Addictive substances can be classified by various criteria. The first is legal status, as seen with alcohol (ethanol), nicotine, and caffeine. All these substances are legal, provided that driving under the influence is not considered. Some substances are permitted only with a medical prescription. However, many people misuse these drugs. For example, benzodiazepines are a large group of drugs that are often abused. Other examples include methadone and ketamine. The last group consists of substances that are illegal in all cases, such as heroin, cocaine, and others. This classification is most relevant from a legal perspective. Another classification is based on the chemical structure. There is the group of amphetamines and methamphetamines; for example, ecstasy belongs to this group. Benzodiazepines include more than forty substances and can be further divided into two types. Another group consists of hemp products. Many people think only of marihuana, but there is also hashish, and, thirdly hemp leaves.
Drugs are a general problem, not only when people drive or operate machinery under their influence. In these cases, the danger resulting from impaired human perception is greater than in other circumstances. For this reason, it would be highly desirable to have a device capable of detecting the presence of illicit drugs in the human body. Zero tolerance for drugs is one of the prerequisites for the successful deployment of such devices. There are many substances that are abused as drugs, and even if we focus on a few selected substances, the scope is not so wide. According to the European Drug Report 2024 [1], more than 90 % of drugs seized in the European Union (EU) in 2022 were accounted for by a few substances or groups of substances. These include hemp products containing tetrahydrocannabinol (THC) – 35 %, cocaine – 28 %, heroin – 16 %, amphetamines (amphetamine and methamphetamine) – 10 %, and ketamine – 2.3 %, which is still significant. This fact allows us to focus on these substances. Even though the problem of addictive drugs is more widely discussed, the number of deaths from overdose has been increasing since recently [2]. The rate of drug overdose deaths in the United States (US) rose from 8.9 deaths per 100000 people in 2003 to 32.6 deaths per 100000 people in 2022. Data from the EU show similar trends to those in the US, with the only difference being in data processing. The number of drug-related deaths in the EU increased by about 38 % from 2013 to 2022 [1]. This further emphasizes the necessity of any tool that can help detect drugs.
There are many types of biological samples that can be used to detect drugs in the human body. Some biological materials, such as hair and nails, allow substances to be detected for extended periods, ranging from weeks to months (see Fig. 1). However, these materials are not suitable for determining if a person is currently under the influence of drugs. In contrast, materials such as saliva, blood, breath, and urine are much more convenient for this purpose [3].
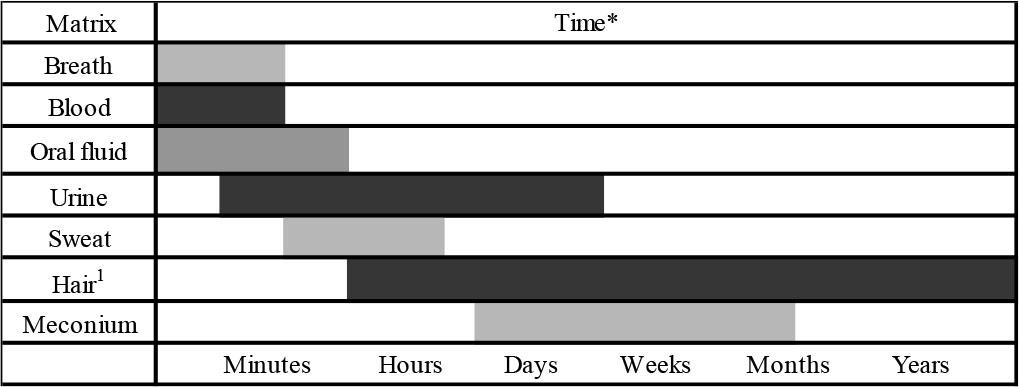
Window of detection for various matrices (Adapted from: Clinical Drug Testing in Primary Care 2012).
* Very broad estimations that also depend on the substance, the amount and frequency of use, and other factors.
1 7–10 days after use to the time required to grow the length of hair, but may be limited to 6 months of hair growth. However, most laboratories analyze an amount of hair equivalent to 3 months of growth.
The period during which drugs can be detected in a specific type of sample is influenced by many factors. These differences are determined by the nature of the substance and the target biological material. The method of use also has considerable influence on the detection window. Significant differences can be observed between one-time use, occasional use, and chronic use [3], [4]. From this perspective, only body fluids such as blood and oral fluid are suitable for relatively rapid detection. Finally, the most important factor in choosing the best sampling method is the simplicity of sample collection. For blood, medical staff must be involved, and the method is invasive. In comparison, collecting a saliva sample is only a small invasion of privacy. Therefore, oral fluid appears to be the best option for drug detection.
