Erdheim-Chester disease (ECD) is a rare non-Langerhans cell histiocytosis characterized by a clonal proliferation of foamy histiocytes affecting multiple organ systems. Its clinical presentation is highly variable, often leading to delayed diagnosis. [1]While bone pain is the most frequent symptom, patients may also present with endocrine, neurological, pulmonary, or cardiovascular involvement.
Cardiac manifestations, seen in up to 70% of cases, include pericardial effusion, myocardial infiltration, and pseudotumoral masses, which can mimic malignancy or infection. Such presentations are clinically significant due to their impact on prognosis and diagnostic complexity. Multimodality imaging—including cardiac magnetic resonance imaging (MRI) and positron emission tomography (PET)/computer tomography (CT)—plays a crucial role in recognizing the disease pattern and guiding biopsy. [2] Detection of BRAF V600E or MAPK pathway mutations has enabled targeted treatments with BRAF or MEK inhibitors, improving outcomes in many patients.
This report presents a rare case of ECD initially diagnosed through cardiac involvement, highlighting the importance of considering this entity in atypical cardiovascular presentations and the value of imaging in early diagnosis. [3]
We present the case of a 68-year-old man with a history of polyvascular diseases (left renal artery stenosis with conservative management and ischemic heart disease following a STEMI with angioplasty for a univascular lesion) who was admitted for the re-evaluation of persistent pericardial effusion despite treatment with standard anti-inflammatory regimen during subsequent visits in the context of ischemic heart disease follow-up. Considering the persistent nature of the pericardial fluid, a cardiac magnetic resonance (CMR) was performed, confirming the circumferential pericardial fluid, without signs of cardiac compression. In addition, a mass-like process sheathing the visceral pericardium in relation to the base of the inferior vena cava, the posterior wall of the right atrium and extending