This narrative review aims to provide an updated, structured analysis of the various transcatheter mitral valve interventions available today. The review focuses on procedural innovations, anatomical indications, trial evidence, and imaging strategies. Particular emphasis is placed on transcatheter edge-to-edge repair (TEER), percutaneous annuloplasty, and transcatheter mitral valve replacement (TMVR), highlighting their role in contemporary heart-team decision-making.
Mitral regurgitation (MR) is a valvular heart disease defined by the failure of the mitral valve to close properly, resulting in the backward flow of blood from the left ventricle into the left atrium during systole. [(1] It is currently recognized as the second most prevalent valvular heart disorder requiring intervention in Europe,[2,3] exceeded only by aortic stenosis, as highlighted by the VHD II survey conducted under the EURObservational Research Programme of the European Society of Cardiology. This registry aimed to assess real-world management of valvular heart diseases and evaluate adherence to clinical guidelines. The study enrolled 7,247 patients across 222 centers and found that severe native valve disease was present in 5,219 individuals (72.0%), with MR identified in 1,114 cases (21.3%), of which 746 were classified as primary and 368 as secondary.[4] Additionally, a population-based study involving 4,755 participants aged ≥ 65 years from seven general practices in Oxfordshire, UK, reported a prevalence of moderate or greater MR of 3.5% (95% CI, 3.1–3.8), with primary MR accounting for the majority of cases (124 out of 203, or 61.1%).[5]
Primary mitral regurgitation (PMR) involves structural abnormalities of one or more components of the mitral valve apparatus. [1] In high-income countries, the predominant causes are degenerative conditions,[1,3,6] such as myxomatous mitral valve disease and calcific mitral valve disease. By contrast, in low-income regions, rheumatic heart disease remains the leading cause of MR.[3,7] Infective endocarditis is another potential etiology of PMR. In secondary mitral regurgitation (SMR), the mitral valve leaflets and chordae tend to be anatomically normal; regurgitation instead arises from a disturbance in the balance between closing and tethering forces, typically due to alterations in left ventricular (LV) and left atrial (LA) geometry.[3,8] SMR is most frequently associated with dilated or ischemic cardiomyopathies, the ventricular SMR—either in the context of severe LV dilation and reduced systolic function, or following an isolated infero-basal myocardial infarction (MI) that causes posterior leaflet tethering despite near-normal LV size and ejection fraction. In some cases, SMR may result from LA enlargement and mitral annular dilatation, particularly in patients with chronic atrial fibrillation (AF), where LV function is usually preserved and ventricular dilation is minimal, becoming the particular phenotype of atrial SMR.[9]
SMR is more prevalent than PMR and is typically linked to worse outcomes,[8] mainly due to underlying cardiomyopathy.[10] Unlike PMR, the prognostic benefit of mitral valve surgery in SMR remains uncertain, with long-term high mortality despite surgical intervention. [2,11] Multiple studies have shown a strong association between SMR severity and increased all-cause mortality or heart failure (HF) hospitalizations. [10,12]
MI can give rise to ischemic MR, a complication increasingly recognized as a strong prognostic indicator during both the acute and subacute phases. In a cohort of 303 patients with Q-wave MI, echocardiographic assessment identified SMR in 64% of cases,[13] highlighting its substantial prevalence in this context. During the chronic phase, SMR remained an independent predictor of increased mortality (RR, 1.88; 95% CI, 1.23–2.86; p = 0.003), regardless of baseline clinical characteristics or the extent of LV dysfunction.[13–15] Furthermore, the magnitude of mortality risk was directly proportional to SMR severity, as quantified by effective regurgitant orifice area (EROA) and regurgitant volume (RV).[14,15]
In a cohort of 1,256 patients with dilated cardiomyopathy from the Mayo Clinic, 24% had severe SMR based on quantitative echocardiographic criteria (EROA > 0.2 cm2; RV > 30 ml; or VC > 0.4 cm).[16] These patients had higher rates of death or HF hospitalization over a median 2.5-year follow-up, independent of LVEF.[[16] The risk persisted across ischemic and nonischemic etiologies. Data from the Duke Cardiovascular Databank showed that moderate-to-severe MR (3+ to 4+ on left ventriculography) was present in nearly 30% of 2,057 patients with HF and LVEF < 40%. SMR was independently associated with increased five-year mortality (adjusted HR, 1.23; 95% CI, 1.13–1.34).[17]
Although SMR is widely recognized as a marker of poor prognosis in patients with primary LV dysfunction and HF, it remains unclear whether this relationship is causal or if reducing MR can meaningfully improve outcomes.
According to the 2025 ESC Guidelines on Valvular Heart Disease, mitral valve repair or replacement carries a class I B recommendation for all symptomatic patients with PMR who are deemed appropriate surgical candidates and do not present with prohibitive operative risk.[2,3] Furthermore, surgery is also recommended in asymptomatic patients with severe PMR and early signs of LV dysfunction—defined by LVEF ≤ 60%, LVESD ≥ 40 mm, or indexed LVESD ≥ 20 mm/m2 (Class I, Level B).[3] In carefully selected low-risk patients with preserved LV function (LVEF > 60%; LVESD < 40 mm; LVESDi < 20 mm/m2), mitral valve repair is similarly recommended (Class I, Level B)[3] when a durable result is highly likely, particularly when at least three adverse prognostic markers are present: AF, resting systolic pulmonary artery pressure > 50 mmHg, significant LA enlargement (LAVI ≥ 60 mL/m2 or LA diameter ≥ 55 mm), or concomitant ≥ moderate secondary tricuspid regurgitation.
For operable patients with severe PMR, surgical mitral valve repair remains the preferred intervention, as it reliably restores valvular anatomy and has been associated with low operative mortality in contemporary cohorts (~1%).[18] Compared with replacement, repair confers lower perioperative mortality and superior long-term survival, functional status, and ventricular remodeling.[19,20] Repair of more complex lesions—such as leaflet or annular calcification or rheumatic pathology—requires considerable expertise and should be performed in experienced heart valve centers.[3,21–23] When anatomical suitability is lacking, mitral valve replacement with preservation of the subvalvular apparatus is recommended.[24]
Minimally invasive mitral surgery via right minithoracotomy is increasingly used in experienced centers, offering equivalent safety and efficacy to conventional sternotomy with shorter hospitalization and faster early recovery, although large registry data show more heterogeneous outcomes.[25]
M-TEER serves as an important therapeutic alternative for selected patients with chronic PMR who are elderly, frail, or present high surgical risk. Although this group constitutes a minority of PMR cases, TEER can provide meaningful symptomatic relief and effective MR reduction when surgery is not feasible.[3,26] Procedural and midterm outcomes are closely linked to residual MR severity and transmitral gradient—parameters that strongly influence clinical prognosis.[26] Technological improvements and cumulative operator experience have expanded the anatomical spectrum amenable to TEER, enabling successful treatment in previously challenging leaflet pathologies.[27,28]
The 2025 ESC Guidelines emphasize that the decision between surgery, TEER, TMVI, or conservative management must be tailored by the multidisciplinary heart team,[3] incorporating clinical characteristics, anatomical suitability, comorbidities, procedural risk, and patient preferences.[3] In operable patients with repairable anatomy, surgery remains the preferred therapy; TEER is best reserved for those with unacceptable surgical risk or anatomical conditions conducive to predictable procedural success but suboptimal surgical durability.
The long-term comparative efficacy of TEER versus surgical repair in PMR remains under investigation. Several ongoing randomized trials—including studies enrolling high-risk (NCT03271762), intermediate-risk (NCT04198870), and all-risk patients > 60 years (NCT05051033)[3]—aim to clarify durability, clinical benefit, and survival following TEER in degenerative MR.[29] Conversely, TMVR has demonstrated excellent MR abolition in highly selected patients, especially those with anatomy unsuitable for TEER. However, TMVI is limited by restricted device availability, high screening failure rates, and meaningful risks such as LVOT obstruction and valve thrombosis, with only modest midterm durability data currently available.[3,30,31]
Chronic SMR is associated with adverse prognosis and presents significant therapeutic challenges. Its management requires meticulous optimization of guideline-directed medical therapy, complemented by individualized consideration of electrophysiological, transcatheter, or surgical interventions based on patient-specific characteristics.[32]
For ventricular SMR, indications for isolated mitral valve surgery are highly restrictive due to substantial procedural risk and the lack of proven mortality benefit.[33] Current guidelines recommend surgical intervention primarily when severe SMR coexists with coronary artery disease requiring coronary artery bypass grafting or when another cardiac procedure is already planned.[3] Although undersized rigid annuloplasty rings can restore leaflet coaptation in selected patients without advanced ventricular remodeling, recurrence remains a major limitation.[3] In randomized trials, restrictive annuloplasty was associated with high rates of recurrent MR—particularly in patients with significant leaflet tethering or large tenting areas (> 1.35 cm2/m2)—and demonstrated limited reverse remodeling.[34,35] As a result, chordal-sparing mitral valve replacement is often favored when ventricular geometry predicts poor durability of repair.[36]
The management of moderate ischemic SMR in patients undergoing CABG remains under debate. Meta-analyses of randomized trials have shown that adding mitral valve surgery reduces recurrent MR yet confers no benefit in mortality or clinical outcomes.[37–39] Therefore, decisions should balance the increased perioperative complexity against the long-term risk of MR progression.