For testing, it was necessary to select a suitable device for which detection limits would be determined. The second part involved preparing reference materials with a defined content of addictive substances.
There are many simple single-use tests that can detect these drugs, but their evaluation depends on the user. Therefore, the results of these tests are not admissible as evidence. For legal purposes, it is preferable to have a device available for evaluation. There are at least three devices on the market that can detect selected drugs in the body. One is the Dräger DrugTest 5000 (DDT 5000), and the other two are the AquilaScan WDTP-80 and WDTP-10. All these devices use single-use kits for collecting body fluid, in this case, saliva. The principle of this method is the same as that of simple single-use tests, which are based on immunoassay. The device from the manufacturer Dräger was included in the DRUID project [5], which focused on measuring the capabilities of various drug detection devices. In this study, results from the DDT 5000 were compared with laboratory analysis using sophisticated analytical systems such as UPLC-MS/MS. The results from this comparison were more than acceptable. The lowest specificity achieved was 87 %, and of the lowest accuracy was 84 %. Both parameters were related to the official detection limits from the manufacturer (see Table 1 and Table 2).
Specificity and accuracy of DDT 5000 obtained in the DRUID project in Belgium.
| Substance | Belgium | |
|---|---|---|
| Specificity [%] | Accuracy [%] | |
| Cocaine | 96 | 93 |
| Opioids | 87 | 89 |
| Benzodiazepines | 99 | 99 |
| Cannabis | 99 | 91 |
| Amphetamine | 100 | 99 |
| Methamphetamine | 100 | 100 |
Specificity and accuracy of DDT 5000 obtained in the DRUID project in the Netherlands.
| Substance | Netherlands | |
|---|---|---|
| Specificity [%] | Accuracy [%] | |
| Cocaine | 97 | 95 |
| Opioids | 100 | 100 |
| Benzodiazepines | 90 | 84 |
| Cannabis | 100 | 100 |
| Amphetamine | 100 | 98 |
| Methamphetamine | 100 | 100 |
Evaluation of detection limits under laboratory conditions using samples prepared to match real saliva from certified reference materials is more reliable [6]. Real saliva was collected from selected volunteers. The DDT 5000 device achieved overall excellent results in all monitored parameters: sensitivity was 99.1 %, specificity was 99.2 %, and accuracy was 98.3 %. For all detection limit evaluations (30 % above, at, and 30 % below), the DDT 5000 detected 208 out of a possible 210 drug-positive samples. In comparison, the AquilaScan WDTP-10 device achieved a sensitivity of only 21.7 % and an accuracy of only 66.2 %; only the specificity was comparable to the DDT 5000, with a value of 99.5 %. The main reason for this result was the failure to detect THC. All samples containing THC were classified as negative, even when the THC concentration was 30 % above the detection limit specified by the manufacturer.
An immunochemical reaction may occur in vivo, where it primarily represents the immune response of an organism to a pathogen. In an in vitro environment, it is used in immunoanalytical laboratories for detection and quantitative determination. To express the result, one of the participants in the reaction (antigen or antibody) is labeled with an indicator that generates a signal. This signal should be easily measurable with high accuracy and reproducibility [7]. Immunoanalytical methods have exceptionally high sensitivity, reaching values of 10−15 to 10−20 mol·L−1. In practice, these values represent a concentration of approximately one molecule per liter. This capability is primarily the result of several properties of antibodies:
Ability to bind a wide range of natural and synthetic compounds, cells, and viruses that act as antigens. Antibodies are proteins, and their large number of binding sites arises from the enormous number of possible amino acid sequence combinations.
Specificity for the reacting substance – the ability to bind precisely to the target substance even in the presence of other molecules.
Binding strength between antibody and antigen – the antibody–antigen complex remains stable even during subsequent processes.
In this case, the specific antibody (Ab) is present in a limited amount. Its binding sites are competed for by the labeled antigen (Ag*), which is in excess, and the same but unlabeled antigen (Ag), which is present in the sample and being measured. Both antigens occupy the antibody binding sites.
In this method, the specific antibody is present in excess. The measured antigen reacts with it, and for quantification, a labeled suitable specific antibody (Ab*) is used. The amount of analyte is directly proportional to the amount of complex [Ag–Ab*].