towards the interatrial septum and the free wall of the left atrium was observed (Figure 1). The pattern and extent of these lesions were pathognomonic of ECD, but further testing was needed to confirm it. Considering the systemic nature of the disease, a PET scan was performed to look for other organ involvement and other possible biopsy sites. At the cardiac level, the PET scan showed moderate diffuse hypermetabolism involving the internal pericardial layer and increased uptake overlapping with the cardiac mass observed during CMR, suggesting an inflammatory pathology (Figure 2). Concerning other organ involvement, the following lesions were observed: hypermetabolic perirenal infiltration with a “hairy kidney” appearance, moderate bilateral hyperfixation of the adrenals, mild hypermetabolic osteocondensation of the distal regions of the femurs and the distal region of the left tibia. A suspicious clavicular cutaneous lesion has been flagged as a possible manifestation of the disease but the results of the biopsy described a seborrheic keratosis. Considering that the renal lesion was the most easily accessible, a biopsy was performed at this level that showed an inflammatory appearance with presence of macrophages confirming ECD. Genetic testing was positive for the BRAF mutation.
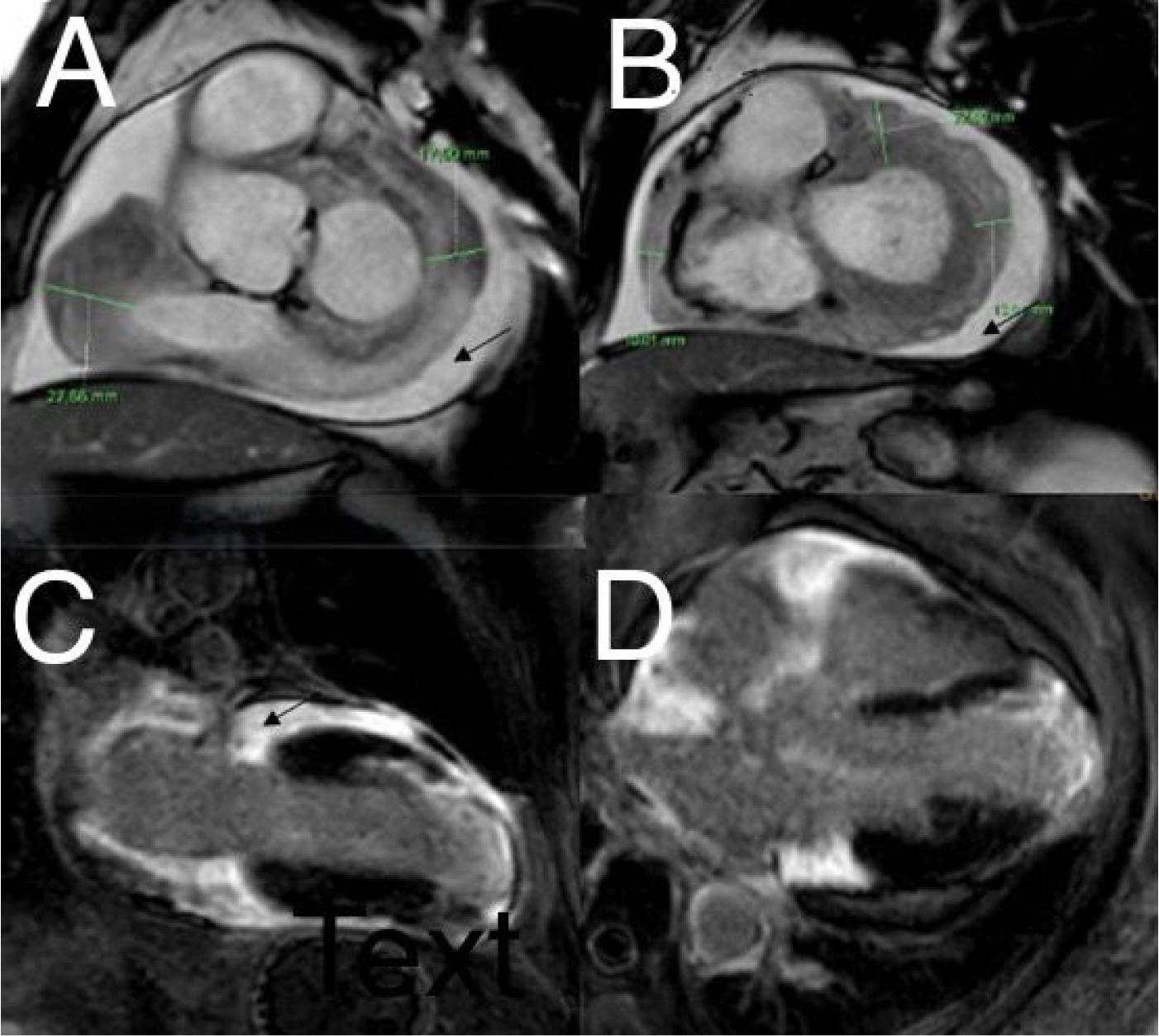
CMR. A, B – Short axis cine steady state free precession sequence showing moderate circumferential pericardial effusion without impact on the cardiac cavities (black arrow). Tumor or pseudotumoral process sheathing the visceral pericardium predominantly facing the right atrium, the base of the inferior vena cava, the posterior wall of the right auriculum extending towards the interatrial septum and the free wall of the left atrium (green lines). C, D – Late gadolinium enhancement sequences showing enhancement zones at the level of the pericardium, superimposable to the topography of the tumor described in the cine images. Additionally, transmural enhancement at the level of the left ventricular apex and the apical segments.
