Eimeria are the absolute gut parasites belonging to phylum Apicomplexa, causing the gut disorders known as coccidiosis (Aljohani, 2024). These parasites reproduce in the intestinal epithelium and cause diarrhea, decreased digestibility, and ultimately reduced growth and death of the bird (Lee et al., 2022). The widespread use of intensive farming systems has made coccidiosis control a significant problem in poultry farms. It has been estimated that £7.7 to £13.0 billion are spent annually in seven countries on the prevention and treatment of avian coccidiosis (Blake et al., 2020). For this reason, researchers and breeders are constantly seeking new and effective methods and substances to manage coccidiosis. The use of coccidiostats/coccidiocidals to prevent coccidiosis has been common and has been used for a long time. However, with the misuse of these drugs, Eimeria has developed resistance to these drugs (Hayajneh et al., 2024). Recently, raising poultry without the use of any drugs is a new approach that aims to produce products free of drug residues (which negatively affect human health and cause the development of more drug-resistant strains) (Hailat et al., 2024). As a result, scientists are looking to assess the effectiveness of various alternatives such as herbal compounds. Many studies have examined the effects of medicinal plants on broiler performance and health (Fahmy et al., 2025). In addition, in recent years, numerous studies have explored the impact of various herbal compounds on coccidiosis control in birds, with promising results (Edleeb et al., 2025; Aljohani and Zaman, 2024; Saeed et al., 2023; Abd-ELrahman et al., 2022). Some researchers have even reported the beneficial effects of medicinal plants (such as Chenopodium quinoa seeds extract) in improving lung diseases in other animals (Majeed et al., 2024). These plant-based additives inhibit pathogen growth through competitive exclusion mechanisms and immune system activation. Additionally, they enhance nutrient digestibility and contribute to lowering blood cholesterol levels (Lee et al., 2003). These effects have been attributed to greater secretion of digestive enzymes (Upadhaya et al., 2019). Moreover, via penetrating the coccidia oocyst wall, tannins destroy its cytoplasm and probably deactivate the endogenous enzymes responsible for the sporulation cycle in chickens. As a result, oocyst numbers will decrease in birds (Muthamilselvan et al., 2016).
The green hull of pistachio (Pistacia vera) is a major by-product, accounting for over 60% of total pistachio residues (Arjeh et al., 2020). This component is particularly rich in bioactive compounds, with phenolic compounds being the most abundant (Rafiee et al., 2018). Pistachio green hull comprises 23.00% dry matter, 11.00% crude protein, 15.00% crude fiber, 12.00% ash, 6.00% crude fat, and 55.50% nitrogen-free extract (Shakerardekani and Molaei, 2020). Various studies have assessed some of the functional attributes of the pistachio green hull extract (PHE) such as phenolic materials (Seifzadeh, et al., 2019), antimicrobial (Rajaei et al., 2010) and antioxidant (Abolhasani et al., 2018) activities and its biological benefits on human health (Sila et al., 2014). Our previous research investigated the impact of PHE on broiler performance, gut morphology, and antioxidant capacity. The findings indicated that the PHE ameliorated the antioxidant capacity and performance, and decreased the excreta oocytes and lesion scores during the coccidiosis challenge (Noruzi et al., 2024). Limited research has been conducted on incorporating pistachio by-products into poultry diets (Ahmadi Kohanali et al., 2022; Hosseini-Vashan et al., 2020; KordZanganeh et al., 2018). It is important to highlight that none of these studies involved a coccidiosis challenge. Given the availability of pistachio by-products in Iran and their properties, this study was conducted to evaluate the anticoccidial properties of PHE in broiler chickens, focusing on carcass traits, meat quality, serum lipid parameters, and ileum microbiology.
In this study, 576 male Ross 308 broiler chicks, aged one day, were purchased from a regional supplier and raised in pens for 42-day period. Each pen (1m × 1m) housed twelve chicks. Throughout the starter phase (1–10 days) and the grower phase (11–24 d), the experiment had four treatments including 1) control (without PHE), 2) 200 ppm PHE, 3) 300 ppm PHE, and 4) 400 ppm PHE, with 12 replicates per treatment. After the grower period (25 days), the birds in each treatment were distributed into two separate groups: infected and uninfected. Treatments comprised 1) without PHE-uninfected (0-No), 2) 200 ppm PHE-uninfected (200-No), 3) 300 ppm PHE-uninfected (300-No), 4) 400 ppm PHE-uninfected (400-No), 5) without PHE-infected (0-Yes), 6) 200 ppm PHE-infected (200-Yes), 7) 300 ppm PHE-infected (300-Yes) and 8) 400 ppm PHE-infected (400-Yes) (with six replicates). Regarding Eimeria species challenge, at day 25, one-half of the broilers were orally administered a 20x dose of trivalent live attenuated vaccine (Livacox T, Biopharm Co., Prague, Czech Republic). Each dose of the vaccine (0.01 ml) contained between 300 and 500 sporulated oocysts each of Eimeria acervulina, Eimeria maxima, and Eimeria tenella. Prior to oral administration, distilled water was added to the vaccine to reach a final volume of 1 ml per bird, corresponding to a 20× standard dose. The control groups were administered 1 ml of distilled water (any anticoccidial drugs were not used in this study). Table 1 shows the dietary ingredients and their nutrient composition (NRC, 1994), which were formulated according to the Aviagen (2022) catalog. For the first three days of the birds' life, the house temperature was maintained at 32°C, then decreased by 3°C each week until it reached 21°C, where it remained constant for the duration of the study. Relative humidity was kept between 50% and 60% throughout the experiment. A light/dark cycle of 18 hours of light and 6 hours of darkness was implemented during the trial, and birds had continuous access to feed and water.