Sophisticated laboratory procedures allow for concentration determination, but these are often associated with blood sampling. Blood collection requires medical personnel, and biological samples are demanding in terms of storage. An interesting alternative is breath sampling, which is non-invasive and does not intrude on privacy. A testing device with a microfilter (Fig. 2) captures particles from breath, with collection taking approximately 2 minutes. A study involving 47 patients compared the analysis of breath, urine, and blood [8]. Breath sampling is more complex than alcohol testing, as it requires chemical extraction from filters. Sample analysis was performed using GC-MS/MS, LC-MS/MS, and CEDIA. Six drugs were monitored: amphetamine, methamphetamine, THC, benzodiazepines, methadone, and heroin. The results showed good agreement between analyses of different samples; for example, amphetamine was confirmed in breath in 17 individuals, and THC in plasma in 9 individuals – 8 of whom also had THC detected in breath. Benzodiazepines were detected in 32 individuals and confirmed in breath in 15. The study confirms that breath tests are a reliable alternative to urine and blood, especially at higher drug concentrations [9].

Sampling device for the determination of drugs from breath (Adapted from: Beck, Stephanson, Sandqvist, Franck [2012]).
Benzodiazepines can be extracted with methanol from various preparations and analyzed by gas chromatography with flame-ionization detection, which, however, may cause thermal degradation [10]. The GC-MS method is highly effective, combining the separation capability of chromatography with the precise identification of compounds by mass spectrometry. In combination with suitable standards, it provides very accurate results [10]. HPLC is less suitable due to the complicated separation of diverse benzodiazepines, though separation efficiency can be improved by the choice of column and solvent.
The identification of drugs in blood, urine, and saliva is more complex than the analysis of such substances in pharmaceuticals or illicit preparations. Cocaine and its metabolites can be determined by GC-MS, but immunological tests targeting benzoylecgonine show low reactivity to cocaine, which may lead to false negatives [11]. A case of death from overdose demonstrated significant differences in concentrations between blood and urine, leading to a negative immunoassay result. The elimination half-life of cocaine and its metabolites varies.
THC can also be determined by mass spectrometry; however, GC-MS/MS is used in this case due to its low concentrations in body fluids [12]. Metabolite extraction is performed by solid-phase extraction or the MEPS method, which is effective for small sample volumes [13]. However, sample preparation is time-consuming and includes protein precipitation with cold acetonitrile, centrifugation, buffering, and homogenization.
Amphetamine and methamphetamine are usually determined from urine, where about 43 % of methamphetamine and 5 % of amphetamine (from methamphetamine conversion) are present after 24 hours [14]. The analysis involves GC-MS and solid-phase extraction; the sample is alkalized, extracted with methanol, and derivatized with trifluoroacetic acid.
Drug content can also be determined from hair, which is easily accessible and stores well. Cocaine, opiates, MDMA, and amphetamines can be detected and are divided into two groups with different extraction procedures. Cocaine and opiates are extracted with methanol and purified on a solid phase, whereas amphetamines undergo hydrolysis, extraction with ethyl acetate, derivatization, and GC-MS analysis. Despite the absence of solid-phase extraction, this remains a complex process.
The first step was the preparation of the reference solution. Reference materials were prepared using the gravimetric method. As a matrix, a test solution intended to simulate human saliva was used. The same composition of synthetic saliva was applied in research on the influence of saliva on dental alloys [15]. This approach was beneficial for achieving more realistic conditions. Nevertheless, the stability of these reference materials was insufficient. Therefore, the reference solutions of drugs were prepared in distilled water to avoid potential interactions between the matrix and the drugs themselves. Each substance was prepared separately at a concentration equal to the detection limit. Certified reference materials with known concentrations were chosen as the starting point for preparation. Measurements were performed on the DDT 5000 with standard saliva collectors. Collectors for six substances were used, except for methadone and ketamine, for which collectors for eight substances were necessary.
Reference solutions were prepared by diluting purchased reference materials, which had a concentration of 1 mg of the given substance per 1 ml of solvent. To achieve very low concentrations, multiple dilutions were necessary, as in the case of THC. Sample dilutions were carried out exclusively in water, since organic solvents cannot be applied to the collectors.
The first measurement for all drugs was performed at the concentration corresponding to the detection limit defined by the manufacturer. There is only one detection limit for all substances except THC. Multiple detection limits are available for THC depending on the device settings (see Table 3).
Detection limits estimated by the manufacturer.
| Substance | Detection limits [ng/ml] |
|---|---|
| Amphetamine | 50 |
| Benzodiazepines | 15 |
| Cocaine | 20 |
| Methamphetamine | 35 |
| Methadone | 20 |
| Ketamine | 300 |
| Opiates | 20 |
| THC | 5/10/25* |
Depends on the device setting
The required amount of a given addictive substance is calculated based on the desired concentration and the required volume of the prepared solution, as shown in (1).
mi – mass of the substance i,
c⊙ – concentration of the desired solution,
V⊙ – volume of the desired solution.