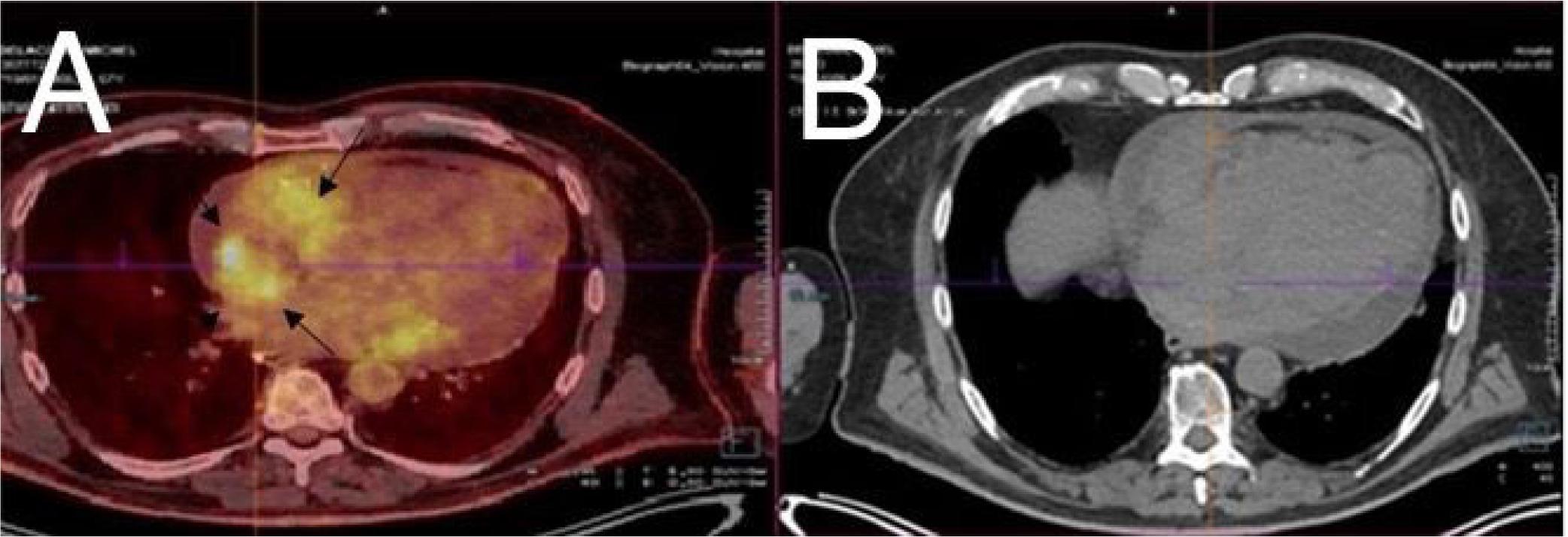
PET-CT SCAN. Transverse section at the level of the heart. A. Moderate diffuse hypermetabolism involving the internal pericardial layer in the context of known pericardial effusion. Increased fixation at the level of known pseudo-tumoral thickenings on the periphery of the right atrium (black arrows) B. Transverse section CT scan without PET at the same level.
In the evaluation of disease extension, a pituitary MRI was performed, showing no detectable pituitary or pituitary stalk abnormality. A renal artery computer tomography (CT) scan showed a moderate stenosis at the origin of the left renal artery, no significant stenosis of the two right renal arteries and periaortic and perirenal infiltration related to known ECD.
Ten months after the initial diagnosis, while investigating the disease, the patient presented with paresthesia in the right arm with visual disturbances. A brain MRI was performed suggesting recent ischemic strokes in the left superficial Sylvian territory. Transesophageal echocardiography (TOE) confirmed the persistent cardiac mass at the roof of the right atrium, sheathing the base of the inferior vena cava and the posterior roof of the right atrium towards the interatrial septum and the free wall of the left atrium, without intracardiac thrombus. Lipomatous hypertrophy of the septum with aneurysm of the interatrial septum without patent foramen ovale visible on color Doppler was also noted.
Re-evaluation after one year of BRAF inhibitor targeted therapy was performed with a control PET scan that showed the disappearance of pericardial hypermetabolism and pericardial effusion and disappearance of pathological hyperfixation at the level of the right atrium (Figure 3). Also, regarding other organ involvement the near-complete metabolic regression of femoral and tibial intramedullary osteocondensations was noted, together with the persistent “;hairy kidney”, appearance with discrete hypermetabolism of the perirenal infiltration.