In contrast, atrial SMR represents a distinct pathophysiological entity, typically affecting elderly patients with long-standing AF and LA dilatation. Observational data suggest that surgical annuloplasty can be effective and durable in this subgroup, mitigating the atrial-driven mechanism of MR progression and potentially improving HF hospitalizations and mortality when compared with medical therapy alone.[40–42] Outcomes may be further enhanced when annuloplasty is combined with surgical AF ablation or LA appendage occlusion, and when concomitant tricuspid regurgitation is addressed during the same operation.[42,43]
According to the 2025 ESC Guidelines, M-TEER is recommended (Class I, Level A) to reduce HF hospitalizations and improve quality of life in hemodynamically stable, symptomatic patients with severe ventricular SMR, impaired LVEF < 50%, and persistent MR despite optimized guideline-directed medical therapy and cardiac resynchronization therapy when indicated.[3,44–47] Eligibility requires fulfillment of specific clinical and echocardiographic criteria that predict improved outcomes, including favorable anatomy for TEER, NYHA class ≥ II, LVEF 20%–50%, LVESD ≤ 70 mm, at least one HF hospitalization in the past year or elevated natriuretic peptides, SPAP ≤ 70 mmHg, absence of severe RV dysfunction, absence of stage D HF, no untreated CAD requiring revascularization, and no severe aortic or tricuspid valve disease.[3,47–50] Collectively, these parameters reflect a phenotype in which TEER is most likely to yield durable hemodynamic and clinical benefit.
Emerging data also support the use of TEER in selected symptomatic patients with ventricular SMR who do not fully meet the guideline-defined criteria for outcome improvement. Observational studies have shown that TEER can enhance functional capacity, facilitate up-titration of guideline-directed medical therapy (GDMT) in patients who previously could not tolerate it, and provide meaningful symptomatic relief in individuals with persistent severe SMR, including those with recent MI.[51,52] For such patients, TEER may be considered (Class IIb, Level B)[(3] after comprehensive heart team evaluation and clarification of candidacy for advanced therapies such as LV assist device implantation or heart transplantation.[48,52] This nuanced approach acknowledges the heterogeneity of ventricular SMR and the potential for TEER to offer clinical value beyond strictly defined prognostic subsets.
Further support for TEER in ventricular SMR comes from the MATTERHORN randomized controlled trial, which enrolled a predominantly ventricular SMR population (84%).[53] The trial demonstrated that TEER was noninferior to surgical mitral repair or replacement for a composite endpoint of mortality, HF hospitalization, MV reintervention, stroke, or need for mechanical circulatory support at 1 year, while exhibiting a superior safety profile. [53] These results reinforce the role of TEER as a viable alternative in patients with high surgical risk or in whom durable surgical repair is uncertain, underscoring its integration into contemporary guideline recommendations.
TEER may also be considered in atrial SMR, with observational studies demonstrating high procedural success and safety,[54–57] although anatomical factors such as planar coaptation, large regurgitant orifices, and small native mitral valve area (MVA) may increase transmitral gradients and require careful evaluation.[58,59] Further studies are needed to refine treatment strategies in this growing patient population.[60]
Overall, TEER has become a central interventional strategy in the management of ventricular SMR and an emerging option for selected patients with atrial SMR, effectively bridging a major therapeutic gap for individuals who remain symptomatic despite maximally tolerated GDMT, CRT, or rhythm–control strategies, and, when appropriate, revascularization. Its expanding evidence base, favorable safety profile, and proven ability to reduce heart-failure burden position TEER as a cornerstone therapy in contemporary practice. Ongoing randomized trials and real-world registries will further refine anatomical selection, procedural durability, and the long-term prognostic impact of TEER across the spectrum of SMR mechanisms.
The MitraClip device received FDA approval in 2013 for the transcatheter treatment of patients with symptomatic severe PMR due to structural abnormalities of the mitral valve.[61] This therapeutic approach was specifically targeted toward individuals considered inoperable or at prohibitive surgical risk, as determined by a multidisciplinary heart team. Compared to patients eligible for surgical correction, candidates for MitraClip often present with more advanced age and multiple comorbidities, which, while precluding surgery, do not exclude them from benefiting from percutaneous repair.
The EVEREST II trial, a landmark multicenter randomized controlled study, enrolled 279 patients with severe degenerative MR, randomized in a 2:1 ratio to undergo either transcatheter repair with MitraClip or conventional surgical intervention.[62] While MitraClip offered a favorable safety profile—with significantly fewer major adverse events within 30 days (15% versus 48%, p < 0.001) and meaningful improvements in functional status, quality of life, and ventricular dimensions—it was less effective in completely eliminating MR at the 12-month mark when compared to surgery.[63,64] Long-term data from this trial, reported by Mauri et al., demonstrated no significant difference in mortality at 4 years (17.4% versus 17.8%, p = 0.914).[65]
The EVEREST II trial focused on a narrowly defined patient group—primarily male individuals, averaging 67 years in age, with preserved LV systolic function (mean LVEF around 60%) and limited comorbidity burden apart from HF.[62] These selection criteria, although suited for a controlled clinical environment, substantially restricted the generalizability of the findings to real-world populations encountered in routine practice. To bridge this gap, Sorajja and collaborators leveraged data from the STS/ACC Transcatheter Valve Therapy Registry in a comprehensive postmarketing evaluation. [66] Their analysis encompassed 2,952 patients who underwent commercial MitraClip implantation at 145 U.S. centers between November 2013 and September 2015. This cohort differed markedly from the EVEREST population, characterized by older age (median 82 years), increased clinical frailty, and elevated procedural risk as reflected by median STS scores of 6.1% for surgical mitral repair and 9.2% for valve replacement.[66] Despite the advanced risk profile, procedural success was achieved in 91.8% of cases, and the in-hospital mortality rate remained low at 2.7%.[66] Following the procedure, a marked improvement in MR severity was observed, with 93% of patients achieving a reduction to grade 2+ or less, and 63.7% reaching grade 1+ MR.[64,66] This clinical benefit is particularly relevant, given the well-established association between more-than-moderate MR and adverse outcomes. The predominant anatomical lesion involved the A2-P2 scallops (78%), and nearly one-third of patients (31%) required the implantation of multiple clips—a notable distinction from the EVEREST II cohort.[64,66]
Additional long-term evidence has been provided by the German TRAMI registry, a large, multicenter, prospective, and industryindependent cohort encompassing 722 patients who underwent MitraClip implantation across 20 centers.[67] With a median follow-up duration of approximately 2.8 years (1,037 days), the registry offers valuable insights into the durability of clinical benefits and long-term survival associated with TEER. Notably, improvements in functional status were sustained over time, with 65% of patients remaining in NYHA class I or II at final follow-up, mirroring the 63.3% reported at 1 year.[67] Patient-perceived quality of life, as assessed by the EQ-VAS score, also remained stable (median 60 [IQR 50–70]). However, cumulative mortality rates increased steadily, reaching 19.7% at 1 year, 31.9% at 2 years, and 53.1% at 4 years, irrespective of MR etiology.[67] Multivariable analysis identified several independent predictors of mortality, including prior aortic valve replacement (HR, 2.21; p < 0.0001), NYHA class IV at baseline (HR, 1.78; p < 0.001), previous hospitalization for cardiac decompensation (HR, 1.63; p < 0.001), impaired renal function (creatinine >1.5 mg/dL; HR, 1.63; p < 0.0001), and severely reduced LVEF (< 30%; HR, 1.60; p < 0.001).[67] These findings highlight the substantial comorbidity burden among TEER candidates and suggest that although MitraClip therapy offers sustained symptomatic and quality-of-life benefits, it may not fully counterbalance the negative prognostic impact of advanced cardiac and noncardiac pathology on long-term survival.