The mass of the available standard of the given substance is calculated using (2).
mstd – mass of the starting standard,
mi – mass of the addictive substance,
cstd – concentration of the starting standard,
ρstd – density of the starting standard.
The amount of solvent needed to prepare the desired volume of the solution can be calculated as the remainder of the total volume, as shown in (3).
Vroz – volume of added solvent,
V⊙ – volume of the prepared solution,
Vstd – volume of the added starting standard.
A similar procedure is followed when preparing a multi-component solution containing several addictive substances. According to (1), the mass of each substance being tested is calculated. Based on this data, the mass of the standard for each substance is determined using (2). The amount of solvent required to achieve the desired final volume of the prepared solution is determined from (4).
Vroz – volume of added solvent,
V⊙ – volume of the prepared solution,
Vstdi– volume of the added starting standard of substance i.
Water is always used as the added solvent. It is necessary to account for changes in its density depending on temperature. This can be calculated using (5) [16].
Given the amount of solvent (water) and the original standard added, the effect of volume contraction is neglected. The final concentration of the prepared solution is calculated using (6).
ci – concentration of substance i in the prepared solution,
ρw – density of the added solvent (water),
mi – mass of added substance i,
msol – mass of the prepared solution.
The uncertainty of the final concentration of the prepared solution is expressed as the expanded uncertainty U, with an expansion factor of 2, which ensures a 95 % probability that the value lies within this interval, assuming a normal distribution. Sources of uncertainty include the repeatability of weighing, the uncertainty of the input standard, characteristics of the balance, uncertainty in the molecular weight of the substances used, and uncertainty in the density of the solvents used.
The sample standard deviation represents the repeatability of the measurements and can be presented as a Type A uncertainty. The sample standard deviation is calculated using (7).
s – sample standard deviation,
uA – Type A uncertainty,
n – number of measurements,
xi – value of the measured quantity,
x̄ – mean of the measured values.
Type B uncertainty includes the other described contributions.
The uncertainty of the balance is taken from the calibration certificate for the given balance. The calibration already accounts for eccentricity. Additionally, the drift of the balance, specified by the manufacturer as 0.2 mg, and the uncertainty due to the resolution of the balance are also included. The uncertainty arising from weighing is given by (8).
ucal – the uncertainty from the calibration sheet,
udrift – the uncertainty caused by drift,
ures – uncertainty caused by the resolution of the display.
The uncertainty stated in the calibration certificate is 0.13 mg for a mass of 1 g. The uncertainty arising from reading the balance display is given by (9).
d – the smallest value displayed.
The next contribution to Type B uncertainty comes from the uncertainty in the calculation of the water volume. The amount of added starting standard is negligible compared to the amount of added solvent. Therefore, the uncertainty associated with the density of water is considered. When using the given equation to calculate the density of water as a function of temperature, this contribution is equal to 9 × 10−7 g/ml [17].
Type B uncertainty uB is calculated using (10).
The standard uncertainty u of the prepared solution is calculated using (11).
The expanded uncertainty U of the prepared solution is obtained by multiplying the standard uncertainty by the expansion factor corresponding to a 95 % probability for a normal distribution, as shown in (12).
The uncertainty is calculated for an amphetamine solution with a concentration of 97.6 ng/ml. First, the relative standard deviation is calculated based on the weighing of the primary standard and the solvent. The standard deviation is determined from repeated weighings using Excel software. This value represents a Type A uncertainty. The weighing is performed ten times. The Type A uncertainty values for the solvent (water), uAmw and the primary standard, uAmi, are as follows:
Type A uncertainty can also be expressed relatively, in percent, for later use.
The first component of Type B uncertainty originates from the calibration and characteristics of the balance. The individual components are summarized in Table 4 for the starting standard and in Table 5 for the solvent (water).
Uncertainty contributions from the calibration and characteristics of the balance for the starting standard.
| Parameter | Uncertainty | Unit |
|---|---|---|
| Calibration | 0.65∙10−3 | g |
| Drift | 0.20∙10−3 | g |
| Resolution | 0.17∙10−3 | g |
| umi | 0.70∙10−4 | g |
Uncertainty contributions from the calibration and characteristics of the balance for the solvent.
| Parameter | Uncertainty | Unit |
|---|---|---|
| Calibration | 0.85∙10−3 | g |
| Drift | 2.20∙10−3 | g |
| Resolution | 0.58∙10−3 | g |
| umsol | 2.43∙10−3 | g |
In the following step, the derivations described (10) are carried out, resulting in (13). The calculated values of uncertainties and the corresponding sensitivity coefficients are presented in Table 6.
