristic U-waves and dysmorphic features, which are suggestive of ATS. The specificity of bidirectional PVCs for the diagnosis of CPVT becomes less relevant in older adults, given the prevalence of myocardial ischemia and other conditions. The exercise tolerance tests in our patient demonstrated only bidirectional couplets, which, albeit suspicious, would not have been diagnostic or even highly suggestive of CPVT, particularly in the absence of the genotype.
Patient adherence to treatment is critical and guides exercise prescription in CPVT. Non-compliance or abrupt discontinuation of beta-blockers can trigger a rebound effect of catecholamines, increasing the risk for malignant arrhythmias[14]. Current guidelines include a Class I recommendation for the use of non-selective beta-blockers, such as nadolol and propranolol, in individuals with a positive genetic test, even in the absence of documented arrhythmias[15].
In AF, rapid atrioventricular node conduction during exercise can cause symptoms like dizziness, syncope, fatigue, and reduced performance. Recommendations for sports participation depend on the ventricular rate during AF episodes. If rate control is achieved and no symptoms of hemodynamic impairment are present, the patient may continue with their exercise[16]. The initial NSVT, as it was very short, did not raise significant concerns regarding exercise without evidence of coronary artery disease or a myocardial substrate. The exercise prescription changed after the genetic testing. According to the 2015 Scientific Statement from the American Heart Association, sports (beyond class IA sports) are not recommended for a patient with CPVT and documented exercise-induced PVCs/NSVT[17]. The American Heart Association classification of sports is based on peak static and dynamic components achieved during competition. Class IA sports are low in both static and dynamic demands, placing the least cardiovascular strain on the heart (low cardiac output and blood pressure). Examples of class IA sports include bowling, curling, cricket, golf, yoga, and riflery[18]. According to the 2021 European recommendations, long-term management of CPVT focuses on limiting exercise to moderate intensity (30-60 minutes, 3-7 days a week). While ventricular arrhythmias can occur at moderate HRs, some patients may engage in sports with appropriate treatment and, most importantly, avoidance of sudden bursts of activity. Based on prior exercise testing, individualized maximal HR limits may be specified. [19]
The case underscores the importance of predictive genetic testing for CPVT, even in the absence of symptoms or characteristic clinical findings. The 58-year-old male patient, who initially presented with AF and monomorphic NSVT without evidence of bidirectional ventricular arrhythmias, was later found to harbor a pathogenic RYR2 mutation following his daughter’s cardiac arrest. Supraventricular arrhythmias may be associated with certain RYR2 mutations which involve the central domain. This case highlights the variable penetrance of the genotype, which is rather unusual, as the patient and the proband are at opposite ends of the clinical spectrum in terms of disease severity. Predictive genetic testing led to a shift in his management, with nadolol initiation and tailored exercise prescription.