TEER using the MitraClip system has emerged as a minimally invasive therapeutic option for patients with SMR. The safety and efficacy of this device have been evaluated initially in two pivotal randomized controlled trials—MITRA-FR and COAPT—which yielded markedly divergent outcomes in HF populations. Despite targeting seemingly similar patient cohorts, defined by symptomatic systolic HF and significant SMR, the trials produced contrasting results: MITRA-FR reported neutral findings,[68] whereas COAPT demonstrated substantial clinical benefit (Figure 1).[69]
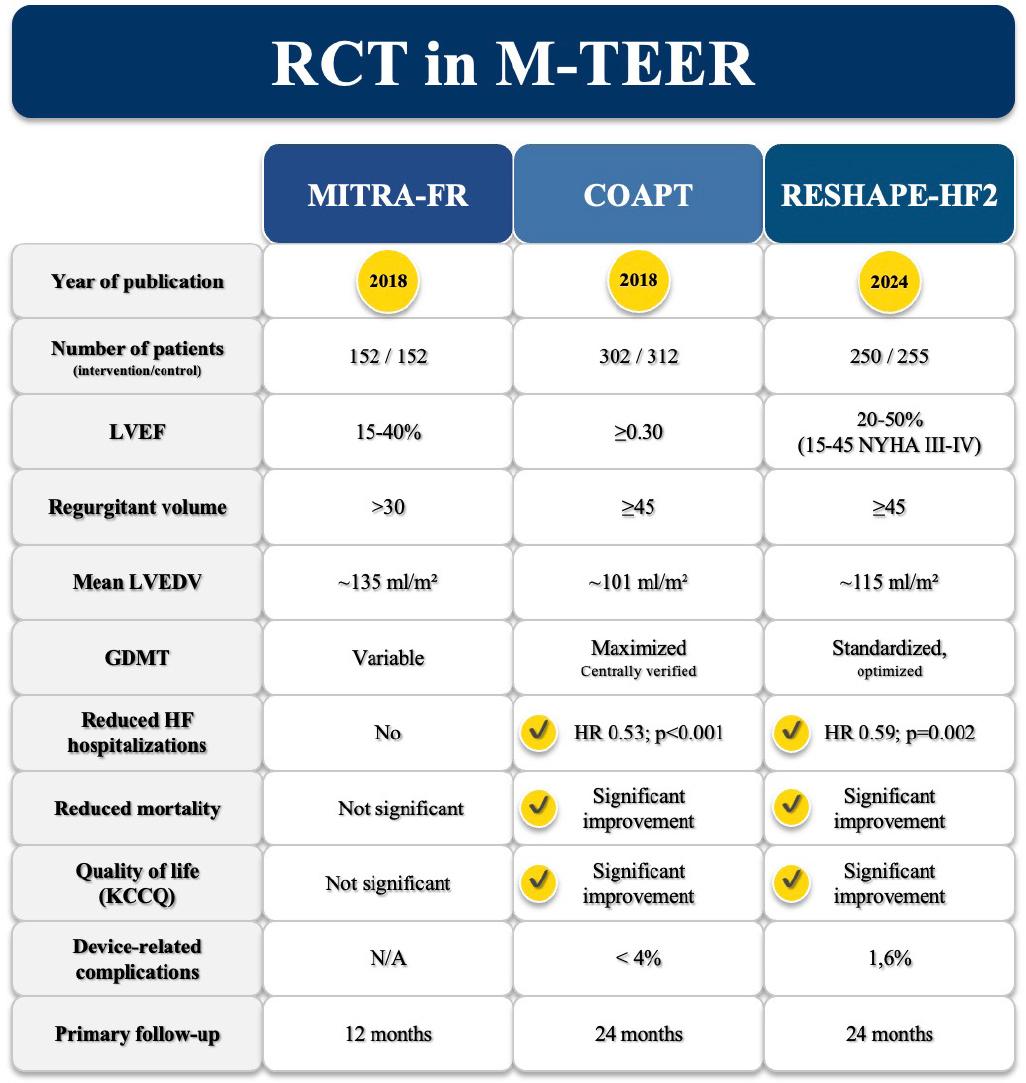
Randomized controlled trials of M-TEER highlighting differences in inclusion criteria and key outcomes for the composite end point of HF hospitalization, mortality, and quality of life.
In the MITRA-FR trial, 304 patients with symptomatic systolic HF and severe secondary MR—defined by an EROA > 20 mm2 and/or a RV > 30 mL—and LVEF between 15% and 40% were randomized in a 1:1 ratio to receive MitraClip on top of optimized medical therapy or medical therapy alone.[68] The primary end point, a composite of all-cause death or unplanned HF hospitalization at 12 months, showed no significant difference between groups (54.6% versus 51.3%, p = 0.53).[68] Likewise, individual components such as mortality (24.3% versus 22.4%) and hospitalization rates (48.7% versus 47.4%) did not differ meaningfully.[68] In contrast, the COAPT trial enrolled 614 patients with symptomatic HF and at least moderate-to-severe SMR (EROA > 30 mm2 and/or RV > 45 mL), with LVEF ≥20%.[69] Patients were randomized in a similar fashion, but the outcomes were distinctly favorable in the intervention group.[68–70] The annualized rate of HF hospitalizations over a 2-year period was significantly lower in the MitraClip arm compared to control (35.8% versus 67.9% per patientyear, p < 0.001), and all-cause mortality was also reduced (29.1% versus 46.1%; p < 0.001) (Figure 1).[69,70]
These conflicting results between MITRA-FR and COAPT can likely be attributed to key differences in patient selection criteria and therapeutic strategy.[70] Patients enrolled in MITRA-FR showed more advanced LV disease, as reflected by larger LV end-diastolic volumes (135 ± 35 mL/m2 in MITRA-FR versus 101 ± 34 mL/m2 in COAPT) and more severe LV systolic dysfunction (LVEF 15%–40% in MITRA-FR versus 20%–50% in COAPT and excluding patients with LV end-systolic diameter ≥ 70 mm) (Figure 1).[68–70] Additionally, MR severity was greater in COAPT (mean EROA 41 ± 15 mm2) than in MITRA-FR (31 ± 10 mm2), despite similar terminology in inclusion criteria (Figure 1).[68–70] This discrepancy is likely due to differences in how MR severity was quantified—MITRA-FR followed European guideline thresholds (EROA < 20 mm2), while COAPT used higher American guideline cutoffs (EROA < 30 mm2).[23] This could have significantly influenced which patients qualified for inclusion, possibly resulting in a less severe MR population in MITRA-FR.
Beyond baseline characteristics, notable differences in therapeutic management may have contributed to the divergent findings. In COAPT, patient selection was more rigorous: individuals had to be on maximally tolerated doses of GDMT, and, when appropriate, CRT, ICD therapy, or coronary revascularization had to be optimized prior to randomization.[69,70] In contrast, the MITRA-FR protocol allowed for ongoing adjustments of medical therapy during follow-up, with some patients not being fully optimized at baseline (Figure 1).[68,70] This variability could have diluted the potential benefits of MitraClip. Moreover, a more aggressive interventional approach was pursued in COAPT, as evidenced by a greater number of clips implanted per patient and a more consistent, sustained reduction in MR severity during follow-up.[69] This difference in procedural intensity may have further limited the ability of MITRA-FR to detect a treatment benefit.
In addition to prior studies, the RESHAPE-HF2 trial was a multicenter, randomized controlled study conducted across 30 centers in nine countries, enrolling 505 patients with HF with reduced ejection fraction (LVEF 20%–50%) and moderate-to-severe SMR. [71] Participants were randomized to either undergo TEER (n = 250) or receive GDMT (n = 255) alone.[71] After 24 months, the TEER group exhibited a significantly lower incidence of the composite end point—comprising HF hospitalization or cardiovascular death—at 37.0 events per 100 patient-years compared to 58.9 in the GDMT cohort (RR 0.64; 95% CI, 0.48–0.85; p = 0.002).[71] When analyzing HF hospitalizations exclusively, TEER maintained a significant advantage (26.9 versus 46.6 events per 100 patient-years; RR 0.59; 95% CI, 0.42–0.82; p = 0.002).[71] Moreover, clinical status, assessed by the Kansas City Cardiomyopathy Questionnaire Overall Summary score, improved more substantially in the TEER arm, with a mean increase of 21.6 points compared to 8.0 points in the medical therapy group at 12 months (p < 0.001) (Figure 1).[71]
Interestingly, TEER in RESHAPE-HF2 did not result in a statistically significant reduction in all-cause mortality.[71] This finding may be attributable to differences in patient characteristics relative to COAPT, including a relatively low baseline use of contemporary HF treatments such as sodium-glucose cotransporter-2 inhibitors, prescribed in fewer than 10% of participants.[70,72] Additionally, the trial’s extended enrollment period involved the use of multiple generations of the MitraClip device, though the impact of device iteration on clinical outcomes has not yet been clearly defined.[71,72]
Given the contrasting findings reported across the three major randomized controlled trials, a contemporary meta-analysis conducted by Anker et al.[72] sought to quantify the overall therapeutic benefit of TEER in patients with SMR. Despite notable variability in trial populations—particularly regarding the severity of SMR, LV dimensions, and inclusion criteria—the meta-analysis demonstrated a significant and consistent reduction in recurrent HF hospitalizations over a 24-month period among patients treated with TEER (HR, 0.63; 95% CI, 0.50–0.80; p = 0.0002).[72] Furthermore, TEER was associated with a lower incidence of the composite end point of HF hospitalization or all-cause mortality (HR 0.72; 95% CI 0.51–0.999; p = 0.0493).[72] Although numerical reductions in all-cause and cardiovascular mortality were observed (HR 0.76 and 0.77, respectively),[72] these trends did not achieve statistical significance in either the primary model or in the more conservative Hartung–Knapp sensitivity analysis.[72]
These findings reinforce the clinical relevance of TEER therapy in attenuating HF-related morbidity among appropriately selected patients with SMR. The variability in outcomes across trials underscores the spectrum of SMR and the need for nuanced patient selection, influenced by underlying ventricular pathology and comorbidity profiles. Future patient-level meta-analyses may provide more granular insight, potentially identifying those who derive the greatest benefit from timely mitral intervention.
A comprehensive imaging workup is central to the diagnostic, therapeutic, and procedural planning pathway in patients considered for TEER. Transthoracic echocardiography (TTE) remains the cornerstone for the initial assessment, providing essential information on MR severity, mechanism, and hemodynamic consequences, including estimates of pulmonary artery pressures, biventricular function, and concomitant valvular lesions. However, due to its superior spatial resolution and ability to visualize leaflet anatomy and motion in detail, transesophageal echocardiography (TOE)— preferably with three-dimensional (3D) capabilities—is mandatory in almost all TEER candidates to determine anatomical suitability (Figure 2).