Calculated values of uncertainties and the corresponding sensitivity coefficients.
| Parameter | Uncertainty | Unit | Sensitivity coefficient | Unit |
|---|---|---|---|---|
| umi | 0.70∙10−4 | g | 2.00∙10−3 | ml−1 |
| umsol | 2.43∙10−3 | g | 1.56∙10−7 | ml−1 |
| uρw | 9.00∙10−7 | g/ml | 7.77∙10−5 | - |
| uB | 1.40∙10−7 | g/ml | - | - |
| uBr | 1.44∙10−7 | % | - | - |
The nearly identical values of uB and uBr are due to the prepared concentration of the reference material being close to 100 ng/ml.
Based on the above, the relative uncertainty urci can be calculated as shown in (14). The main contribution to the overall uncertainty is from Type A uncertainty, which results from the very small sample weights of the addictive substance.
The standard uncertainty expressed in absolute terms and the corresponding expanded uncertainty with a coverage factor of k = 2 for a 95 % confidence level are as follows:
Before measurement on the tested device, the samples are applied onto collection collectors. The collector from un-packing to removal is shown in Fig. 3. These collectors absorb the reference solution, which is applied in a volume of 0.3 ml (under laboratory conditions, the collected volume may differ during actual use). The collectors contain a control section that changes color from white to blue when enough sample has been absorbed. In part 3 of Fig. 2, a clear color change in the lower part of the collector can be observed after collecting a sufficient amount of sample (the change is highlighted with a red circle).

Sampling kit for DDT 5000; (a) New kit; (b) Kit prepared for use; (c) Kit with collected sample; (d) Kit after evaluation.
Each collector also includes a cap that is inserted into the device. After sample evaluation, the device pushes the cap onto the collector, preventing any further contact with the collected sample. The entire collector is then inserted into the device, which automatically performs the evaluation. This is the greatest advantage of the device, as the evaluation cannot be influenced by the operator.
After inserting the collector into the device (see Fig. 4) and closing the collector compartment, the mode for THC to be applied to the given sample is selected. Once the selected THC mode is confirmed, the device performs a collector check. After this check is completed, the device prepares the sample, and after preparation, the evaluation starts automatically. Depending on the chosen THC mode, the evaluation time varies: for rapid mode, it is 4 minutes; for standard mode, it is 7 minutes; and for sensitive mode, it is 15 minutes. The collector check takes approximately 20 seconds, and sample preparation takes 25 seconds.

Collector inserted into the DDT 5000 device.
The DDT 5000 device does not provide results as the exact concentration of detected substances. The result is presented as positive/negative (Fig. 5). For this reason, it is crucial to know the detection limits of this device.
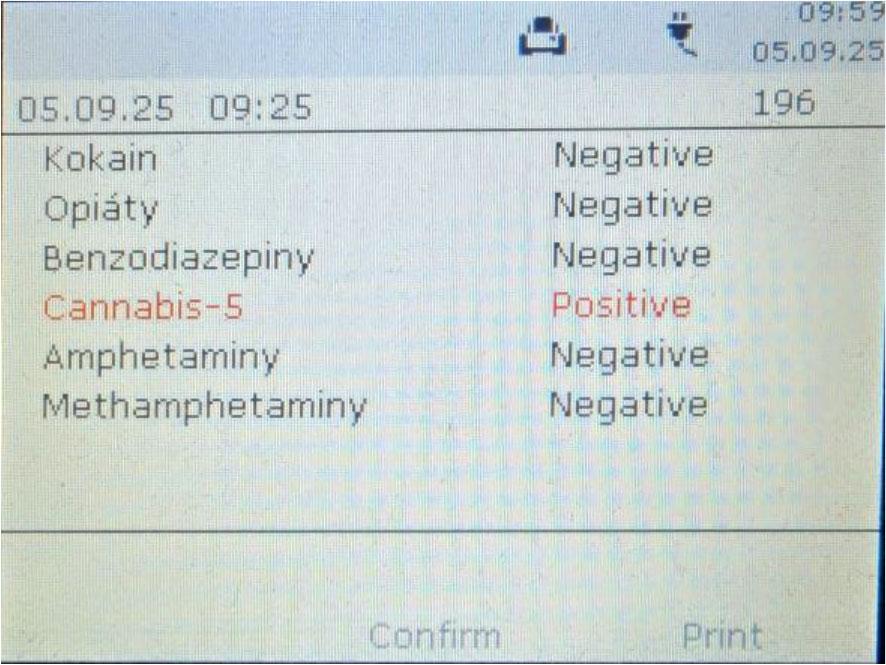
Results presentation by the DDT 5000 analyzer.