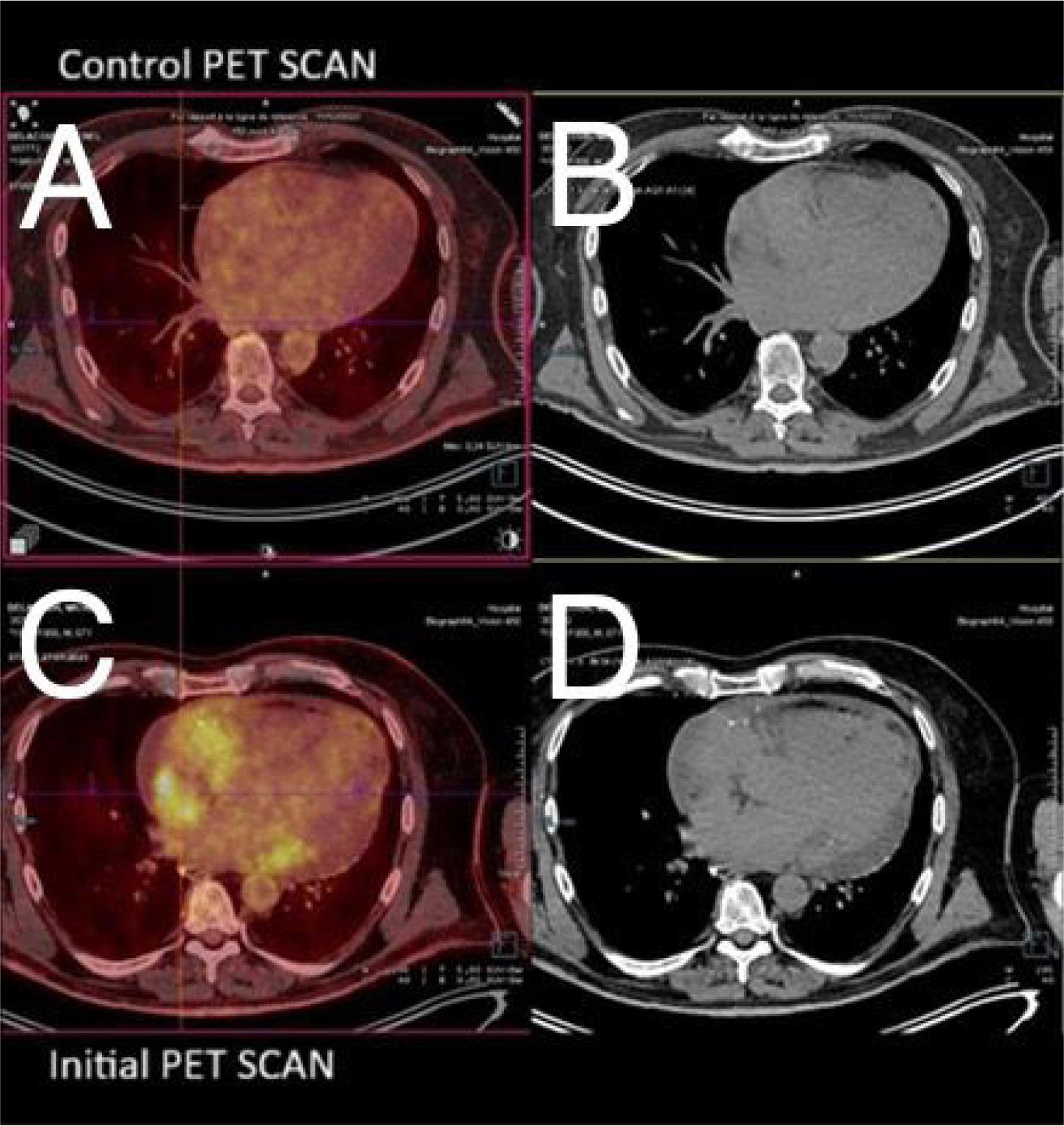
Control PET-CT Scan. A. Transverse PET/CT scan. Disappearance of pericardial hypermetabolism and pericardial effusion. Disappearance of pathological hyperfixation at the level of the right atrium. B. Transverse CT scans at the same level. C. Initial PET/CT scan. View Figure 2 for image details. D. Transverse CT scans at the same level.
A control CMR was performed, indicating a slightly impaired left ventricular systolic function due to the sequela of apical infarction and absence of left ventricular dilation. Also, significant improvement of pericardial effusion with persistence of a minimal residual layer was observed. Regarding the atrial mass, it maintained similar dimensions but with a significant regression of inflammatory and perfusion signs on T2 weighted sequences and on late gadolinium enhancement sequences, with absence of intramyocardial fibrosis (Figure 4).
