In recent years, leadless devices have become increasingly common, particularly in hemodialysis patients, those at high risk of infection, and patients lacking an appropriate pathway for conventional access. Implantation of such devices is usually performed via the femoral vein. In sporadic cases, venous access is impossible due to anatomical malformations, stenosis, or occlusions caused by other factors. Leadless pacemakers are implanted directly into the right ventricular septum, thereby avoiding intravascular complications associated with conventional pacemakers.
We present the case of a 58-year-old male patient, a smoker with a very complex medical history, who presented to the Emergency Department of the Heart Institute, Cluj-Napoca, for repeated syncopal episodes. His past medical history included bilateral nephrectomy due to renal carcinoma, chronic hemodialysis for the past 5 years, partial colonic resection, and aortic valve endocarditis.
On admission, the electrocardiogram revealed complete atrioventricular (AV) block with a junctional escape rhythm at 15 beats per minute. The patient was hemodynamically stable, with a blood pressure of 120/80 mmHg and no requirement for vasoactive support. Oxygen saturation was 93% while receiving 2 L/min oxygen via nasal cannula. Cardiac auscultation revealed a grade IV/VI systolic murmur audible across the entire precordium. The patient was immediately transferred to the angiography laboratory for temporary pacing.
An initial temporary pacing attempt was performed, but lead displacement occurred within hours. The position of the temporary pacing probe was reassessed, revealing right ventricular positioning with intermittent capture. The patient was completely dependent on pacing. Given the absence of other venous access, a new