For the same reason, it is not possible to determine the detection limit directly. Therefore, a procedure was chosen to identify two closest concentrations, with the lower one identified by the device as negative and the higher one as positive. Based on these data, it can be concluded that the detection limit lies within the given concentration interval.
To verify the composition of reference materials, a gas chromatograph with a quadrupole combined with a time-of-flight analyzer (GC/Q-TOF) was selected. This method also allows verification of the composition of prepared mixtures using the National Institute of Standards and Technology (NIST) spectral libraries. The analysis of selected addictive substances is considerably lengthy and takes several hours, mainly due to the presence of benzodiazepines in the mixture, with Alprazolam exhibiting the greatest retention on the column. It eluted only at the 189th minute of the analysis (see Fig. 6.).
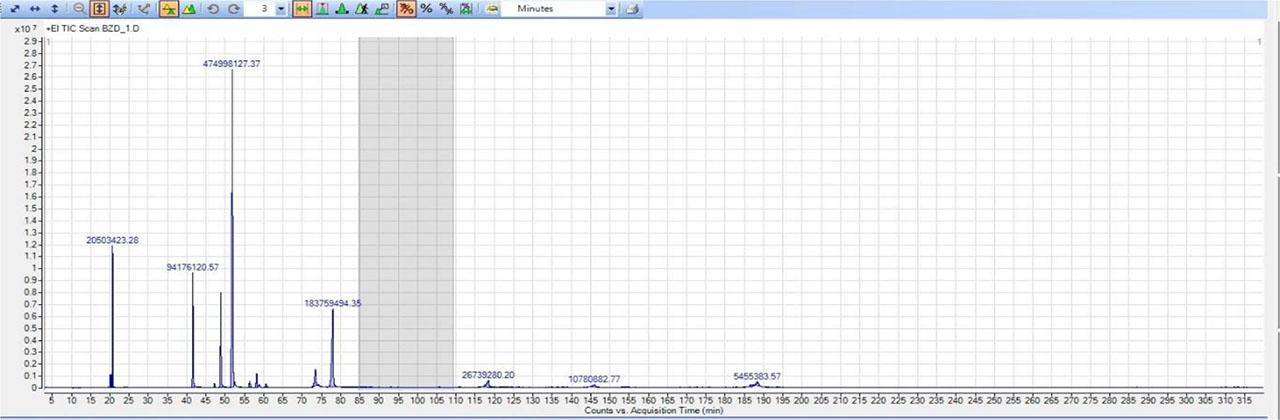
Result from analysis of benzodiazepines reference material.
The final method used for the analysis is presented in Table 7.
Parameters of analytical method.
| Parameter | Value |
|---|---|
| Initial temperature | 80 °C |
| Initial hold time | 15 min |
| Ramp | 10 °C/min |
| Final temperature | 200 °C |
| Final hold time | 180 min |
| Carrier gas flow | 1.0 ml/s |
| Injection | 2 µl |
| Column | HP-5ms; 30 m; 0.32 mm; 25 µm |
Each concentration of all substances was measured three times using the same reference solution at the specified concentration. The reported limits are the average of the highest concentration evaluated as negative and the lowest concentration evaluated as positive. For all substances, at least the detection limits specified by the manufacturer were confirmed. For most drugs, the limits determined during measurements were below these values. Ketamine was the only substance with an official detection limit in the hundreds of nanograms per milliliter – specifically, 300 ng/ml. In comparison, the detection limit estimated by measurement was an acceptable 157.2 ng/ml (see Fig. 7). Thus, the real detection limit appears to be approximately half of the official detection limit.