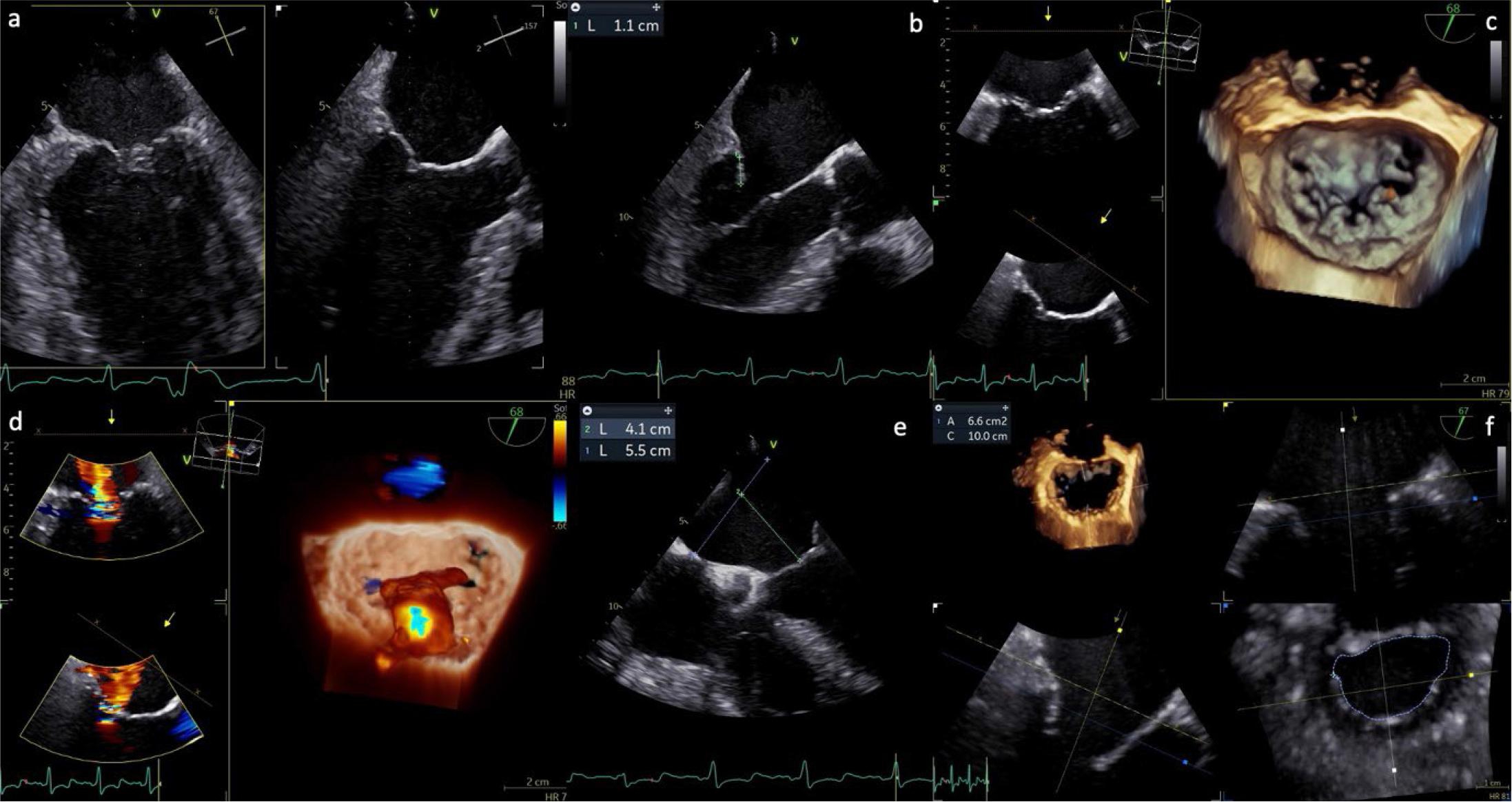
Preprocedural TOE assessment in a case of SMR. (a) X-plane bicommissural and LV long-axis view (67°/157°); (b) measurement of the posterior leaflet, showing adequate length for device grasping; (c) 3D reconstruction of the mitral valve, en face (surgical) view; (d) 3D reconstruction demonstrating severe SMR due to leaflet restriction, with a centrally originating jet directed posteriorly; (e) evaluation of septal puncture feasibility; (f) multiplanar reconstruction (MPR) for calculation of the anatomical MVA.
In the context of SMR, accurate assessment of regurgitation severity presents notable challenges. A comprehensive evaluation (Figure 2) using a multiparametric and integrative approach is essential, incorporating qualitative, semiquantitative, and quantitative echocardiographic indicators.[2] Quantitative thresholds traditionally associated with severe MR include a vena contracta (VC) width ≥ 7 mm, regurgitant fraction ≥ 50%, EROA ≥ 40 mm2, and RV ≥ 60 mL.[2] However, in SMR, reliance on EROA and RV—typically derived using the proximal isovelocity surface areamethod—can be misleading, as the assumptions of a circular regurgitant orifice and stable flow may not hold true. The regurgitant orifice is often elliptical, and low-flow states common in advanced LV dysfunction can further distort these measurements. As a result, lower thresholds (e.g., EROA ≥ 30 mm2 and RV ≥ 45 mL) are frequently proposed to define severe MR in SMR populations.[2i Nonetheless, such cutoffs must be interpreted in clinical context, since modeling studies suggest they may correspond to unrealistically low cardiac outputs. [64,73] Therefore, the integration of advanced imaging parameters, such as 3D VC measurements, alongside indirect markers—such as systolic flow reversal in pulmonary veins, LA dilatation, LV systolic function and diameter, evidence of pulmonary hypertension, and signs of right ventricular dysfunction—remains critical for accurate grading and therapeutic decision-making in SMR.
TEE enables a meticulous evaluation of mitral valve morphology, including leaflet thickness, mobility, prolapse or tethering, clefts, and calcification—particularly within the intended grasping zone of the clip arms.[64,73]
Precise anatomical assessment (Figures 2,3) is fundamental in the preprocedural evaluation of patients with MR considered for transcatheter repair. The flail gap—the vertical distance from the ventricular aspect of the flail leaflet segment to the atrial edge of the opposing leaflet—is measured during systole, perpendicular to the annular plane, using TOE views in the four-chamber long axis and LV outflow tract (LVOT). The flail width, defined as the extent of the flail segment along the coaptation line, is typically assessed in transgastric short-axis or commissural views. Identifying calcification within the grasping zone (approximately the distal 7 mm of both anterior and posterior scallops) is essential to distinguish it from leaflet thickening or fibrosis, which may affect device anchoring. Additional measurements such as coaptation length (the leaflet overlap) and coaptation depth (the apical displacement of leaflet meeting point relative to the mitral annulus) further inform anatomical suitability— especially in secondary MR. Key quantitative thresholds, including a flail gap <10 mm, flail width <15 mm, and coaptation depth <11 mm, are instrumental for procedural planning in degenerative MR, while also aiding risk stratification in functional cases (Figure 3).[(64] Moreover, three-dimensional multiplanar reconstruction (3D MPR) plays a critical role in delineating jet origin and quantifying the VC area, which are predictors of procedural success and long-term durability.