Ingredients and nutrient composition of the experimental diets (as-fed basis)
| Ingredients (%) | Starter (1–10 d) | Grower (11–24 d) | Finisher (25–42 d) |
|---|---|---|---|
| Corn | 49.14 | 53.96 | 60.75 |
| Soybean meal (44%) | 42.23 | 38.08 | 32.12 |
| Soybean oil | 4.25 | 4.36 | 3.84 |
| Limestone | 0.97 | 0.72 | 0.67 |
| Dicalcium phosphate | 2.04 | 1.61 | 1.32 |
| Vitamin premix1 | 0.25 | 0.25 | 0.25 |
| Mineral premix2 | 0.25 | 0.25 | 0.25 |
| L-Lysine HCl | 0.07 | 0.02 | 0.07 |
| DL-Methionine | 0.29 | 0.24 | 0.23 |
| L-Threonine | 0.01 | - | - |
| Choline | 0.05 | 0.05 | 0.05 |
| Sodium Bicarbonate | 0.11 | 0.11 | 0.11 |
| Salt (NaCl) | 0.35 | 0.35 | 0.34 |
| Nutrient composition (%) | |||
| Metabolizable energy (kcal/kg) | 2975 | 3050 | 3100 |
| Crude protein | 23 | 21.50 | 19.50 |
| Lysine | 1.32 | 1.18 | 1.08 |
| Methionine + Cystine | 1.00 | 0.92 | 0.86 |
| Threonine | 0.88 | 0.80 | 0.72 |
| Calcium | 0.95 | 0.75 | 0.65 |
| Available Phosphorus | 0.50 | 0.42 | 0.36 |
| Sodium | 0.18 | 0.18 | 0.18 |
| Potassium | 0.99 | 0.92 | 0.82 |
| Chlorine | 0.26 | 0.25 | 0.26 |
Provided the followings per kg of diet: vitamin A (trans-retinyl acetate), 13000 U; vitamin D3 (cholecalciferol), 5000 U; vitamin E (D L-α tocopherol acetate), 80 U; vitamin K (menadione), 4 mg; riboflavin, 9 mg; pantothenic acid (D-Ca pantothenate), 25 mg; pyridoxine (pyridoxine-HCl), 5 mg; thiamine, 5 mg; vitamin B12 (cyanocobalamin), 0.02 mg; biotin, 0.35 mg; folic acid, 2.5 mg; nicotinic acid, 70 mg; ethoxyquin (antioxidant), 2.5 mg.
Provided the following per kg of diet: Fe, 20 mg; Zn, 120 mg; Mn, 120 mg; Cu, 16 mg; I, 1.25 mg; Se, 0.30 mg.
Pistachio green hulls of the Ahmadaghaei variety were sourced from Fakhrabad, Bajestan, Iran. The pistachio hulls were dried, powdered, and kept at −80°C until further analysis. One gram of ground pistachio green hull was mixed with water in a liquid-to-solid ratio of 15:1 and incubated at 25°C for 8 hours, as described by Rajaei et al. (2010). The total phenolic content was determined using the Folin–ciocalteu colorimetric method (Waterhouse, 2002). The calculations were performed using a calibration curve prepared with gallic acid. The total phenolic content was reported as milligrams of gallic acid equivalents per gram of dry weight. The total tannin content was determined by subtracting the non-tannin phenols and precipitating the tannins using insoluble polyvinyl pyrrolidone. The total tannin content was determined by subtracting the non-tannin phenols from the total phenols (Makkar et al., 1995). The total flavonoid content was quantified using gallic acid as the standard, as described by Heimler et al. (2005). The values of total phenol, total tannin, and flavonoid compounds in PHE were 91.32 mg gallic acid equivalents per gram, 32.03 mg gallic acid equivalents per gram and 30.46 mg catechin equivalents per gram of dry pistachio hull, respectively. The dried pistachio green hull powder contained 3931.67 kcal/kg of gross energy, 94.16% dry matter, 12.14% crude protein, 5.81% crude fat, 15.16% crude fiber, and 11.98% crude ash, as reported by AOAC (2005).
The performance data was reported in our previous study (Noruzi et al., 2024). To avoid repetition, we have omitted them in this paper. At 42 days of age, two birds from each pen were euthanized via cervical dislocation; subsequently, carcass components were dissected and weighed to analyze relative weights. The carcass and organ weights, including breast, thigh, liver, abdominal fat, bursa of Fabricius, thymus, and spleen, were measured relative to live weight.
Two broilers, representing the average body weight of each replication, were chosen and euthanized by cervical dislocation at 42 d of age. To assess meat quality parameters, the right breast muscle was excised and stored at 4°C. The pH levels of the meat samples at 45 minutes and 24 hours after death were measured by a pH meter (WTW pH 330i model). To evaluate the color values of the meat (lightness L*, redness a*, and yellowness b*) A CR410 chromameter (Konica Minolta Sensing Inc., Japan) was used (24 h postmortem). A 2×2×5 cm piece of breast meat was weighed, placed in a plastic bag, and kept suspended at 4°C. After 24 hours, the samples were blotted dry and reweighed. Drip loss percentage was determined by the formula: [(initial weight − final weight) ÷ initial weight] × 100.
Breast meat samples were weighed, sealed in zip-locked plastic bags, and cooked in a water bath at 85°C. After 30 minutes, they were cooled at 4°C, blotted dry on filter paper, and reweighed. The percentage of cooking loss was determined using the formula: [(initial weight − final cooked weight) ÷ initial weight] × 100.
Shear force was determined using a Texture Analyzer (TA.XT plus, Stable Micro Systems, UK) on the cooked samples (Zhang et al., 2014). The water-holding capacity (WHC%) of breast meat was determined after overnight thawing at 4°C, following the method of Wilhelm et al. (2010).
At 42 days of age, blood samples were collected from the wing vein of two birds per pen. Serum was separated by centrifuging the samples at 2000×g for 10 minutes. Afterward, serum samples were kept at −20°C until they were analyzed for lipid profile assessment. Serum triglycerides, total cholesterol, and high-density lipoprotein cholesterol (HDL) were measured using laboratory kits (Pars Azmun, Tehran, Iran) via spectrophotometer (UV-VIS 2100, Unico, USA). Very low-density lipoprotein cholesterol (VLDL) was calculated by dividing the triglyceride concentration by 5, while low-density lipoprotein cholesterol (LDL) was determined using the equations of Friedewald et al. (1972).
At 42 days of age, two chickens per pen were randomly selected and slaughtered to evaluate the ileal microbial population. Subsequently, one gram of ileal digesta was obtained from each bird, diluted, and homogenized in physiological serum. Lactobacillus and Coliform populations were enumerated using MRS Agar and MacConkey Agar, based on the method described by McCartney et al. (1996). The total bacterial count was reported as log10 CFU/g.
This study was carried out using a completely randomized design (CRD) during the starter and grower phases, with 4 treatments and 12 replicates per group. During the finisher phase (challenge period), data were analyzed using CRD in a factorial arrangement (4×2: four levels of PHE (0, 200, 300, 400 ppm) and two conditions (uninfected and infected broilers, No and Yes)) with six replicates. The General Linear Model (GLM) procedure of SAS 9.4 (2012) software was applied, and the results were expressed as means ± standard error of the mean (SEM). Tukey's test was employed for mean comparisons (P<0.05).
Eimeria infection significantly reduced carcass, breast, thigh, and spleen weight percentages and increased bursa of Fabricius and thymus relative weights (P<0.05) (Figure 1). For main effects of PHE, broilers fed 200 ppm of PHE showed a significantly lower abdominal fat percentage compared to the control group (without PHE) (P<0.05) (Figure 2). No significant differences were observed for the other effects (P>0.05).