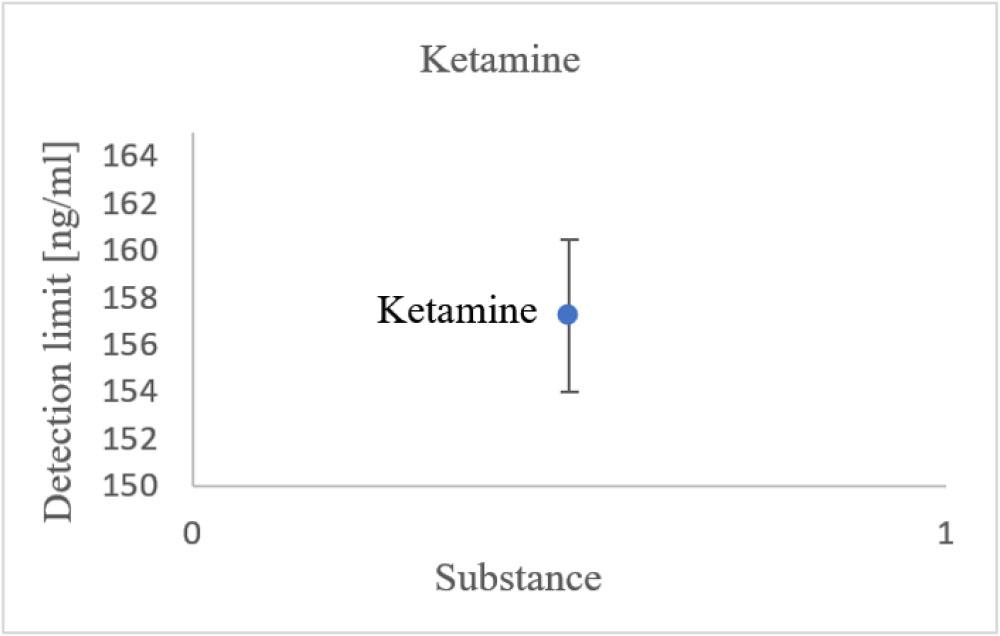
Estimated detection limit of ketamine.
The detection limits for benzodiazepines and methadone were relatively close to the values specified by the manufacturer. Amphetamine was slightly further from the declared limit. For only one substance, heroin, the determined limit was higher than the manufacturer’s value. In this case, the result may have been influenced by the uncertainty of the reference solution (see Fig. 8). It can be stated that heroin's detection limit is at the level defined by the manufacturer.
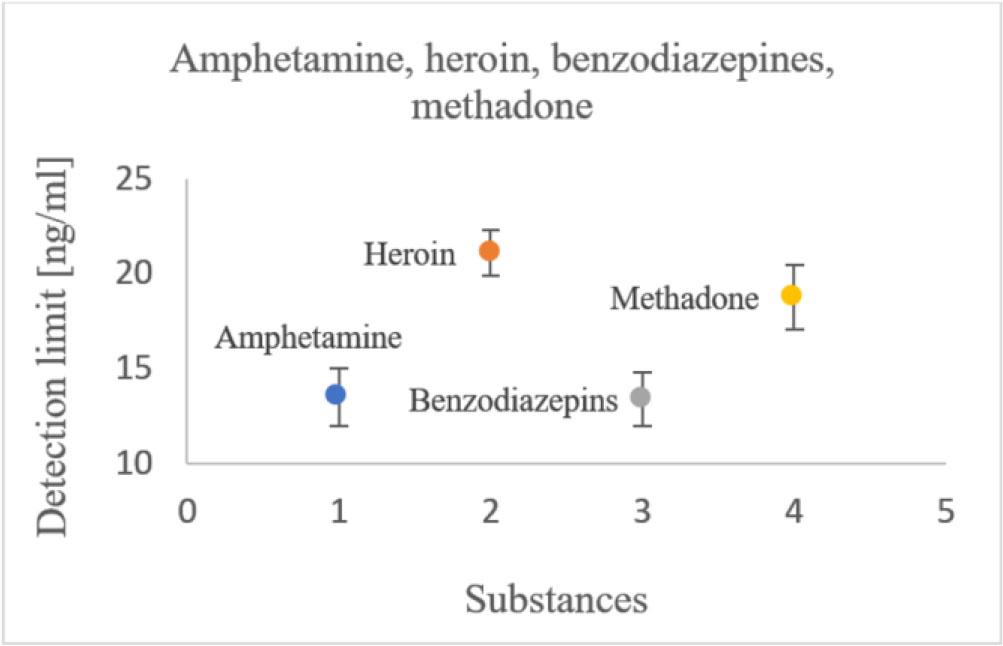
Estimated detection limits for amphetamine, heroin, benzodiazepines, methadone.
Detection limits for methamphetamine and cocaine were also below the values specified by the manufacturer (see Fig. 9). The limits given for these drugs by the manufacturer are low; the estimated limits were even lower, yet still acceptable.