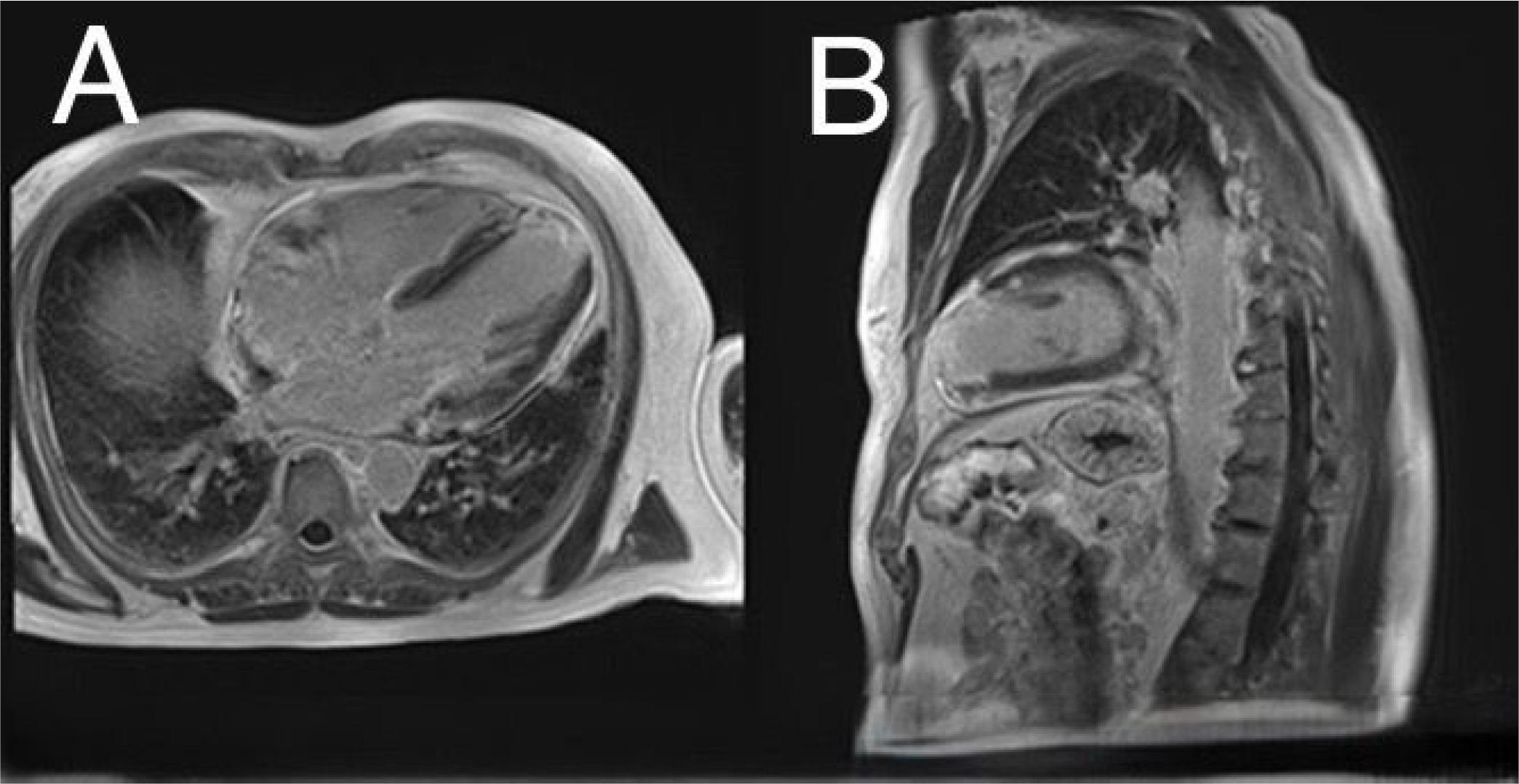
Control CMR – A, B – late gadolinium enhacement (LGE) sequences in four chambers and two chambers view showing significant improvement of pericardial effusion with persistence of a minimal residual layer. Persistence of a marked infiltration of the atrial mass of superimposable dimensions but with a very significant regression of inflammatory and perfusion signs on late enhancement sequences (black arrows). Apical scar post myocardial infarction (white arrow).