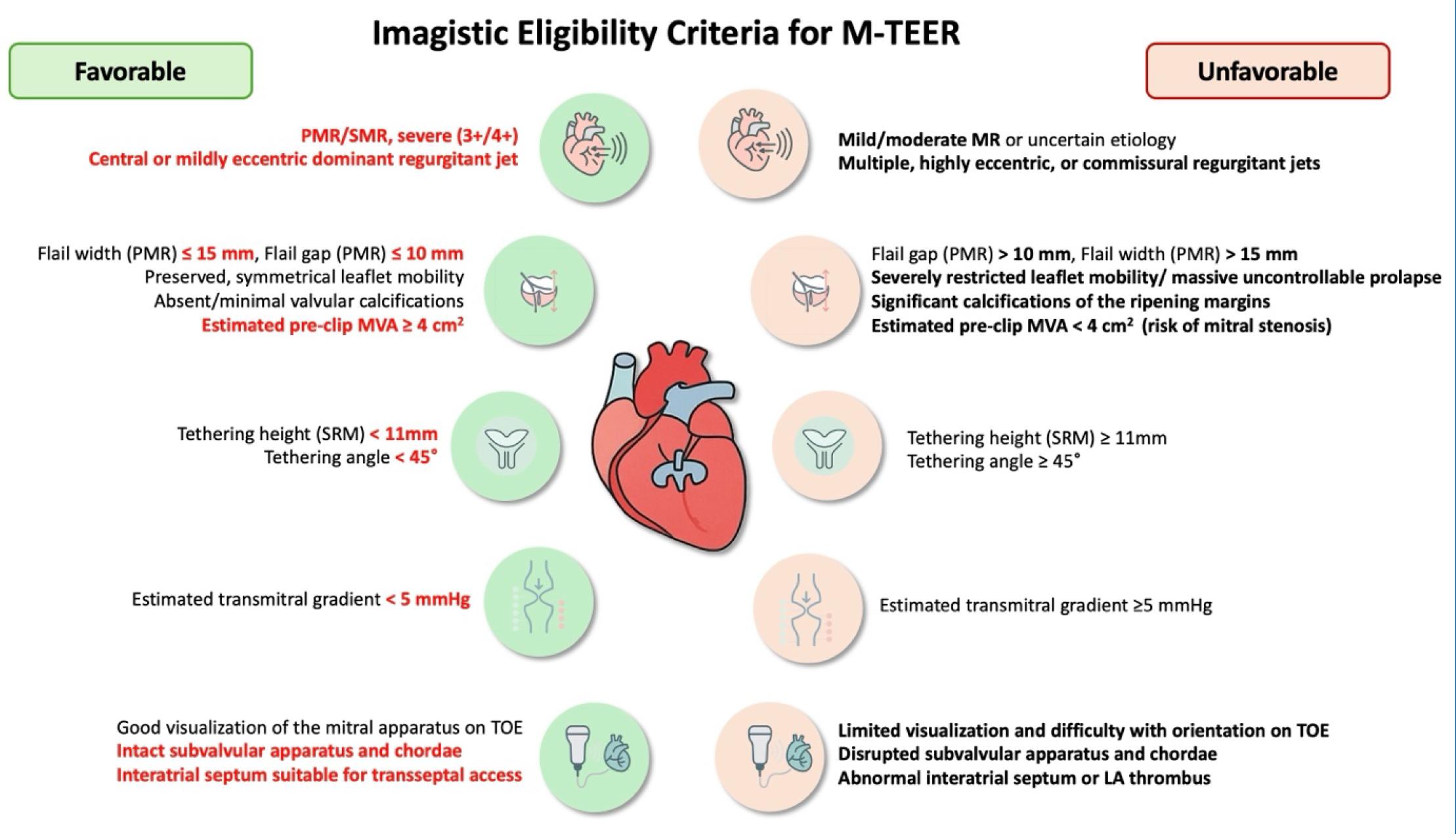
Imagistic Criteria for Selection of Patients Eligible for M-TEER.
In selected cases, exercise or pharmacologic stress echocardiography may be helpful to unmask dynamic MR worsening and pulmonary hypertension, particularly when symptoms seem disproportionate to resting MR findings. Additionally, coronary angiography is routinely performed to exclude significant coronary artery disease, while coronary computed tomography (CT) angiography may be an alternative in low-risk patients. Right heart catheterization may be considered for patients with pulmonary hypertension, right ventricular dysfunction, or significant tricuspid regurgitation to further refine risk and treatment strategy.
Ultimately, all imaging findings are discussed by a multidisciplinary Heart Team, which includes an interventional cardiologist, a cardiac surgeon, an imaging specialist and an HF expert. The team integrates imaging, clinical, and procedural data to guide decision-making, ensuring that TEER is pursued in anatomically feasible, clinically appropriate candidates. While initial procedural selection was based on strict EVEREST II criteria, real-world experience has broadened inclusion, although data suggest that deviations from these anatomical thresholds may be associated with higher rates of persistent MR or reintervention.[64] Hence, careful anatomical and functional evaluation remains a pivotal step in optimizing outcomes for patients undergoing TEER.
Both PASCAL and MitraClip procedures rely on high-quality intraprocedural imaging—chiefly TOE with 3D MPR and single-plane fluoroscopy—to guide transseptal access, device steering, leaflet grasping, and clip deployment. The use of biplane TOE during transseptal puncture provides excellent spatial orientation and has largely eliminated the routine need for biplane fluoroscopic imaging, which is now reserved for select anatomically challenging cases. Precise coordination between the imaging specialist and the interventional cardiologist is critical to ensure spatial alignment and effective device delivery.
Optimal trans-septal puncture is performed under TOE guidance, typically in a posterior-superior position, at least 4 cm above the mitral annulus to ensure a favorable trajectory for clip delivery. The procedure begins with a bicaval view (90°–120°) using an X-plane to localize and adjust the needle tip in the antero-posterior plane (Figure 4). Fusion technology allows these 2D views to be overlaid on fluoroscopy, and in recent advancements, full 3D heart segmentation can identify the optimal puncture zone. After securing LA access, the guide catheter is advanced into the left atrium. Following successful septum crossing, the position of the curved stiff wire in the left pulmonary veins (typically the left upper) or left atrium is confirmed. Fusion imaging aids in guiding this step and steering the catheter toward the mitral plane (Figure 4), avoiding interaction with the left pulmonary veins and LA appendage.[73]
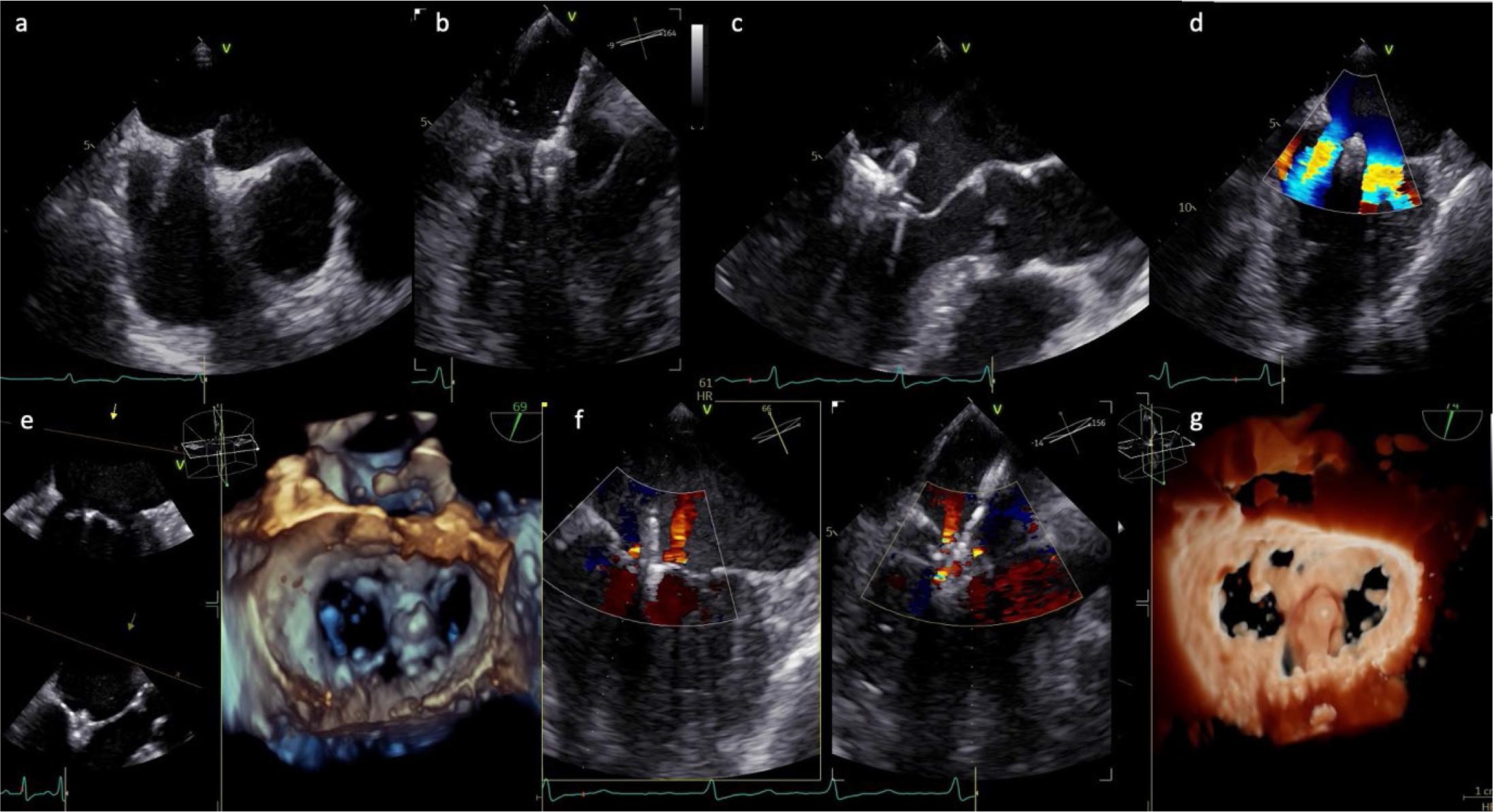
Intraprocedural TOE guidance during TEER for SMR. (a) Transseptal puncture in the aortic valve short-axis view (45°); (b) advancement and steering of the catheter towards the mitral plane; (c) catheter orientation perpendicular to the mitral plane, followed by valve crossing and leaflet grasping; (d), (f) device positioned above the mitral annulus, perpendicular to the leaflet coaptation line, typically at the origin of the regurgitant jet, which is reduced after device deployment; (e), (g) 3D reconstruction of the mitral valve, en face (surgical) view, after device deployment.
The grasping view is, by definition, a targeted long-axis image at the intended landing zone that simultaneously displays the anterior and posterior leaflets for optimal capture. The commissural view is used primarily for jet localization and medial–lateral orientation, whereas precise leaflet grasping is performed in a dedicated long-axis plane.