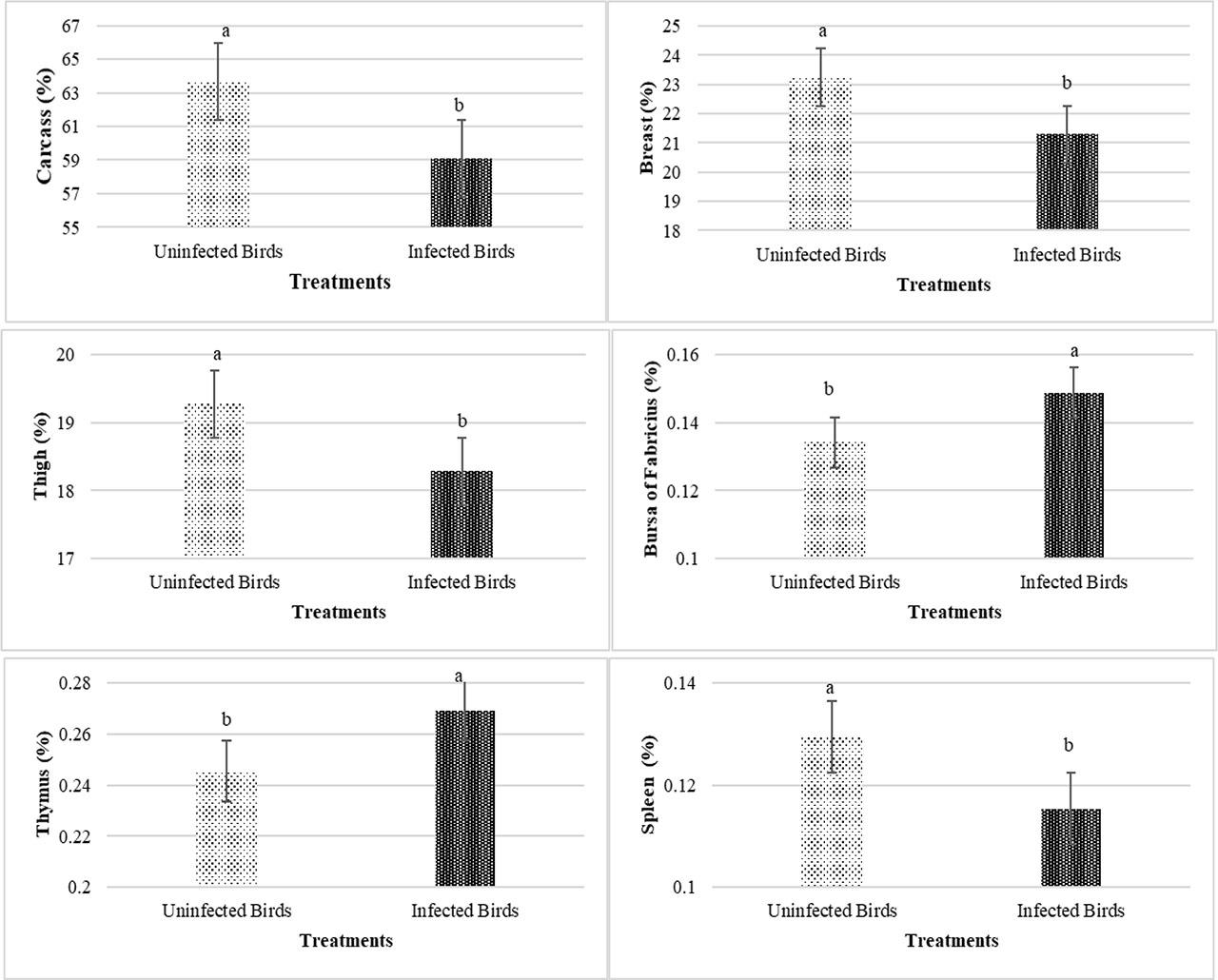
Main effects of Eimeria infection (uninfected vs. infected) on percentages of carcass, breast, thigh, bursa of Fabricius, thymus and spleen weight of broiler chickens at 42 d of age (P<0.05)
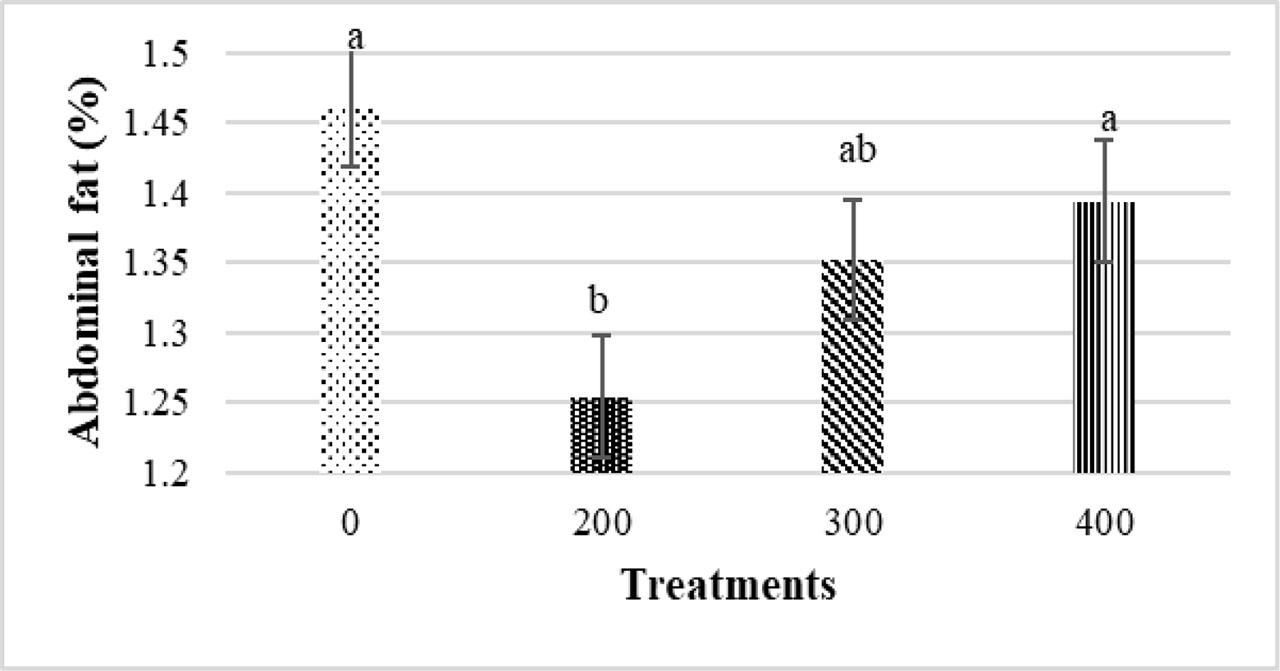
Main effects of different levels of pistachio green hull aqueous extract (0, 200, 300, 400 ppm) on abdominal fat percentage of broiler chickens at 42 d of age (P<0.05)
Figures 3 and 4 present the results related to the quality of chicken breast meat. Lightness (L*) increased as PHE levels in the diet rose, while drip loss and cooking loss significantly decreased (P<0.05) (Figure 3). The highest L* and lowest drip and cooking losses were observed in the 300 and 400 ppm PHE treatments, respectively. Regarding the main effect of infection, infected birds had significantly higher drip loss values in their breast meat compared to uninfected birds (P<0.05) (Figure 4). Other effects did not show significant results (P>0.05).