pacing probe was introduced through a 23 cm 6F sheath via the right femoral vein and positioned in the right ventricle with optimal parameters, ensuring effective stimulation. The femoral sheath was removed, and the pacing probe maintained stable right ventricular apical capture at 80 bpm and 12 V. No immediate post-procedure complications were observed. The patient was transferred to the intensive care unit, where continuous telemetry confirmed complete pacemaker dependency.
Laboratory tests revealedgrade III anemia (hemoglobin 6.8 g/dL), creatinine 9.12 mg/dL, urea 140 mg/dL, potassium 6.2 mmol/L, sodium 134 mmol/L, and CRP 70 mg/L. Three sets of blood cultures were negative, as were tracheal aspirate and other secretion cultures.
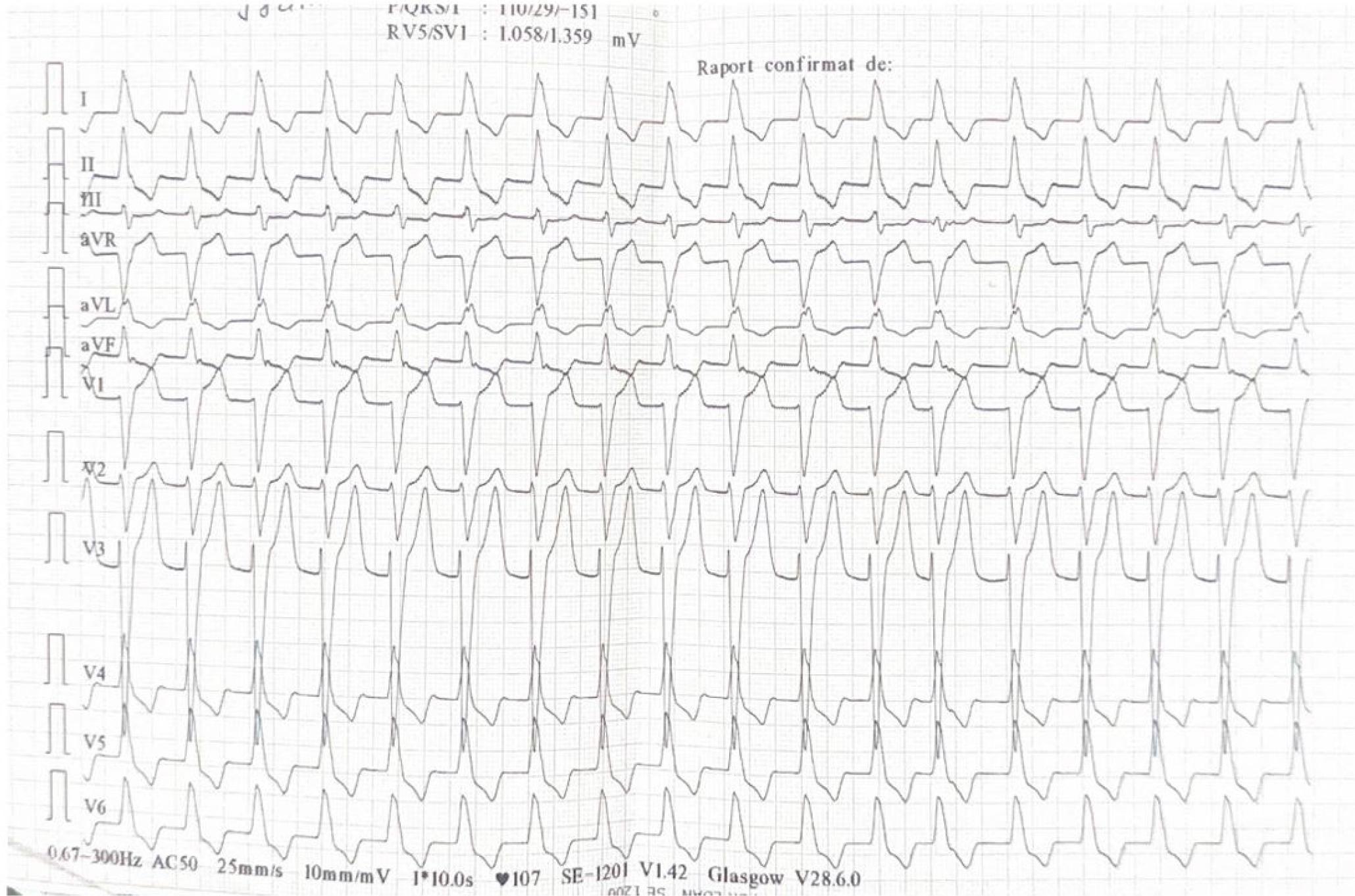
Atrial fibrillation, left bundle branch block.
Cardiac ultrasound demonstrated severe mitral stenosis with extensive calcifications, significant left ventricular hypertrophy (IVS 21 mm) with intraventricular obstruction (resting gradient >70 mmHg), and impaired LV function (Summary Figure 2). Additional findings included a nondilated aorta (Ao asc 31 mm, annulus 22 mm, sinus 33 mm), markedly enlarged left atrium (55 mm, 114 ml), concentric LV hypertrophy (IVS 21 mm, PW 13 mm, LV 40/20 mm) with reduced GLS (–12.6%, apical sparing), severe calcific mitral stenosis (Vmax 2.5 m/s, mean gradient 12.7 mmHg, valve area 0.9 cm2), dynamic LVOT obstruction (late-peaking Vmax 4 m/s, Gmax 66 mmHg) with only mild aortic stenosis (Vmax 2.3 m/s, mean gradient 22 mmHg, valve area 1.6 cm2), normal stroke volume (70 ml), mildly enlarged but functional RV (35 mm, TAPSE 19 mm), mild tricuspid regurgitation (grade I/II, RV–RA gradient 36 mmHg), no pulmonary hypertension, and a normal IVC (14 mm with >50% inspiratory collapse).
