Ischemic heart disease is the leading cause of death and premature mortality, both globally and in Europe [1]. Long-term mortality data for ischemic heart disease and cardiovascular mortality from any cause are typically under-reported in Eastern Europe. Information on CVD morbidity and mortality in Eastern Europe is usually based on country-specific aggregate data rather than individual patient-level data [1, 2]. In terms of social and economic differences, there are important disparities between Eastern Europe and Western countries. Romania, one of the main countries in Eastern Europe, had 34% of its population at risk of poverty or social exclusion, the highest in the European Union (EU) and about 70% higher than Western Europe [3]. Romania ranked last in the European Union in terms of Gender-equality-index with a value of 57.5 compared to the European average of 71 points, reflecting also the lower access to health care for women [4]. Gender-related disparities can also be observed in the diagnosis, treatment, and prognosis of atherosclerotic cardiovascular disease, with significant gaps in short and long-term outcomes for women [5, 6]. These discrepancies emphasize the importance of identifying pathophysiologic and treatment variations for stable ischemic heart disease, as well as acute coronary syndromes with and without ST elevation. The current study aims to analyze and identify gender differences in long-term all-cause and cardiovascular-cause mortality in ischemic heart disease treated by PCI, with a focus on risk factors, comorbidities, and differences in therapeutic approach.
The details of the analyzed all-comers prospective PCI registry have been previously described [7, 8]. In summary, this registry has prospectively included all patients aged 18 years or older who underwent PCI at the Emergency Institute for Cardiovascular Diseases and Transplantation in Târgu Mureș. Enrollment began at hospital discharge in January 2016, and data have been systematically recorded in the institute’s local PCI Registry, accessible online at http://pci.cardio.ro/. The registry adheres to the guidelines of the Cardiology Audit and Registration Data Standards (CARDS), a framework developed by the Department of Health and Children, European Society of Cardiology, Irish Cardiac Society, and the European Commission [9]. The CARDS criteria emphasize a structured approach to data collection, encompassing anthropometric and demographic details, pertinent medical history, comorbidities, clinical status at admission, PCI indications, coronary artery segments affected and treated, use of invasive diagnostic and therapeutic devices, periprocedural complications, in-hospital medications, clinical progression, discharge status, discharge medications, and follow-up outcomes. Comprehensive data corresponding to these variables were gathered for each patient included in the registry. All patients listed in the registry as of December 2023 were included in the current analysis. Each participant (or their legal representative) provided informed consent for the PCI procedure and their inclusion in the study. Ethical approval for the study was granted by the institution’s ethics committee (decision number 8646, dated December 22, 2015). The study protocol aligns with the ethical principles outlined in the Declaration of Helsinki, ensuring the confidentiality of all patients’ data.
The primary clinical objective of this study was to evaluate the incidence of mortality from cardiovascular causes and all-cause mortality. Data on in-hospital mortality were sourced from the PCI registry, while mortality rates as of December 31, 2023, were provided by the Romanian National Health Insurance Service (NHIS) database. For deceased patients, the Regional Statistics Office of the Romanian National Institute of Statistics provided the date and cause of death, classified according to the International Classification of Diseases, 10th Revision (ICD-10). Deaths attributed to diseases of the circulatory system were classified as cardiovascular-related.
Analyses were conducted with a significance level of α = 0.05 and a 95% confidence interval (CI). Tests were two-sided. The Shapiro-Wilk test was used to check for normality in continuous variables. Those with a parametric distribution were presented as mean ± standard deviation and analyzed using the non-paired Student’s t-test. Non-parametric continuous variables and discrete variables were expressed as medians (interquartile ranges) and assessed using the Mann-Whitney U test. Categorical data were expressed as absolute and relative frequencies and compared using either the χ2 test or Fisher’s exact test, depending on suitability. The cumulative incidence of both all-cause and cardiovascular mortality was calculated through the Kaplan-Meier method, and differences were evaluated using the log-rank test. Predictors of survival were examined using Cox proportional hazard models. Univariable models assessed the relationship between survival and individual predictors, while multivariable models evaluated multiple predictors simultaneously, with results presented as hazard ratios (HR). To avoid overfitting, the number of predictors in multivariable models was limited to ensure at least 15 events per covariate. Strong correlations among independent variables were flagged if the variance inflation factor exceeded 2.5. Stepwise methods were employed to build multivariable models, with Akaike’s Information Criterion (AIC) used at each stage to evaluate potential overfitting. The final prediction models and their performance may be optimistic, as they were assessed using the same dataset. To address this, 95% confidence intervals (CIs) were calculated using bootstrapping with 1000 samples to ensure internal validation. All statistical computations were performed using R version 4.1.1 and R Studio version 1.4.17.