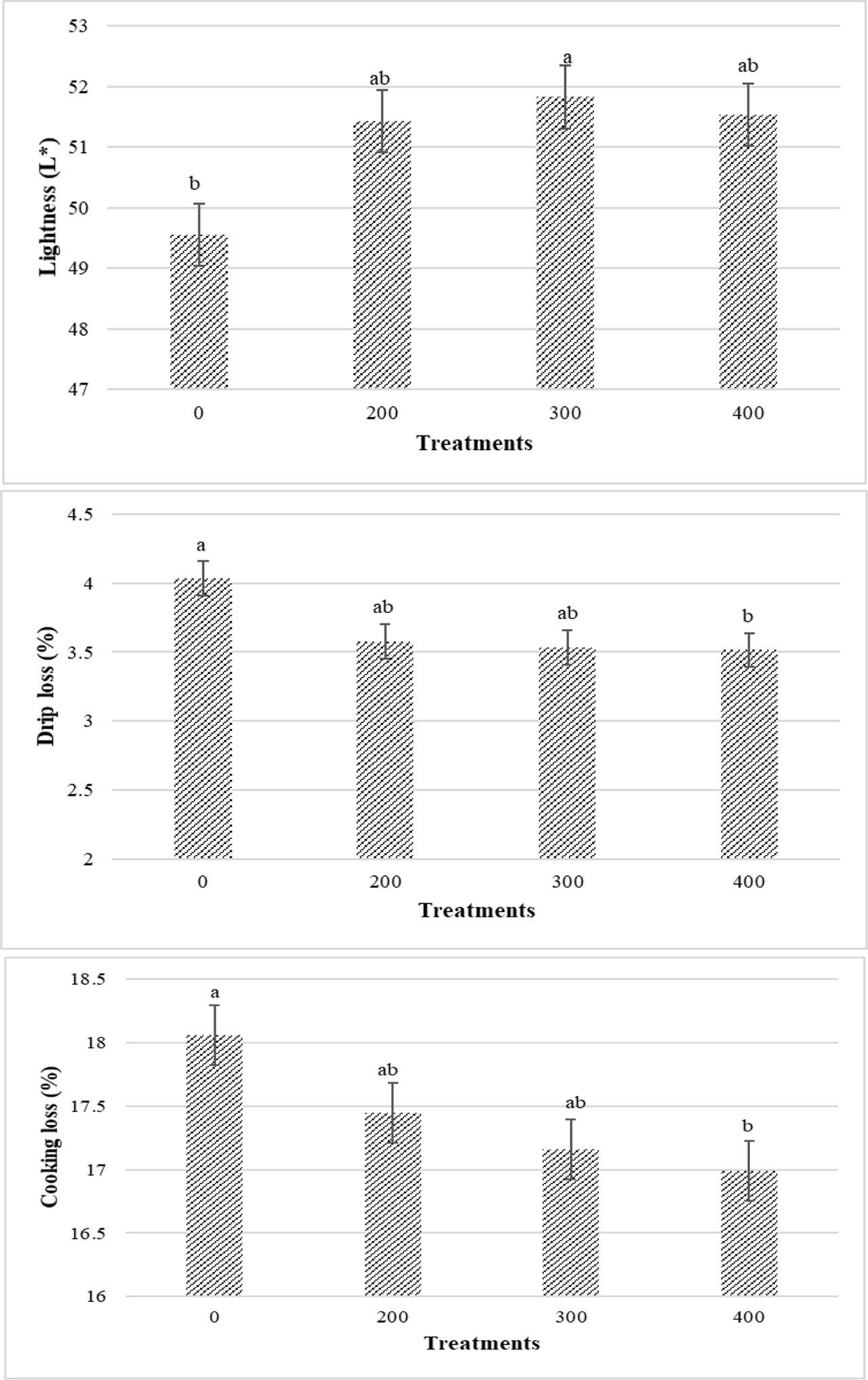
Main effects of different levels of pistachio green hull aqueous extract (0, 200, 300, 400 ppm) on breast meat lightness, drip loss and cooking loss of broiler chickens at 42 d of age (P<0.05)
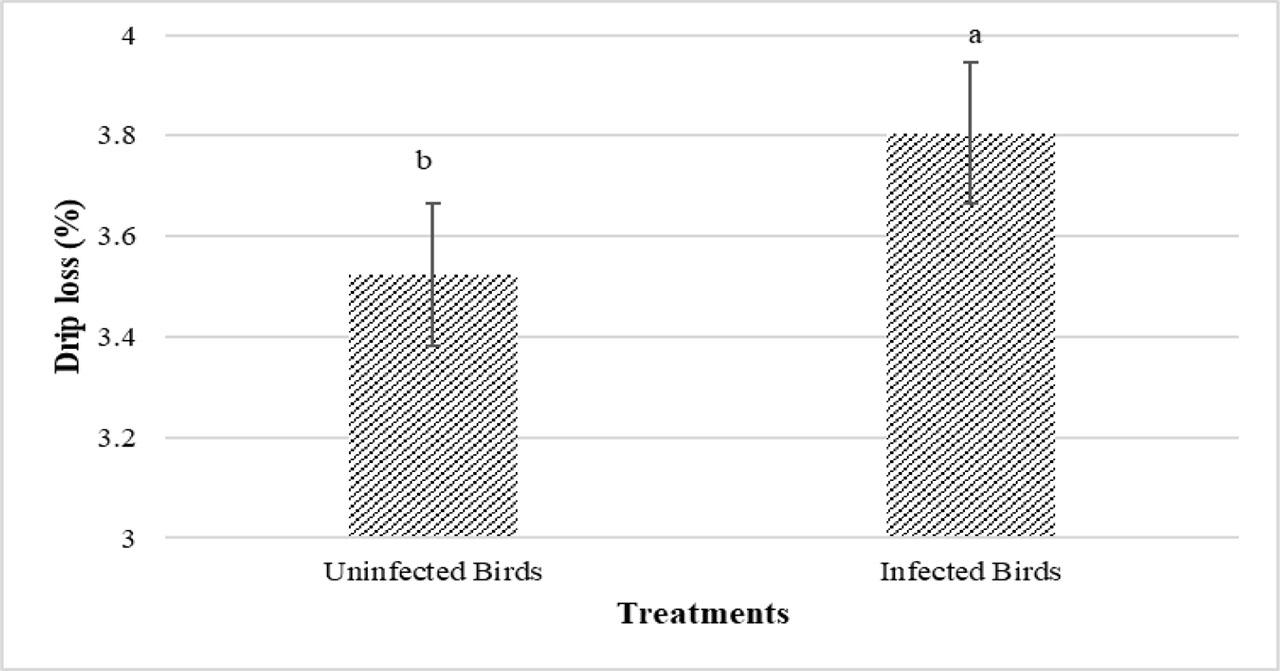
Main effects of Eimeria infection (uninfected vs. infected) on breast meat drip loss of broiler chickens at 42 d of age (P<0.05)
The use of 300 and 400 ppm of PHE in the broiler's diet notably reduced total cholesterol levels in serum compared to the control group (without PHE) (P<0.05), while the use of 200 ppm did not show a significant difference with other treatments (0, 300 and 400 ppm) (Figure 5). As shown in Figure 6, regarding the interaction effects of PHE and infection, serum total cholesterol levels in the 400-No group were considerably lower than the 0-No and 200-No groups (P<0.05). However, the 400-No treatment did not show significant differences compared to the other treatments (300-No, 0-Yes, 200-Yes, 300-Yes and 400-Yes) (P>0.05). In relation to serum HDL levels, birds consuming diets containing 300 and 400 ppm of PHE indicated the highest HDL content, which was significantly different from the control (without PHE) (P<0.05) (Figure 5). The treatments did not significantly affect the other parameters.