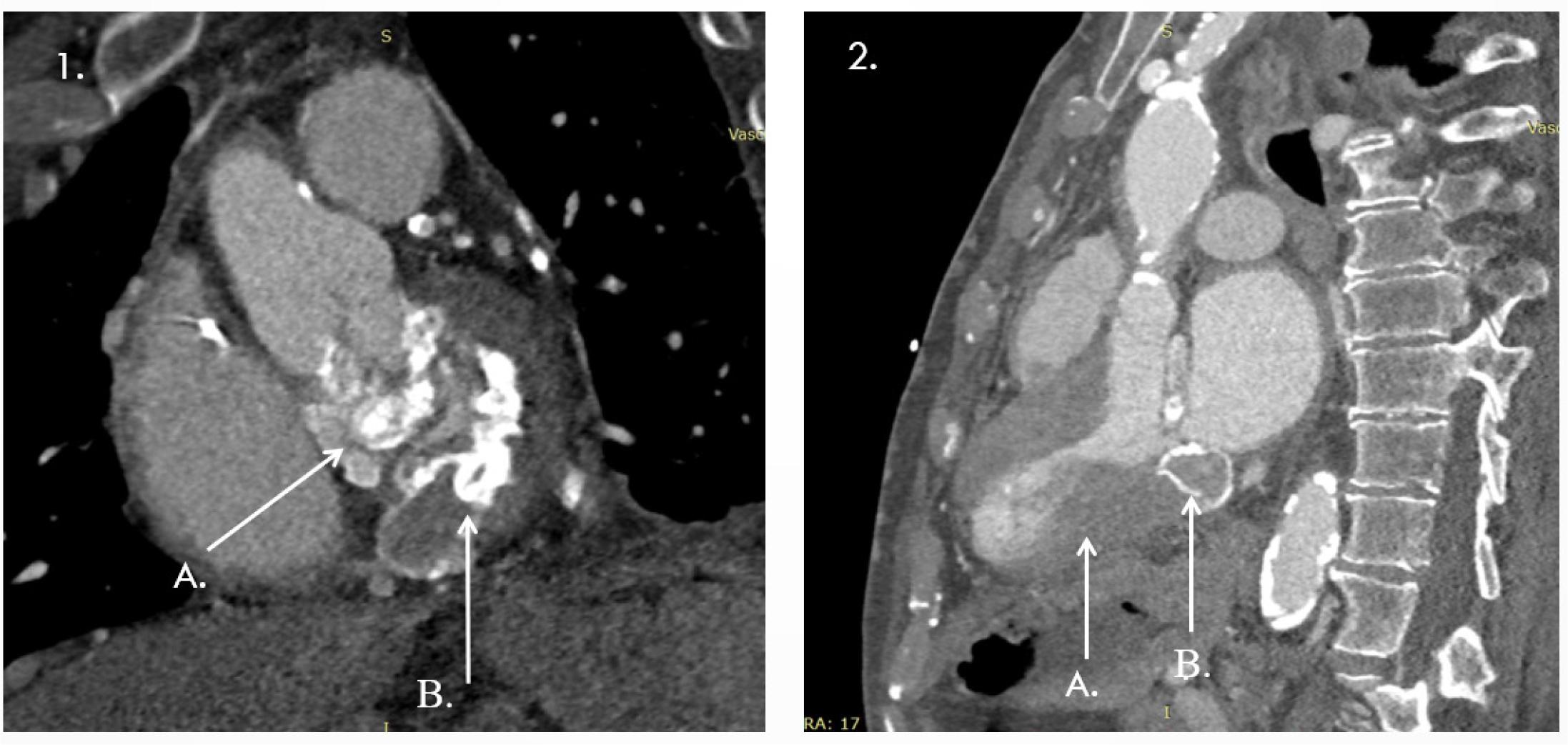
1 A, B Annular calcification extending to the mitral-aortic continuity and aortic root; 2 A-Important left ventricular hypertrophy; B-Toothpaste tumour.
A multidisciplinary Heart Team (clinical cardiologist, electrophysiologist, cardiovascular surgeon, interventional cardiologist, radiologist, and anesthesiologist) was convened to guide further management. The consensus was to perform a CT scan to obtain detailed imaging of the venous axis of both upper and lower limbs.