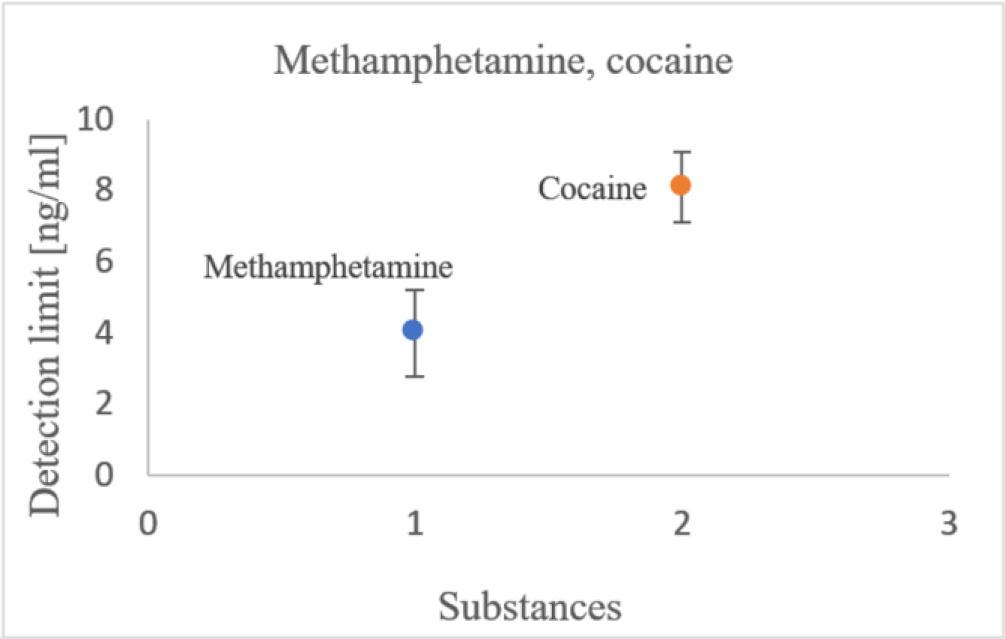
Estimated detection limits for methamphetamine and cocaine.
All previous results are acceptable and consistent with other research results. However, the results obtained during testing of detection limits for THC are questionable. The first concern is the minimal influence of device settings on the detection limits. The limits specified by the manufacturer are 5/10/25 ng/ml. The lowest value is five times smaller than the highest and the middle value. The differences among the measured results follow this trend: the lowest is approximately half of the middle value. The difference between the highest and middle value is twice as large as expected. This is still acceptable, especially considering the uncertainty of the reference materials. However, the actual level of the detection limits is questionable. The estimated limits are 0.59 ng/ml, 0.07 ng/ml, and 0.03 ng/ml. These values are not only significantly lower than the manufacturer's limits but also very low in general. Preparation of the reference solutions required more than two dilutions of the original reference materials, resulting in higher uncertainty for these mixtures than their concentration. The testing was repeated with similar results, but further verification will be necessary. The uncertainty of the detection limit does not arise solely from the reference material; a substantial part comes from the method of its determination. As mentioned, the detection limit is defined as the mean of the boundary values of the interval, which is delimited by the lowest concentration evaluated as positive and the highest concentration evaluated as negative. This may cause the relatively higher uncertainties of the detection limits, particularly for THC. Half of this interval is included as a component of the detection limit uncertainty. For THC, the combination with multiple dilutions of the reference materials may account for the relatively high uncertainties. Future work will aim to narrow the intervals in which the detection limits are located, thereby reducing their associated uncertainty.
Overall, the results indicate that the DDT 5000 has the potential to provide more than just indicative information when a prohibited substance is present in the human body. The capabilities of this device have been confirmed in previous research. Although it will require significant time and effort, it may be possible to establish formal rules for using this device as an evidential tool. This approach requires zero tolerance for addictive substances, as the device only confirms the presence of a given drug in the sample. Nevertheless, it remains a highly valuable tool for detecting addictive drugs in the human body, not only for checking drivers but also for protecting health.
All results obtained are acceptable, with no reason for doubt. Only in two cases would it be suitable to verify the results. The first case is heroin where the estimated detection limit appeared slightly higher than the limit stated by the manufacturers. This could be due to the uncertainty of the prepared reference materials. However, in all other cases, the estimated limits were slightly lower than the detection limits specified by the manufacturer. The second case was THC, where the measured detection limits were unexpectedly low. For THC, the tested device offers three possible detection settings. Among these settings, some differences in detection limits would be expected, but the observed variations were smaller than anticipated.