The physiopathology of ECD comprises a multiorgan infiltration of foamy CD68+CD1a histiocytes (resident mononucleate macrophages) and “Touton” cells (multinucleated giant cells) which can typically be found in lesions with increased lipid content such as xanthelasmas, xanthomas, and fat necrosis.[3] The group of disease commonly termed histiocytoses, are very rare with an annual incidence of less than five cases per million inhabitants and present in a heterogeneous fashion, ranging from isolated cutaneous manifestations to life-threatening neoplastic conditions. [4] The pathogenesis of ECD involves a mutation in the mitogen-activated protein kinase (MAPK) pathway. The BRAF mutation being the most frequent that occurs, in more than 50% of cases. The MAPK pathway is responsible for its role in cell growth, division, differentiation, survival, and response to stress. This classifies ECD as a neoplastic disorder among the various types of histiocytoses. To summarize, the genetic alterations in the MAPK pathway position ECD as a clonal hematopoietic disorder that expresses clinically due to the neoplastic histiocytic infiltrates.[5]Among the most common regions of involvement are the long bones, central nervous system, retroperitoneum, lung, skin, facial sinus, endocrine glands, and cardiovascular system. The metaphysis of the long bone is affected in 95% of patients with ECD by osteosclerosis. Another common site among the endocrine glands is the hypothalamuspituitary axis, central diabetes insipidus (CDI) representing the first clinical manifestation of the disease in such cases, averaging around 25% to 48% of all first clinical manifestations of ECD.[6] The goal in a newly diagnosed patient is to evaluate the extent of the disease involvement, the risk of end-organ failure and comprise a therapeutic and surveillance plan. Irrespective of the presenting cardinal symptom, a FDG PET-CT scan including the brain and distal extremities should be performed, accompanied by a cardiac and brain MRI with gadolinium. The FDG PET-CT scan serves the purpose of assessing organ involvement and choosing the best biopsy target. To explore all possible sites, a dedicated CT of the chest, abdomen, and pelvis is recommended to demonstrate pulmonary, periaortic, and perinephric infiltrates. CMR is a valuable diagnostic tool for assessing persistent pericardial fluid because it can provide information on the extent of the disease, as well as information on the surrounding structures and superior tissue characterization including an estimate of inflammation.[7] Laboratory testing should include a screening for any endocrinopathies, peripheral blood count abnormalities, evaluation of the renal and hepatic function, together with immune and inflammatory tests. Genetic testing for BRAF 600 gene should also be performed, since it guides the treatment. [8] Presence of the BRAF gene mutation is correlated with a more aggressive form of ECD that is associated with cardiac involvement in more than 70% of cases and cardiac complications such as pericarditis, cardiac tamponade, and conduction disorders but not with an increase in mortality.[9] The lesion distribution in this case is considered pathognomonic for ECD but differential diagnosis still has to be made with Langerhans cell histiocytosis, metabolic disorders, malignancy and sarcoidosis. Only the biopsy can provide definite diagnosis.[10] Concerning the treatment for patients that have a BRAF V600 mutant ECD with both cardiac and neurological lifethreatening involvement the first line therapy are BRAF-inhibitors such as vemurafenib and dabrafenib. The experience of the treating clinician, the clinical status of the patient as well as the toxicity profile should guide the decision of which of the two drugs should be used on a case-to-case basis. In patients without the mutation, an alternative therapy to BRAF inhibitors is cytotoxic therapy, balancing ECD symptom management and side effects of the treatments[11]
This case highlights a rare presentation of ECD with predominant cardiac involvement, diagnosed early through cardiac MRI before the development of compressive physiology. Despite the presence of a significant right atrial mass and persistent pericardial effusion, the patient exhibited no hemodynamic compromise—an uncommon finding in such extensive cardiac infiltration. The diagnosis was confirmed through multimodality imaging and histopathology, followed by targeted BRAF-inhibitor therapy, which led to substantial clinical and metabolic improvement over long-term follow-up. This case underscores the critical role of imaging in early recognition and the impact of molecular-targeted treatment in altering the disease course.