A thoracic CT scan confirmed the temporary pacemaker lead at the right ventricular apex. It also showed extensive mitral annular calcification extending to the mitral-aortic continuity and aortic root, with two small cavitary defects (up to 30 mm), as well as vascular calcifications involving the aorta, supra-aortic trunks, and proximal coronary arteries (Summary Figure 3). Additional findings included distal SVC occlusion by a ~4 cm thrombus with a hyperdense peripheral component, likely a fractured catheter fragment. Another catheter fragment (35 × 6 mm) was identified in the left brachiocephalic vein with a thrombosed lumen and partial obstruction. Extensive thoraco-abdominal collateral venous circulation was noted. The left subclavian, brachiocephalic, jugular veins, and IVC were patent, though the proximal and mid-left subclavian vein showed stenosis, likely residual thrombus (Summary Figure 4).
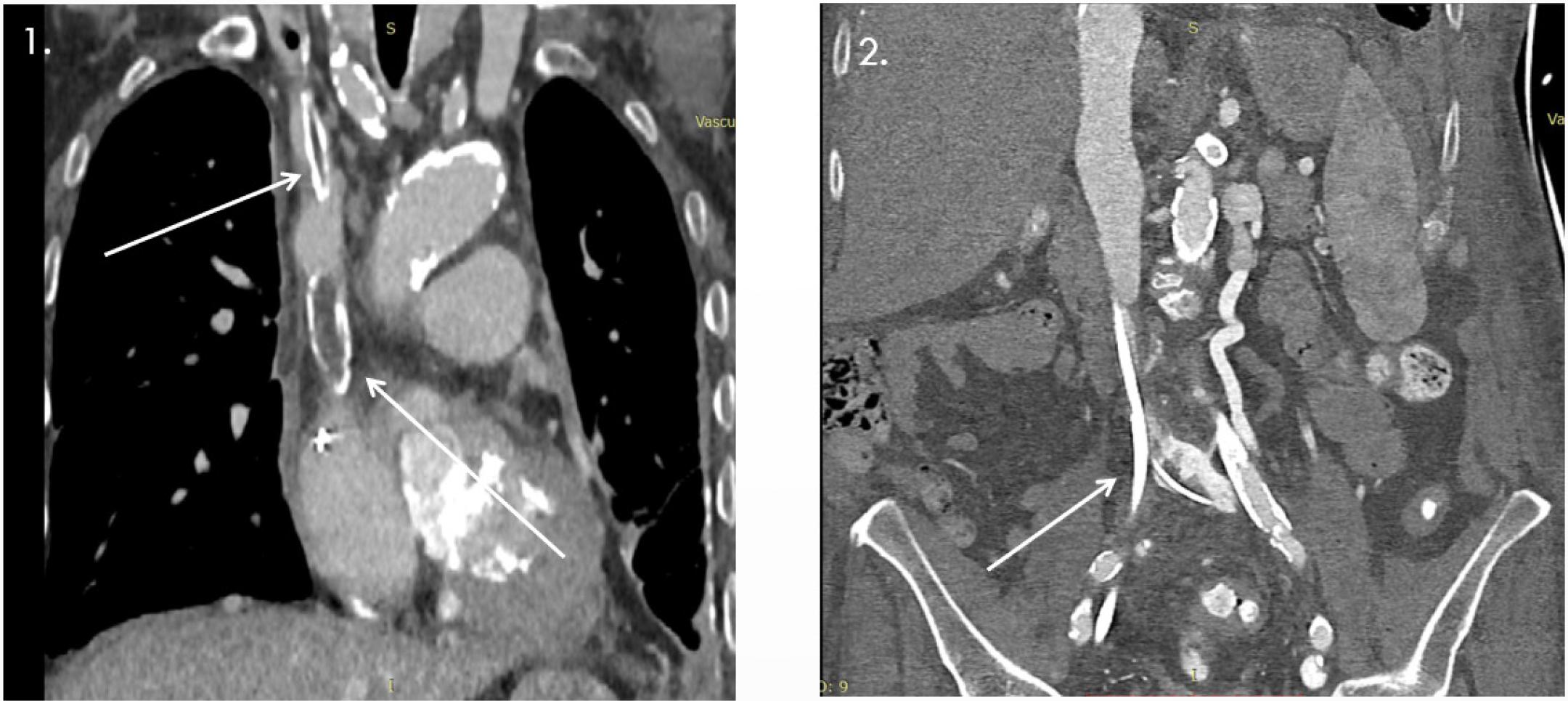
1- Oclussion VCS due to ‘forgotten* dialysis catheter; 2 - Stenosis and oclussion of inferior vena cava and collateral circulation between the left common iliac vein and the left renal vein.