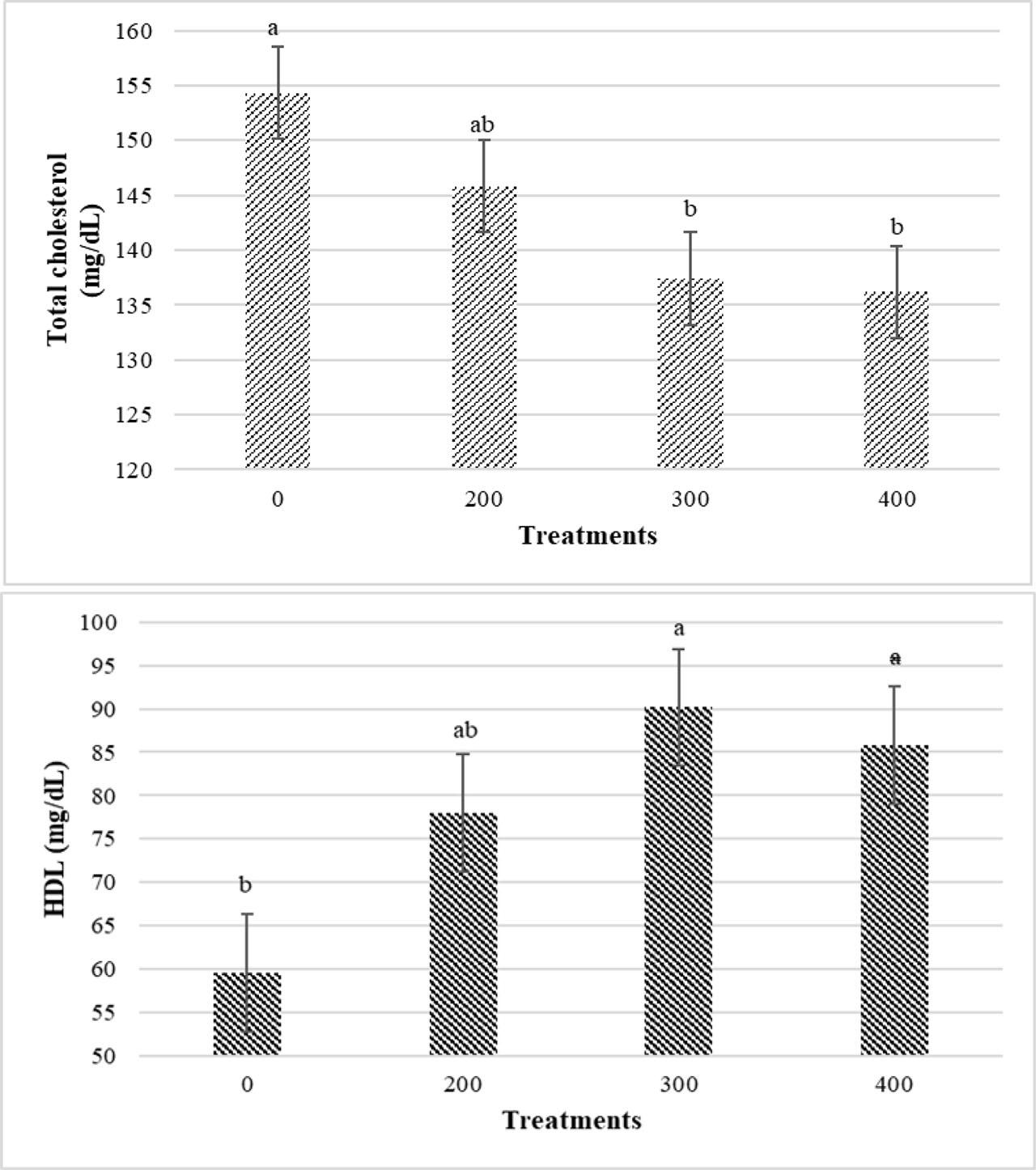
Main effects of different levels of pistachio green hull aqueous extract (0, 200, 300, 400 ppm) on serum total cholesterol and high-density lipoprotein cholesterol (HDL) levels of broiler chickens at 42 d of age (P<0.05)
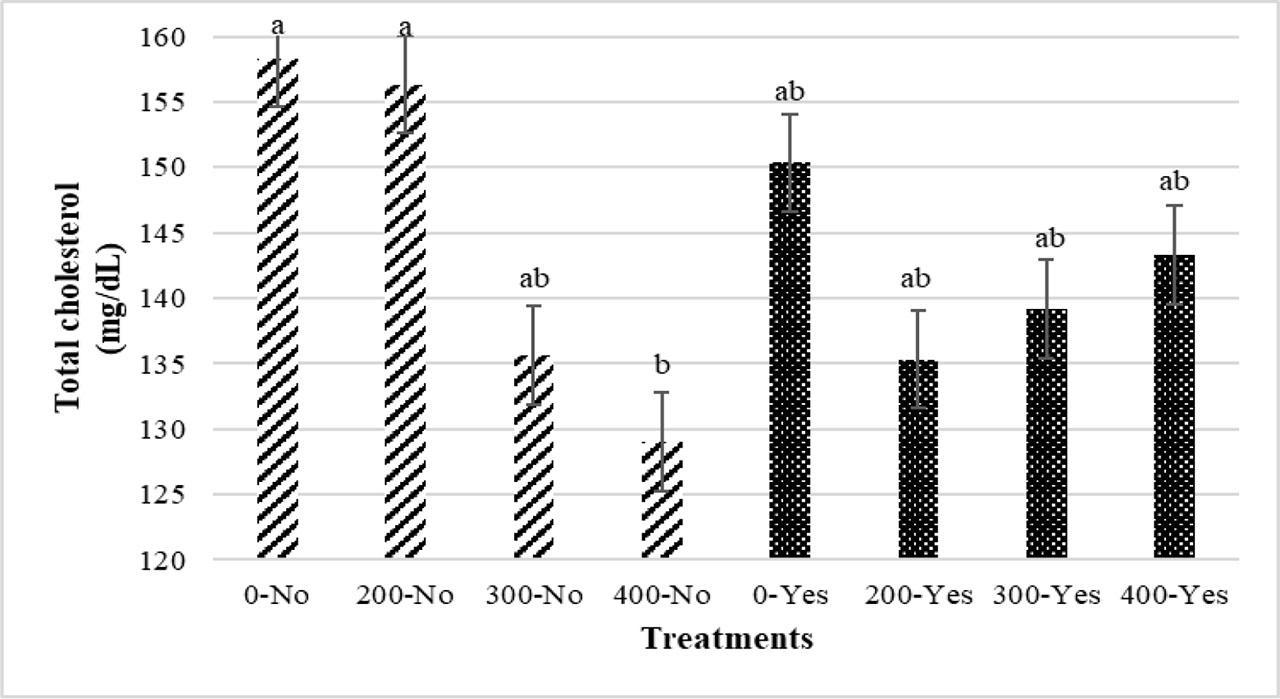
Interaction effects of different levels of pistachio green hull aqueous extract (0, 200, 300, 400 ppm) and Eimeria infection on serum total cholesterol level of broiler chickens at 42 d of age. No, uninfected birds; Yes, infected birds (P<0.05)
The results of the effects of PHE and infection on the ileal microbial population are presented in Figures 7 and 8. The challenge significantly increased the number of Coliforms (P<0.05) (Figure 7). The number of ileal Coliforms in treatments 0-No, 200-No, 400-No, and 300-Yes was significantly lower than 0-Yes (P<0.05) (Figure 8). The highest number of Coliforms was obtained in 0-Yes. There was no significant impact of the experimental treatments on the Lactobacillus population (P>0.05).
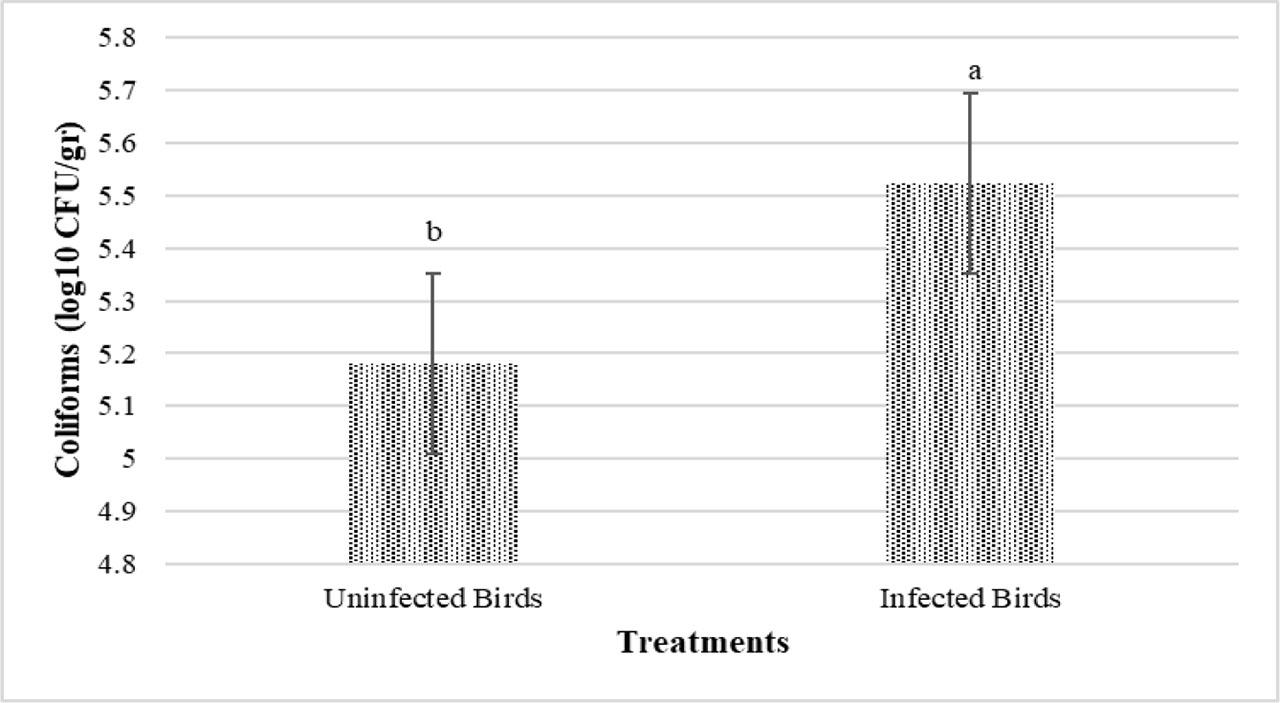
Main effects of Eimeria infection (uninfected vs. infected) on ileum Coliforms population of broiler chickens at 42 d of age (P<0.05)
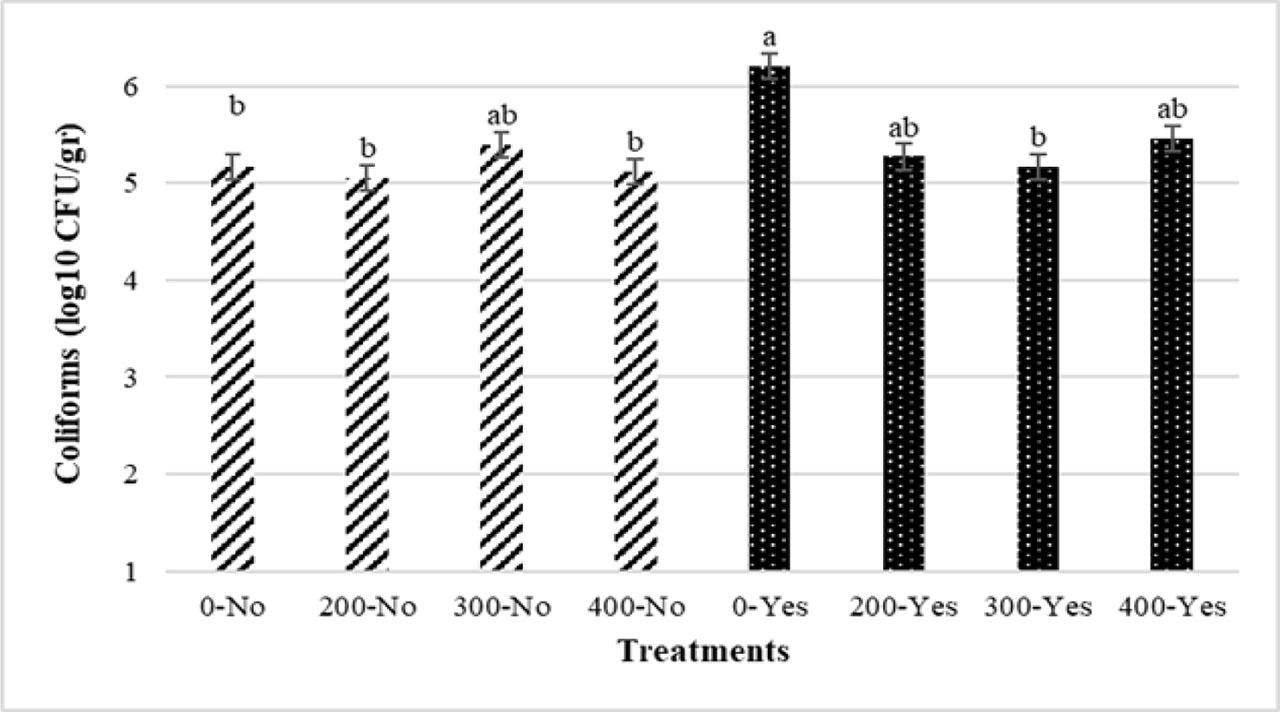
Interaction effects of different levels of pistachio green hull aqueous extract (0, 200, 300, 400 ppm) and Eimeria infection on ileum Coliforms population of broiler chickens at 42 d of age. No, uninfected birds; Yes, infected birds (P<0.05)
It has been proven that plants rich in antioxidant substances such as phenols, flavonoids, tannins, and saponins have been applied as alternative agents for the treatment of avian coccidiosis and have shown strong anticoccidial effects (Hussain et al., 2021). Due to the limited studies on the use of pistachio by-products in birds and its widespread cultivation in Iran, the authors decided to design this experiment. Previous studies have shown that coccidiosis in broilers, due to stimulation of the immune response, leads to lower feed consumption and reduced body weight gain (Cai et al., 2023; Beski, 2023; Cha et al., 2020). As a result of the metabolic disorders associated with coccidiosis, broilers consumed fewer nutrients (Choi et al., 2023), resulting in reduced meat performance, body weight and carcass weight (Oikeh et al., 2019). Our previous study showed that the coccidiosis significantly decreased body weight gain and feed intake in broilers (Noruzi et al., 2024). Therefore, this decrease in the relative carcass, breast, and thigh weights can be attributed to a decrease in the bird's growth performance (Noruzi et al., 2024).
Alsulami and El-Saadony (2023) observed similar results regarding relative carcass and breast weight. On the other hand, Upadhyaya et al. (2019) stated that plant extracts improved the performance of coccidiosis-infected birds by increasing nutrient digestibility (which itself is due to increased secretion of digestive enzymes). Almahallawi et al. (2024) also observed that birds treated with flaxseed essential oil (during coccidiosis challenge) had a positive response in a dose-dependent manner. They reported the best results at a concentration of 3% flaxseed essential oil. The thymus, spleen, bursa of Fabricius, and liver are considered important immune organs in poultry and their relative weight changes often reflect the immune status of the birds (Das et al., 2021). In the present study, coccidiosis enhanced the relative weights of the bursa of Fabricius and thymus, while it decreased the spleen's relative weight. Consistent with the current findings, Jelveh et al. (2023) also observed that the relative weight of the bursa in challenged birds was significantly higher than in unchallenged birds. It has been suggested that the increase in organ weights could be due to immune stimulation from the coccidiosis vaccine, which causes more nutrients to be consumed for antibody production and immune organ development (Liu et al., 2015). In our previous study (Noruzi et al., 2024), we measured serum NO concentration during coccidiosis challenge. Our results revealed that the levels of NO was