Precise clip placement during mitral M-TEER is critical, starting with positioning the device above the mitral annulus, perpendicular to the leaflet coaptation line, typically at the regurgitant jet’s origin (Figure 4). Device orientation is guided using a 3D TOE view from the left atrium in the standardized “surgeon’s view,” with the aorta positioned at 12 o’clock. This orientation allows intuitive alignment of device movements with the intercommissural plane during steering and implantation.[73] Effective collaboration between the cardiac imaging specialist and structural interventionalist is vital, requiring a shared understanding of 3D mitral anatomy and integration of fluoroscopic and echocardiographic imaging for real-time guidance. After confirming perpendicular orientation via 3D echocardiography, adjusting the X-ray gantry angle ensures consistent device alignment during fluoroscopy-guided grasping.
For valve crossing and leaflet grasping, standardized views are employed, including an intercommissural view (50°–70°) with an X-plane to obtain a LVLVOT view (140°–160°) of the A2/P2 segment, displaying both leaflets clearly Figure 4).[73] Simultaneous visualization of these planes allows trajectory corrections, ensuring the catheter remains in the left ventricle to facilitate grasping and prevent valve distortion. Catheter adjustments—pulling/pushing, flexing/unflexing, or rotating clockwise/counterclockwise—shift the device medially/laterally or toward the posterior/anterior, respectively, as seen in the intercommissural and LVOT views. Flexing the guide sheath corrects an “aorta-hugger” position or adjusts height, while posterior rotation increases height. For commissural pathologies, the intercommissural plane is adjusted to 30°–60° for A3/P3 or 60°–90° for A1/P1 to align with the coaptation line, with optimal views derived from 3D MPRs.[73]
Once aligned, the clip arms are opened perpendicular to the coaptation plane. Depending on the operator’s preference, the device may advance into the left ventricle with arms open or closed, with open-arm advancement requiring intraprocedural orientation adjustments (Figure 4). Before grasping, both mitral leaflets must be confirmed as freely mobile above the open clip arms, precisely positioned along the coaptation line.[73] Grippers are then deployed to secure the leaflets between the clip arms and grippers, with partial clip closure confirming initial leaflet capture (Figure 4).
Following leaflet grasping, residual MR must be carefully evaluated, and transmitral gradients should be measured to exclude significant iatrogenic mitral stenosis. Insights from prior device generations provide useful benchmarks regarding the expected reduction in the MVA postimplantation. For instance, the PASCAL P10 system has been reported to reduce MVA by approximately 47%. Importantly, the extent of this reduction is strongly influenced by the device’s position along the coaptation line, with the greatest decrease occurring when the device is deployed centrally at the A2/P2 scallops—often referred to as the “hot zone”—and a more modest reduction is observed with commissural placement. Beyond the implantation site, the location of the regurgitant jet and the chosen therapeutic approach are critical determinants in selecting the appropriate device and strategy. In cases involving multiple jets, where two devices may need to be placed at a considerable distance from one another, a baseline MVA of at least 6 cm2 is generally advised to avoid inducing clinically significant mitral stenosis (Figure 4).
Mitral annuloplasty is a cornerstone of surgical mitral valve repair, utilized in virtually all procedures addressing primary and secondary MR. In recent years, significant efforts have focused on developing transcatheter methods to emulate the precision of surgical annuloplasty, offering new therapeutic avenues for patients deemed too high-risk for open surgery. Percutaneous mitral annuloplasty is divided into two main strategies: direct and indirect approaches.[74]
Direct annuloplasty entails implanting a ring or band securely fastened to the mitral annulus, closely replicating the surgical technique. This method demands high technical precision and relies on sophisticated imaging to accurately position anchors on the dynamically shifting mitral annulus.[64] Conversely, indirect annuloplasty reshapes the mitral annulus by modifying the geometry of nearby structures, such as the coronary sinus. While simpler to perform, this approach is constrained by anatomical challenges, as the coronary sinus and mitral annulus often lie in different planes, potentially limiting the effectiveness of annular remodeling.[64,74]
Direct annuloplasty is guided predominantly by real-time echocardiography—most commonly 3D TOE—which allows accurate visualization of annular anatomy and precise positioning of anchors. Fluoroscopy plays a complementary role, primarily to ensure spatial safety and to avoid injury to the circumflex coronary artery during anchor deployment. In contrast, indirect annuloplasty integrates both fluoroscopic and echocardiographic guidance, reflecting its reliance on coronary sinus anatomy.[74]
Transcatheter direct annuloplasty devices represent a transformative approach to managing SMR, offering alternatives for patients at high surgical risk. The Cardioband Mitral Valve Reconstruction System (Edwards Lifesciences, Irvine, CA), the most widely adopted device in this category, is delivered via a transfemoral, transseptal route using a 25 French steerable sheath.[64] It features a flexible polyester band with 12 to 17 helical anchors, spaced 8 mm apart, affixed along the posterior mitral annulus from the anterolateral to posteromedial commissure.[74] Preprocedural CT guides device sizing based on the mitral valve perimeter. Anchors are sequentially deployed under fluoroscopic and TOE guidance, starting at the lateral commissure and moving posteriorly. A premounted contraction wire, adjusted via a spool, cinches the band to reduce the annular septolateral dimension, effectively alleviating MR.[64,74] In a prospective multicenter study of 60 high-risk patients with moderate to severe SMR (mean LVEF 33% ± 11%), Messika–Zeitoun et al. reported one-year outcomes for 39 patients, demonstrating 87% survival, 66% freedom from HF rehospitalization, and 78% freedom from reintervention. [75] Notably, 95% of patients achieved MR reduction to moderate or less, with significant improvements in NYHA functional class and quality-of-life metrics.[75] Early technical challenges, such as anchor disengagement in the initial 30 patients, were addressed through device modifications, though rare complications included two nondevice-related in-hospital deaths, one stroke, two coronary artery issues, and one tamponade.[75] The ACTIVE trial (NCT03016975),[76] currently active but not recruiting, randomizes patients with significant SMR to Cardioband plus GDMT versus GDMT alone, evaluating MR reduction, cardiovascular mortality, HF hospitalizations, and functional outcomes over 5 years.
The Millipede IRIS device (Boston Scientific, Maple Grove, MN), under active clinical investigation, is a nitinol-based, semirigid complete annuloplasty ring with eight pre-attached stainless-steel helical anchors. Delivered transfemorally via a transseptal sheath, it is maneuvered above the mitral annulus using TOE and fluoroscopy, then expanded to engage the annulus. A distinguishing feature is its integrated intracardiac echocardiography (ICE) catheter, which offers unobstructed near-field visualization of the annulus and anchor deployment. However, ICE complements rather than replaces TOE, as its close proximity to the target limits the assessment of device trajectory and its relationship with global anatomical structures. In contemporary practice, both modalities are often used in combination to optimize procedural guidance.[64,74] Anchors can be independently rotated, retracted, and repositioned, ensuring precise deployment. Postanchoring, 8 sliding collars allow individualized tensioning to tailor annular reduction.[64,74] In a first-in-human study of 7 patients with moderate-to-severe (3–4+) ischemic or nonischemic MR (mean LVEF 42%±19%), Rogers et al.[77] reported a 31% reduction in septolateral diameter (from 38.0 ± 4.1 mm to 25.9 ± 4.9 mm) and MR reduction to 0–1+ at 30 days, accompanied by improvements in NYHA class, LV, and LA volumes.[77)] The device’s fully repositionable and retrievable design enhances procedural adaptability, with ongoing trials further assessing its performance.
The AMEND system (Valcare, Israel) introduces a rigid, D-shaped annuloplasty ring, currently delivered transapically, with a transfemoral transseptal approach in development. Delivered linearly through a catheter, it reforms into a rigid ring within the left atrium. Barbed anchors first secure the posterior annulus, followed by anterior fixation and detachment from the delivery system.[64,74] Early human implantations via transapical delivery have shown promise, though peer-reviewed data remain scarce. The system’s potential compatibility with a docking transcatheter ViR strategy suggests future versatility, pending further validation.
These devices underscore the dynamic evolution of transcatheter mitral annuloplasty. The Cardioband’s robust clinical track record, the Millipede’s precision through ICE-guided delivery, and the AMEND’s innovative rigid-ring design collectively highlight their potential to expand therapeutic options for SMR. Continued research is essential to refine procedural techniques, address technical challenges, and establish long-term efficacy in diverse patient populations.
The Carillon Mitral Contour System stands as the leading device for indirect transcatheter mitral annuloplasty, tailored for managing SMR in patients deemed ineligible for surgical intervention. It features 2 self-expanding anchors deployed within the proximal coronary sinus and great cardiac vein, interconnected by a nitinol cable of fixed length.[64] Introduced via a jugular vein approach and guided by fluoroscopy, the system modifies the mitral annulus geometry by exploiting the coronary sinus’s anatomical adjacency, thereby reducing MR severity.[(64] Its safety and efficacy have been rigorously assessed through three prospective trials: TITAN, TITAN II, and REDUCE-FMR.