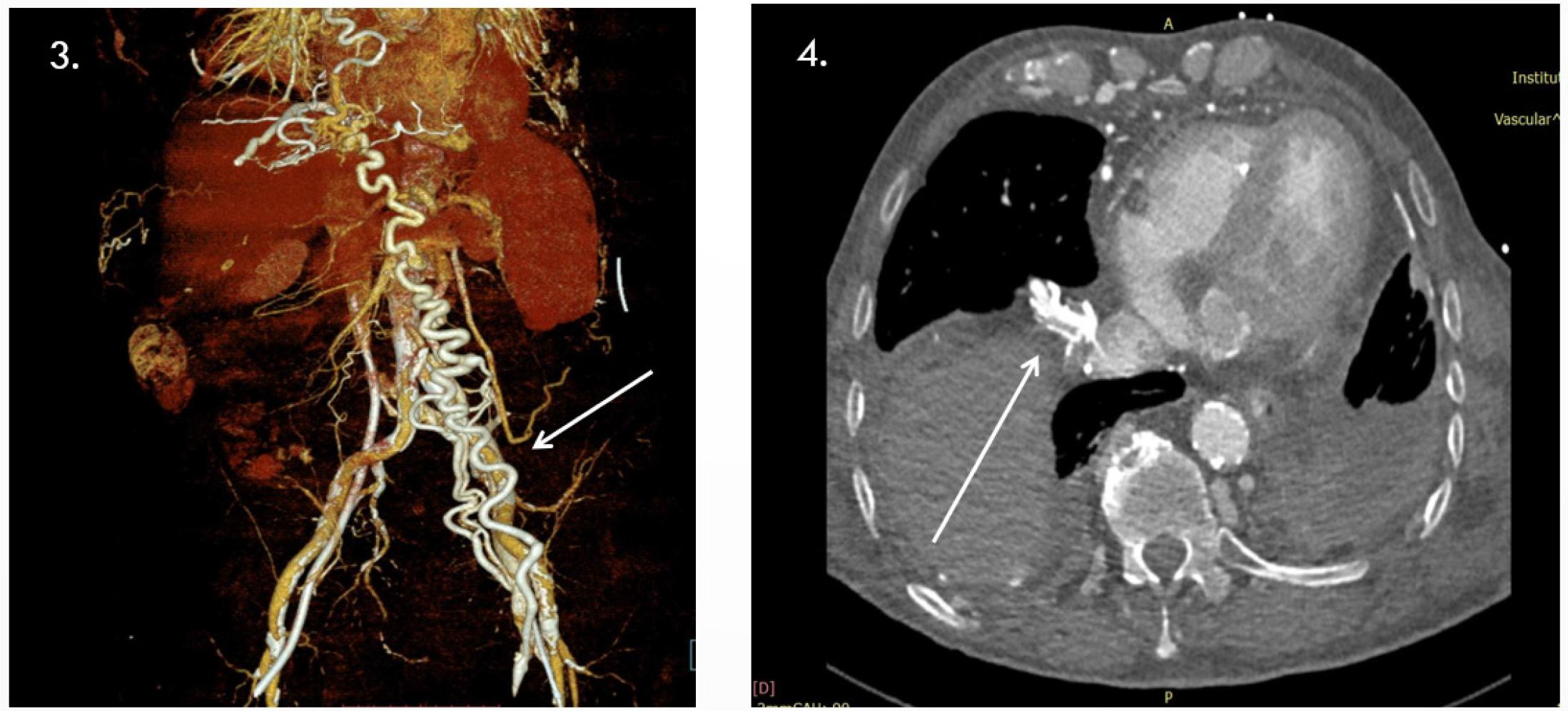
3- Important collateral circulation thoraco-abdominal; 4- Important collateral circulation thoraco-abdominal.