considerably higher in infected birds. Nevertheless, NO amounts in chickens that consumed 300 and 400 ppm PHE were significantly decreased. This outcome can confirm the strong antioxidant property of PHE and its effectiveness (Noruzi et al., 2024). In contrast, Alqhtani et al. (2023) observed a decrease in bursa and thymus weights in birds infected with Eimeria. However, some studies did not report a significant effect of challenge on the weight of immune organs and other carcass components (Qaid et al., 2022). In the current trial, the percentage of abdominal fat reduced significantly in broilers consumed 200 ppm of PHE. However, 300 and 400 ppm of PHE did not show significant differences from the control (without PHE). In line with the current outcomes, Kishawy et al. (2019) declared that the use of the pomegranate peel extract in the diet of broiler chickens significantly reduced the amount of abdominal fat. They suggested that pomegranate peel extract or pomegranate leaf extract had an inhibitory impact on lipid metabolism and consequently reduced body fat deposition due to its hypocholesterolemic and hypolipidemic effects (Kishawy et al., 2019). Since PHE is rich in flavonoids, its phenolic compounds are expected to inhibit adipose tissue growth (via anti-angiogenic mechanisms and metabolic regulation), leading to a decrease in abdominal fat percentage (Gonzalez-Castejon and Rodriguez-Casado, 2011). Some studies attributed the reduction in abdominal fat to a decrease in the activity of the pancreatic lipase enzyme and activation of the adenosine monophosphate protein kinase cascade system, which is a main enzyme in fat metabolism (Park et al., 2014). The discrepancy between the results is attributed to the differences in the unique nature of each herb species, their distinguished chemical characteristics, and different environmental situations. In addition, the choice of extraction solvent and testing method can significantly affect the concentration and classification of secondary bioactive compounds in the plant (Abubakar et al., 2024).