Despite its promise, indirect mitral annuloplasty remains a still largely investigational approach in the management of SMR. Initial feasibility studies such as the AMADEUS trial and TITAN trial reported reductions in annular diameter, modest decreases in MR grade, and improvements in functional status or 6-minute walk distance. [78–80] However, in those early experiences, a substantial proportion of patients initially screened for the procedure could not receive a permanent implant—for reasons including inability to cannulate the coronary sinus, lack of sufficient MR reduction, or compression of coronary structures.[78,80,81]
More importantly, although the randomized REDUCE-FMR trial provided some encouraging data—showing a modest but statistically significant regurgitant volume reduction and reverse LV remodeling at 1 year in treated versus control groups—the overall patient numbers remain small, and roughly 14% of planned implants in the treatment arm could not be performed because of anatomical unsuitability or risk of coronary compromise.[82–84] Long-term data are still sparse; reports of survival and MR-status at 6-year follow-up in single-center series exist but involve very limited cohorts, often with substantial selection bias.[85,86] As a consequence, the real-world applicability of coronary-sinus–based annuloplasty remains restricted, many patients being anatomically unsuitable, and the durability, safety over time (especially risks such as coronary compression, device dislocation, or loss of MR reduction), and comparative efficacy versus other transcatheter repair strategies remain uncertain.[81,82]
The TITAN trial recruited 53 patients with ischemic or nonischemic cardiomyopathy, LVEF < 40%, and moderate-to-severe SMR.[87] Due to anatomical constraints, 17 patients were unsuitable for implantation.[87] However, those who received the device showed marked improvements at 12 months, including a significant decrease in RV (from 34.5 ± 10.2 mL/beat to 17.4 ± 8.1 mL/beat, p < 0.001), alongside reductions in LV end-diastolic volume (from 208.5 ± 45.3 mL to 178.9 ± 40.1 mL, p = 0.015) and end-systolic volume (from 151.8 ± 38.7 mL to 120.7 ± 34.2 mL, p = 0.015).[87] Nonimplanted patients, conversely, experienced progressive ventricular enlargement.[87] Functional gains, evidenced by improved 6-minute walk distances, persisted through 24 months.[64,87]
Subsequent trials further validated these findings. The TITAN II study confirmed the device’s favorable safety profile and sustained functional enhancements,[88] while the REDUCE-FMR trial, a doubleblind, sham-controlled, randomized study, demonstrated robust efficacy.[89] In REDUCE-SMR, Carillon device recipients exhibited a notable reduction in RV (−7.1 mL/beat versus +3.3 mL/beat in controls) and beneficial LV remodeling, with decreases in end-diastolic (−10.4 mL versus +6.5 mL) and end-systolic volumes (−6.2 mL versus +6.1 mL) (89). Patients also reported significant improvements in NYHA functional class compared to the sham group, highlighting the device’s therapeutic potential.[89]
Contemporary developments in transcatheter mitral annuloplasty underscore the critical role of patient-specific anatomical evaluation and tailored device application, guided by MR severity and etiology. Advanced imaging modalities, such as 3D echocardiography and CT, are now integral to preprocedural planning and real-time procedural guidance, ensuring precise anchor placement and minimizing complications. Innovations in indirect annuloplasty devices focus on enhancing procedural safety and achieving durable annular reduction while safeguarding adjacent structures, notably the circumflex coronary artery. The Carillon system illustrates the potential of minimally invasive SMR treatment, with evidence suggesting improvements in mitral valve competence and quality of life in selected high-risk patients. Nevertheless, further studies are essential to refine patient selection algorithms, optimize long-term device performance, and address anatomical variability.
TMVR has carved a niche in treating mitral valve disease for patients too high-risk for surgery. Initially, clinicians repurposed aortic transcatheter heart valves for mitral valve-in-valve (ViV), ViR, or ViMAC procedures, but these off-label uses faced substantial anatomical limitations, particularly the risk of neo-LVOT obstruction and the challenge of securing stable anchoring—rather than issues related to annular dynamics, which are often reduced in patients with long-standing MR or severe MAC. Purpose-built mitral THVs now address these challenges, offering tailored solutions for native, noncalcified mitral pathology. However, TMVR’s complexity—marked by risks like LVOT obstruction and coronary artery compression— demands meticulous preprocedural planning to ensure success.
Advanced imaging drives TMVR’s narrative. TOE evaluates mitral pathology, from regurgitation to prosthetic dysfunction, while cardiac multidetector computed tomography (cMDCT) provides the definitive anatomical road map. cMDCT measures leaflet heights, annular geometry, and identifies features like the “myocardial shelf” in dilated ventricles or caseous calcification, a rare MAC variant that heightens procedural risks.[89] It also predicts LVOT obstruction by assessing the neo-LVOT area (≤ 1.7 cm2, indicating high risk) and optimizes fluoroscopic angles for precise deployment.[89] During procedures, fluoroscopy guides real-time positioning, and TTE confirms valve function. As TMVR evolves with refined devices and imaging, it promises enhanced outcomes for complex mitral disease, though optimizing patient selection and long-term durability remains critical.
The MViV approach has emerged as the cornerstone of TMVR, offering a lifeline to patients with failing mitral bioprostheses who face high surgical risks. Early MViV procedures leaned on balloone-xpandable aortic THVs, such as the Sapien 3, delivered via transseptal or transapical routes.[64] Registries like VIVID and TVT reported transseptal access in 18.5% and 49% of cases, respectively, with 30-day mortality rates of 8%–9%.[64] The MITRAL trial, focusing exclusively on transseptal MViV, achieved a lower 30-day mortality of 3.3% in a select cohort with experienced operators, and at 5 years, MViV patients showed a remarkable 21.4% all-cause mortality, with 94.7% in NYHA class I or II and stable valve function (mean mitral gradient 6.6 ± 2.5 mm Hg).[90] Similarly, the PARTNER 3 MViV study in intermediate-risk patients reported no mortality or stroke at 1 year, with 87.2% of patients improving NYHA class and all showing mild or no MR, highlighting MViV’s efficacy in less severe cases.[91]
Furthermore, real-world evidence underscores the clinical benefits associated with MViV procedures. Whisenant et al.’s analysis of over 1,500 SAPIEN 3 MViV cases from 2015–12019 revealed a 96.8% procedural success rate, with 30-day and one-year mortality at 5.4% and 16.7%, respectively.[64,92] Transseptal access outperformed transapical, with one-year mortality of 15.8% versus 21.7%, cementing its preference when anatomically feasible.[64,92] Yet, challenges persist: 60%–80% of MViV patients exhibit elevated transvalvular gradients postprocedure, often underestimated by intraprocedural TOE. In vitro studies attribute this to stent frame irregularities, such as eccentricity or underexpansion, prompting exploration of intravascular ultrasound to optimize stent and leaflet deployment.[93,94] Both the MITRAL and PARTNER 3 studies underscore sustained quality-of-life improvements, with significant Kansas City Cardiomyopathy Questionnaire score gains, though higher residual MR in MITRAL’s ViR and valve-in-MAC cohorts correlated with increased mortality.[64,91] As MViV evolves, refining imaging, device design, and patient selection will be critical to maximizing long-term outcomes.
MViR addresses the complex challenge of failed surgical annuloplasty rings, where anatomical and hemodynamic constraints demand precise intervention. Early data from the VIVID and TVT registries revealed 30-day mortality rates of 11%–13%, reflecting the procedure’s initial hurdles.[64] The MITRAL trial marked a turning point by adopting an exclusively transseptal approach, reducing 30-day mortality to 6.7% and achieving a one-year mortality of 23.3%, notably lower than the predicted STS score of 7.6%.[95] At 2 years, mortality reached 43.3%,[64,90] comparable to outcomes with purpose-built mitral devices like Tendyne and aligning with TEER using MitraClip,[66] underscoring MViR’s viability for high-risk patients. These advancements led to FDA approval for MViR in high surgical risk patients in May 2021.
Despite progress, MViR faces challenges, particularly with smaller valve sizes and rigid annuloplasty rings, which often result in elevated transvalvular gradients. Contemporary TVT registry data show a decline in 30-day mortality from ~11% in 2015-2016 to 6% in 2020,[64] driven by increased transseptal access adoption, mirroring MITRAL trial outcomes. This shift highlights the importance of access route optimization and patient-specific anatomical planning to mitigate hemodynamic complications. Advanced imaging, such as TOE and CT, remains critical for tailoring device selection to annular geometry and ring characteristics. As MViR evolves, ongoing research into optimizing valve sizing, reducing gradients, and refining procedural techniques will be essential to enhance longterm outcomes and broaden its applicability in complex mitral valve disease.
ViMAC tackles one of the most significant challenges in structural heart interventions, targeting patients with severe MAC who face elevated surgical risks due to advanced age, female predominance, and cardiovascular comorbidities. Multimodal imaging, particularly cardiac CT, is pivotal, enabling precise grading of MAC severity and guiding procedural planning with tools like the validated MAC-CT score, which predicts outcomes in mitral interventions.[96] Early ViMAC experiences, as captured in the TMVR in the MAC global registry (n = 116; median STS score, 15.3%), revealed sobering 30-day and one-year mortality rates of 25% and 53.7%,[97] respectively, with similar findings in the Cedars-Sinai registry (n = 58; 30-day mortality, 34.5%).[98] These outcomes were driven by complications such as paravalvular leak (PVL), valve embolization, and LVOT obstruction, with the latter identified as the strongest predictor of one-year mortality (adjusted HR 2.63).[64,97]
Advancements in ViMAC have focused on mitigating LVOT obstruction through septal reduction and anterior leaflet modification strategies. Techniques like alcohol septal ablation (MITRAL trial) [99] and percutaneous leaflet laceration (LAMPOON) [64] have improved outcomes, with preprocedural CT assessing neo-LVOT area (< 200 mm2 indicating high risk) and Skirt Neo-LVOT dimensions to guide strategy selection.[64] In the MITRAL trial, trans-septal ViMAC with preemptive alcohol septal ablation reduced 30-day mortality to 6.7% compared to 21.4% for transatrial access,[64] with a two-year mortality of 39.9%, comparable to PARTNER 1 TAVR outcomes (43.3%).[100] Stable mitral gradients (6.7±2.5 mm Hg at two years) and improved Kansas City Cardiomyopathy Questionnaire scores underscored sustained functional benefits.[90] The ongoing MITRAL II trial evaluates transseptal ViMAC in high-risk patients with severe MAC and mitral dysfunction, targeting all-cause mortality HF hospitalization at one year.[64] As ViMAC evolves, refining imaging, preventive strategies, and patient selection will be critical to improving survival and expanding its role in complex mitral disease management.