Contrast-enhanced CT via the right antecubital vein revealed severe proximal IVC stenosis (~9 mm length, residual lumen 1–2 mm, 2.5 cm above iliac confluence) with partially calcified thrombus, extensive collateral venous circulation, ruptured calcified catheter fragments, and a right iliac venous stent. The left common iliac vein was ligated with collateral flow; the left renal vein was dilated. The aorta showed diffuse calcification and stenosis, with focal ectasia and thrombus at the IMA origin raising suspicion of a small aorto-duodenal fistula. Significant stenoses were also noted at the SMA and celiac origins, with heavily calcified iliac arteries (Summary Figures 5).
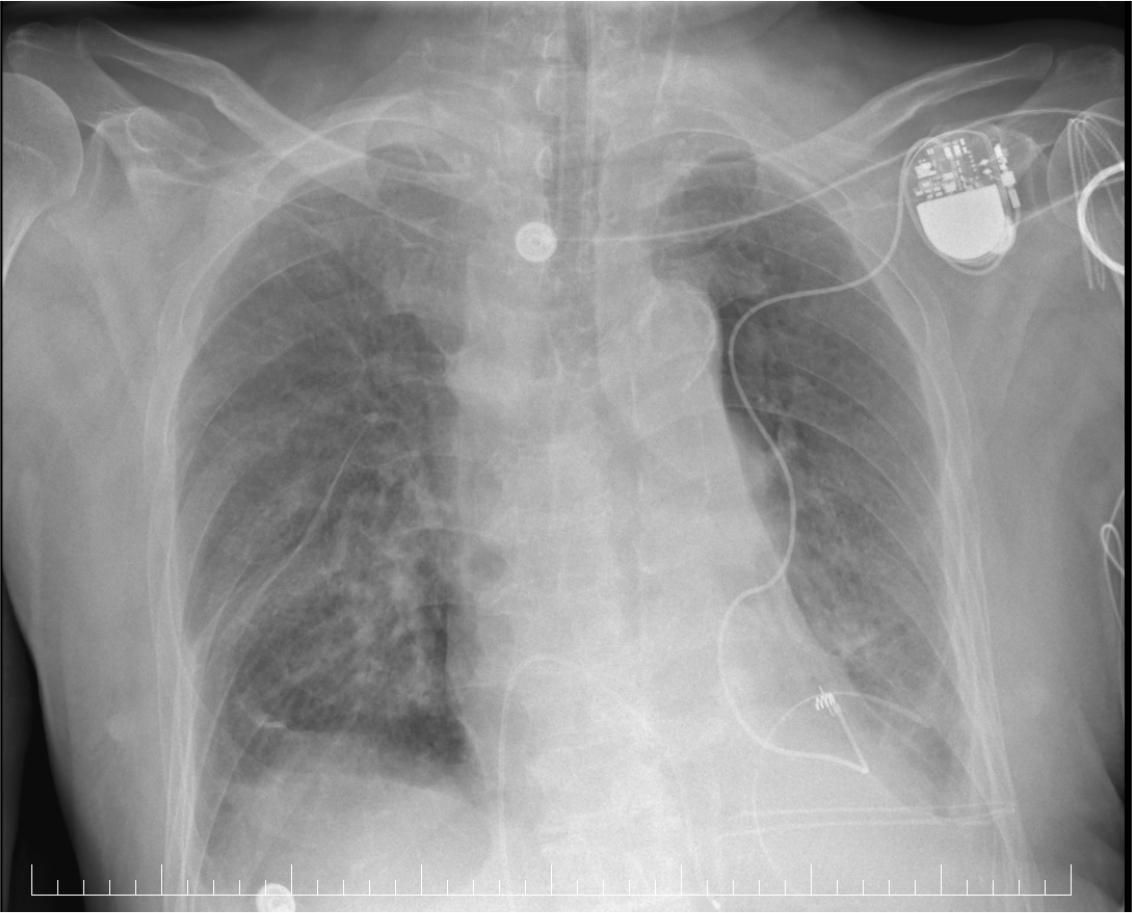
X ray-pacemaker and epicardial lead placement.