Poultry products are considered one of the most economical and sustainable sources of animal protein. Evaluation of poultry meat quality includes basic parameters such as taste, tenderness, color, shelf life, and consumer health factors, including fat content, fatty acid composition, cholesterol level, and bioactive compound profile (North et al., 2019). Polyphenols in medicinal plants have been shown to improve meat quality due to their antioxidant properties (Xie et al., 2022). Qaid et al. (2023) observed that the L*, a*, and b* parameters increased in the Rumex nervosus leaf powder group (3 g/kg). They stated that this herb can act as a natural additive in poultry feed to improve meat quality. They attributed this improvement to natural antioxidants, such as phenolic, and flavonoids such as gallic acid in Rumex nervosus. Since the PHE used in the present study also contains phenolic compounds, the increase in L* can be attributed to these compounds. Herbal compounds ameliorate meat quality by increasing healthy polyunsaturated fatty acids, reducing lipid oxidation, and improving meat shelf life (Galli et al., 2020). The increase in L* values can occur due to alterations in surface reflectance, resulting in denaturation and aggregation of myofibrillar protein (Kruk et al., 2011). In the present study, higher levels of PHE significantly reduced drip loss and cooking loss. It has been stated that the phenols and flavonoids present in Moringa oleifera interacted with muscle proteins, increased cross-linking, inhibited oxidation, and maintained moisture during meat cooking, ultimately leading to reduced cooking loss in broiler meat (Akib et al., 2024). In agreement with our result, Rehman et al. (2018) reported similar results. Drip loss values were considerably higher in infected broilers compared to uninfected ones. Čobanović et al. (2024) reported that coccidia challenge enhanced drip loss values. Indeed, coccidiosis acts as a stressor in chicken organs, causing inflammation and oxidative stress, and changing postmortem metabolism in skeletal muscle, resulting in a negative impact on the meat quality (Čobanović et al., 2024). Various studies have reported the negative effect of Eimeria species in reducing meat WHC and drip loss and color traits (Qaid et al., 2022; Qaid et al., 2021; Jaramillo et al., 2021; Partovi et al., 2019). It has also been reported that the protein content in the meat of infected birds was low (Partovi et al., 2019). Because parasites, by affecting the digestive tract, reduce digestion and absorption of nutrients, this leads to reduced protein synthesis and increased degradation (Partovi et al., 2019). Water is bound to protein molecules in meat, this reduced protein synthesis could explain the increased drip loss (Van Laack, 1999).
It has been reported that the Acacia nilotica aqueous extract decreased triglyceride and total cholesterol amounts in uninfected and infected broiler chickens (during coccidia challenge) (Eldeeb et al., 2025). This effect has been attributed to epicatechin in the extract, stating that this effective compound reduced pancreatic lipase activity in the digestive tract and ultimately inhibited fat absorption (Eldeeb et al., 2025). In addition to regulating fat absorption, epicatechin also acts by controlling transcription factors expressed during triglyceride and cholesterol synthesis (Eldeeb et al., 2025). In this regard, we found that the total cholesterol level in the 400-No group was significantly lower than the 0-No group. In addition, treatments containing 300 and 400 ppm of PHE had the highest HDL content compared to the control. The phenolic compounds in the PHE probably caused a decrease in serum lipid concentrations by disrupting the absorption and metabolism of fat in the bird's body (Sharifian et al., 2019). It has been said that phenolic compounds, through their effect on the activity of the 3-hydroxy-3-methylglutaryl coenzyme A reductase and the sterol-o-acyl transferase, reduced the absorption of cholesterol, and ultimately decreased the concentration of cholesterol and triglycerides in the blood (Esmaillzadeh et al., 2004). Aziz-Aliabadi et al. (2025) and Aziz-Aliabadi et al. (2024) reported cholesterol-lowering effects of herbal substances. However, Youssefi et al. (2023) and Lee et al. (2020) found that the serum lipid profile of chickens was unaffected by Eimeria challenge and herbal compounds.
The main purpose of using herbal compounds in poultry diets is not simply to alleviate coccidia infection. Indeed, these alternatives improve the bird performance via regulating the intestinal microbiota structure, reducing the availability of pathogen receptors, and protecting the gut barriers (Zhang et al., 2025). The beneficial microbial population of the gastrointestinal tract plays an important role in the response to gut inflammation and the maintenance of gut homeostasis, which are linked to the efficient digestion and absorption of nutrients and bird performance (Broom et al., 2018). One of the factors that disrupts the balance of the intestinal microbial population is the challenge of coccidiosis (Lien et al., 2024). In the current study, the lowest number of Coliforms was observed in treatments 0-No, 200-No, 400-No, and 300-Yes, which were significantly different from treatment 0-Yes. During the challenge, treatment 300-Yes showed the lowest number of Coliforms, which was not different from treatments 200-Yes and 400-Yes but was different from the treatment without PHE (0-Yes). It has been suggested that the use of PHE in challenged birds improved performance traits (Noruzi et al., 2024), so it can be said that using PHE would probably be more efficacious if there were infection in the poultry flock (Noruzi et al., 2024). Perhaps one of the reasons for the improved performance is the reduction in the Coliforms number. Due to its phenolic compounds and antibacterial effects, PHE provided an appropriate environment for effective digestion and absorption and improved the broilers performance by changing the gut microbiota structure (Sharifian et al., 2019). An increase in the Lactobacillus number suggests gut acidification and increases the growth of beneficial bacteria. These bacteria reduced intestinal pH by producing short-chain fatty acids (Giannenas et al., 2018). Thus, the growth of harmful bacteria (Salmonella and Coliform) will be reduced (Giannenas et al., 2018). In agreement with these results, various studies reported the positive effect of herbal compounds (oregano essential oils, mulberry leaf extract and nettle, dill, coriander and thyme extracts, respectively) on modulating the gut microbial population (Elbaz et al., 2025; Song et al., 2023; Sigolo et al., 2021). In addition, it has been shown that by penetrating the coccidia oocyst wall, tannins destroy its cytoplasm and probably deactivate the endogenous enzymes responsible for the sporulation cycle in chickens. As a result, oocyst numbers will decrease (Sharma et al., 2021; Ugwuoke and Pewan, 2020). Giannenas et al. (2003) said that the antimicrobial properties of herbal substances might decrease Eimeria oocysts and thus reduce the intestinal damage caused by this protozoan. Given that limited research has been conducted on the effects of pistachio by-products in poultry, further studies are needed to determine the optimal dosage and form of pistachio green hull (e.g., powder, essential oil, fermented, or phytosome), across different poultry species and under varying environmental conditions, such as heat and cold stress, necrotic enteritis, and other challenges.
In conclusion, the negative effects of the coccidiosis challenge were observed on some measured parameters such as carcass composition percentage. However, despite these adverse effects, the inclusion of PHE was found to reduce abdominal fat percentage, cholesterol levels, and coliform population, while improving HDL levels and meat quality. In terms of reducing abdominal fat, the authors recommend PHE at levels of 200 ppm, and in terms of improving other measured traits, levels of 300 and 400 ppm. The data related to Coliform population, emphasize that the PHE will perform efficiently in the conditions of exposure of the bird to the contamination.