TMVR for native, noncalcified mitral valves represents an expanding therapeutic frontier for patients with severe MR who are unsuitable for surgery or TEER. The Tendyne system, a selfexpanding, trileaflet porcine bioprosthesis delivered transapically, has become the most clinically mature platform in this space.[64] Its dual-frame design and apical tether enable stable anchoring across a wide range of annular anatomies.[64] In the Tendyne Global Feasibility Study of 100 high-risk patients (90% SMR; mean STS 7.9%; mean LVEF 46.6%), procedural success exceeded 95%, with 0% intraprocedural mortality, 6% 30-day mortality, and one-year mortality of 26%, comparable to outcomes reported for MitraClip in the TVT registry and early TAVR cohorts.[64,97,101] At one year, 98% of patients had none or trace MR,[97,101] accompanied by significant symptomatic and quality-of-life improvement, supporting its CE Mark approval in 2020 [64,102] and subsequent full commercial approval in both the EU and the US in 2025 for patients with severe MR and MAC not amenable to repair.
Long-term follow-up demonstrates sustained hemodynamic benefit: at two years, MR elimination remained stable and HF hospitalizations decreased markedly (0.51 versus 1.30 events/patient-year pre-procedure).[103] Early experience in patients with severe MAC showed feasibility but with high late mortality,[104] mirroring results from the MITRAL ViMAC cohort. The ongoing SUMMIT trial [105] continues to evaluate Tendyne in both non-calcified MR and MAC anatomies and will refine the device’s optimal clinical niche.
Alongside Tendyne, the TMVR field is rapidly evolving. The SAPIEN M3 system, a fully transseptal, transfemoral TMVR platform, recently reported promising one-year results in the pivotal ENCIRCLE trial, with MR ≤ 1+ achieved in ~95% of patients, improvement in functional class, and acceptable procedural risk.[106] These developments signal a transition toward truly percutaneous TMVR solutions for anatomically complex MR. Nevertheless, TMVR remains restricted by anatomical suitability, risk of LVOT obstruction, durability concerns, and the need for long-term comparative data versus TEER.
PVLs emerge as a significant challenge following surgical mitral valve replacement, affecting 5%–17% of patients,[64] with most (approximately 74%) identified within the first postoperative year.[107] Risk factors driving PVL formation include prior infective endocarditis, extensive MAC, chronic corticosteroid therapy, mechanical prostheses, supra-annular valve positioning, and continuous suturing techniques.[108–111] While frequently asymptomatic, PVLs can lead to serious complications such as congestive HF, hemolytic anemia due to blood shear stress, and recurrent endocarditis, prompting percutaneous closure when anatomy permits. Absolute contraindications, such as active infection or dehiscence exceeding one-third of the sewing ring,[64] restrict intervention, underscoring the need for careful patient selection to optimize outcomes.
Imaging is the cornerstone of PVL diagnosis and management, guiding clinicians through a complex interventional landscape. TTE, despite limitations from acoustic shadowing, detects transvalvular flow abnormalities and supports postprocedural monitoring. 3D-TOE stands as the gold standard, offering detailed assessment of defect location, severity, and hemodynamic parameters like color Doppler jet characteristics, VC width, EROA, RV, and pulmonary pressures. Cardiac CT, often integrated with fluoroscopy via CT-fluoroscopy fusion, provides high-resolution anatomical insights and facilitates intraprocedural wire crossing and device deployment.[64,107]
Percutaneous PVL closure employs three main approaches— anterograde, retrograde, and transapical—selected based on defect location and complexity. The anterograde transseptal approach, most frequently used, involves femoral venous access, transseptal puncture, and a venoarterial loop formed by snaring the wire in the ascending aorta, allowing precise delivery of occluder devices through the leak.[64] The retrograde approach, using femoral arterial access, creates an arteriovenous loop for better alignment with certain defects, while the transapical method, though less common due to higher complication risks, simplifies wire manipulation.[64] Post deployment, fluoroscopy and TOE verify device positioning, residual regurgitation, transvalvular gradients, and the absence of complications (pericardial effusion), ensuring procedural success.
Large registries demonstrate the growing promise of percutaneous PVL closure despite technical challenges and variable device availability. A Spanish multicenter[112] registry of 469 patients (70% mitral PVL) reported technical success rates of 86.6%, with low major complication rates.[110,112] A UK–Ireland registry (n = 259; 44% mitral PVL) achieved 91% device implantation success, with low in-hospital mortality (2.9% for elective, 6.8% for urgent procedures); 74.7% of patients had no or mild residual PVL, and NYHA functional class improved significantly.[113] These findings highlight the safety and efficacy of PVL closure, but ongoing research into device optimization and patient selection remains critical to further enhance outcomes in this complex mitral intervention.
Although transcatheter mitral interventions have expanded rapidly across Western European countries, their adoption in Central and Eastern European (CEE) states remains markedly heterogeneous. A recent survey by EAPCI found that the penetration of percutaneous mitral valve repair (PMVR) procedures across Europe remains marginal compared with transcatheter aortic valve replacement, with many centers citing economic, regulatory, and logistic hurdles as limiting factors.[114]
A practical consideration is the learning curve and procedural volume required to achieve proficiency in TEER: analyses from the TVT registry and multicenter studies demonstrated a clear inflection in performance metrics after approximately 50 implants, with continued improvements observed up to roughly 200 procedures per operator/center.[115] An EAPCI survey similarly reported low annual PMVR volumes across many European centers in 2014—about 58% of centers performed ≤ 10 procedures, 15.5% performed 10–40, and only ~26% exceeded 40 procedures per year—highlighting a structural barrier to rapid diffusion of these therapies.[114] National and registry data further indicate that higher-volume institutions tend to achieve better in-hospital outcomes and lower complication rates, supporting the rationale for concentrating TEER programs in experienced centers or implementing structured proctoring and mentoring pathways for newly adopting sites.[116]
National reimbursement policies and institutional infrastructure strongly influence access. In several middle- and lower-income European countries, structural-heart programs are underfunded or constrained by administrative caps, limiting patient access to newer mitral therapies.[117,118]
Operator experience and availability of advanced imaging modalities represent additional barriers. High-quality multimodality imaging (3D echocardiography, CT) and experienced interventional-imaging teams are essential for safe and effective mitral interventions—resources which are unevenly distributed across the region. This imbalance echoes broader inequalities in interventional cardiology across Europe, shaped by socioeconomic, geographic, and policy-related disparities.
Moreover, institutional factors, such as the presence of a dedicated structural-heart program, a hybrid cath lab, and the required ancillary staff (anesthesiology, echo, nursing) further impact the feasibility of establishing mitral intervention services. In many CEE countries, the lack of such comprehensive infrastructure constrains the introduction of newer devices beyond high-volume tertiary centers.
Despite these obstacles, the landscape is gradually evolving. Some centers in Central and Eastern Europe have begun integrating structural-heart therapies, supported by growing participation in European registries and increased efforts to harmonize training and procedural pathways. Continued emphasis on expanding imaging capabilities, standardizing training, and aligning reimbursement frameworks remains crucial to ensure equitable access to transcatheter mitral therapies across Europe.
Transcatheter therapies have substantially expanded the therapeutic options for patients with MR who are unsuitable for surgery. Among these, TEER is now the only established modality with guideline-supported indications and robust randomized data demonstrating reductions in HF hospitalizations and improvements in quality of life in appropriately selected SMR patients. In contrast, both indirect and direct percutaneous annuloplasty techniques, as well as TMVR, remain investigational. Their current evidence is derived mainly from small early-phase studies, anatomically restricted patient cohorts, and feasibility registries rather than large randomized trials.
Advanced multimodality imaging plays a central role across all interventions, informing MR mechanism, anatomical feasibility, procedural strategy, and anticipated device durability. A structured, patient-centered approach—integrating imaging findings, clinical status, and device-specific considerations—remains essential to achieving safe and durable outcomes.
Looking forward, key evidence gaps persist, including harmonized imaging-based selection algorithms, standardized definitions of procedural success, durability data for emerging devices, and guidance on therapy sequencing in complex anatomical subsets. Ongoing randomized trials, prospective registries, and European real-world data will be crucial to refining indications, improving accessibility, and firmly establishing the long-term role of transcatheter mitral therapies in contemporary cardiovascular care.