Transesophageal echocardiography (TOE) was performed due to the patient’s history of endocarditis. Despite being limited by intolerance, TOE excluded left atrial thrombus and revealed no signs of infectious endocarditis. Findings confirmed transthoracic results: severe mitral stenosis, grade II mitral regurgitation, severe aortic stenosis, grade I aortic regurgitation, and grade I/II tricuspid regurgitation. During hospitalization, the patient experienced recurrent episodes of acute pulmonary edema, managed with oxygen therapy and medical treatment, as well as dialysis sessions following the administration of contrast medium. According to ESC pacing and CRT guidelines, the patient had an indication for Micra™ leadless pacemaker implantation, particularly given the infeasibility of conventional transvenous access. However, due to complete venous obstruction, epicardial pacing was selected. Under general anesthesia, left anterior thoracotomy was performed in the 5th intercostal space (~7 cm). Partial pericardiotomy was performed with careful adhesiolysis. The lateral-apical LV wall was identified, and an epicardial electrode was implanted. The lead was tunneled into a subclavicular pocket housing a Medtronic Viva CRT device. The pocket was closed in layers, hemostasis was achieved, a suction drain was placed, and the thoracotomy incision was closed in layers with skin suturing.
Postoperative chest X-ray confirmed correct epicardial lead placement with no pneumothorax or complications (Summary Figure 5). The patient remained hemodynamically stable, was extubated within 24 hours, and the ECG showed atrial fibrillation with left bundle branch block. (Summary Figure 1). Hemodialysis sessions were performed as scheduled. Laboratory tests showed stable grade II anemia, persistent azotemia, and elevated CRP, with no clinical or paraclinical signs of infection. Serial chest radiographs confirmed pulmonary status and lead placement.
At one week post-procedure, the patient developed acute pulmonary edema refractory to medical therapy, followed by cardiorespiratory arrest unresponsive to resuscitation. Autopsy confirmed acute pulmonary edema as the immediate cause of death.
When femoral access is not feasible (e.g., occlusion, thrombosis, prior surgery, post-thrombotic syndrome), leadless pacemaker implantation becomes highly challenging. At present, no officially approved alternative venous route exists. The literature describes rare cases of Micra implantation via the internal jugular vein and, less commonly, via subclavian or other central veins. These techniques are off-label, technically demanding, and should be undertaken only in centers with advanced expertise.
In our case, both superior and inferior venous access were occluded: the SVC due to a retained fractured dialysis catheter fragment with thrombus, and the IVC due to severe stenosis/occlusion. Extensive collateral circulation was present. Although a temporary pacing lead was advanced via the IVC, attempting to introduce the 27-French Micra introducer through stenotic and fibrosed veins carried a prohibitive risk of vessel perforation. The lack of appropriately sized covered stents further argued against this approach.
This case highlights the complexity of managing a polyvascular patient with multiple comorbidities. Leadless Micra pacemakers are increasingly recognized in the ESC guidelines on pacing and resynchronization as lifesaving in dialysis patients, cachectic patients, those at high risk of infection, or when conventional venous access is not available.
In this patient, Micra implantation was not feasible due to venous obstruction. Literature reports rare alternative approaches (internal jugular or subclavian), as well as isolated cases of successful venous dilation for transvenous access; however, following Heart Team discussion, these were deemed inappropriate.
To our knowledge, this is the first case at the Institute of the Heart in which Micra implantation could not be performed due to the absence of a viable venous approach.