A total of 7,442 patients were included in the present study, with the male group comprising 5,208 patients (70%) and the female group comprising 2,234 patients (30%). The clinical characteristics of the included population are reported in Table 1. In-hospital mortality was observed in 256 patients (3.44%) overall: 116 (5.19%) in the female group and 140 (2.69%) in the male group (p = 0.005). Regarding long-term survival data, survival probability curves for all-cause mortality and cardiovascular mortality (Figures 1 and 2) revealed lower survival rates for females in both categories (p = 0.0317; p = 0.002). Survival probability curves for acute and chronic coronary syndromes are illustrated in Figures 1 and 2. Among patients presenting with acute coronary syndrome with ST-segment elevation (ACS-STE), survival probability was significantly lower for females for both all-cause and cardiovascular mortality (p<0.0001). In contrast, in patients with acute coronary syndromes without ST-segment elevation (ACS-NSTE), there were no differences in mortality between women and men. However, in patients with chronic coronary syndromes, male patients demonstrated a lower survival probability for all-cause mortality compared to women (p=0.024). Female patients were significantly older and had higher rates of stroke history, atrial fibrillation, chronic kidney disease, and valvular heart disease. However, they had lower rates of smoking, chronic obstructive pulmonary disease (COPD), chronic coronary syndrome, peripheral artery disease, chronic heart failure, and dilated cardiomyopathy compared to male patients. ACS-STE was more common in females than males (27.3% vs. 25.6%, p=0.04) and was associated with a worse condition at admission, as reflected by significantly higher GRACE scores (90.1 vs. 84.7, p<0.0001) and ACEF scores (1.38 vs. 1.33, p<0.0001) compared to males.
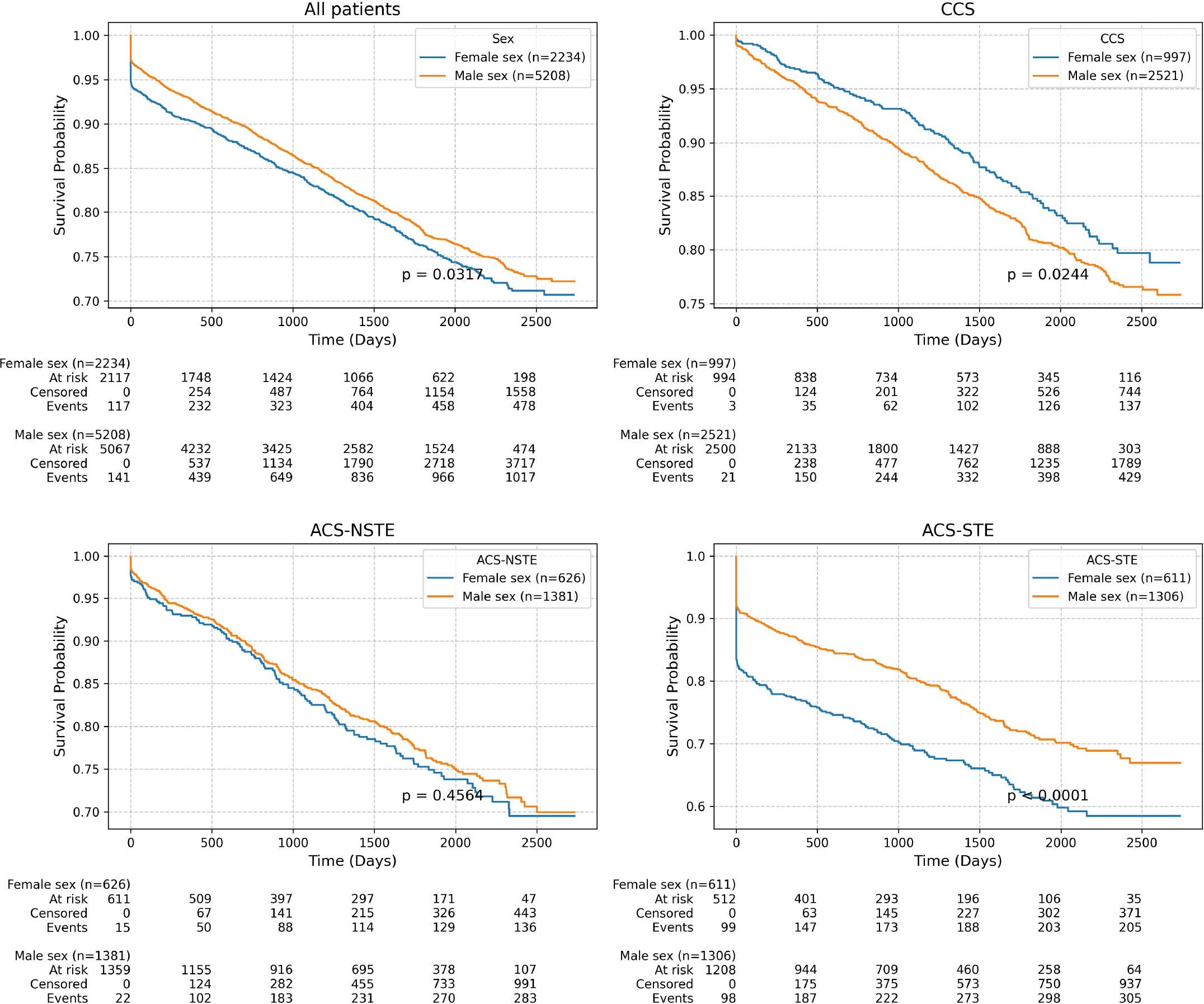
All-cause mortality among the studied population.
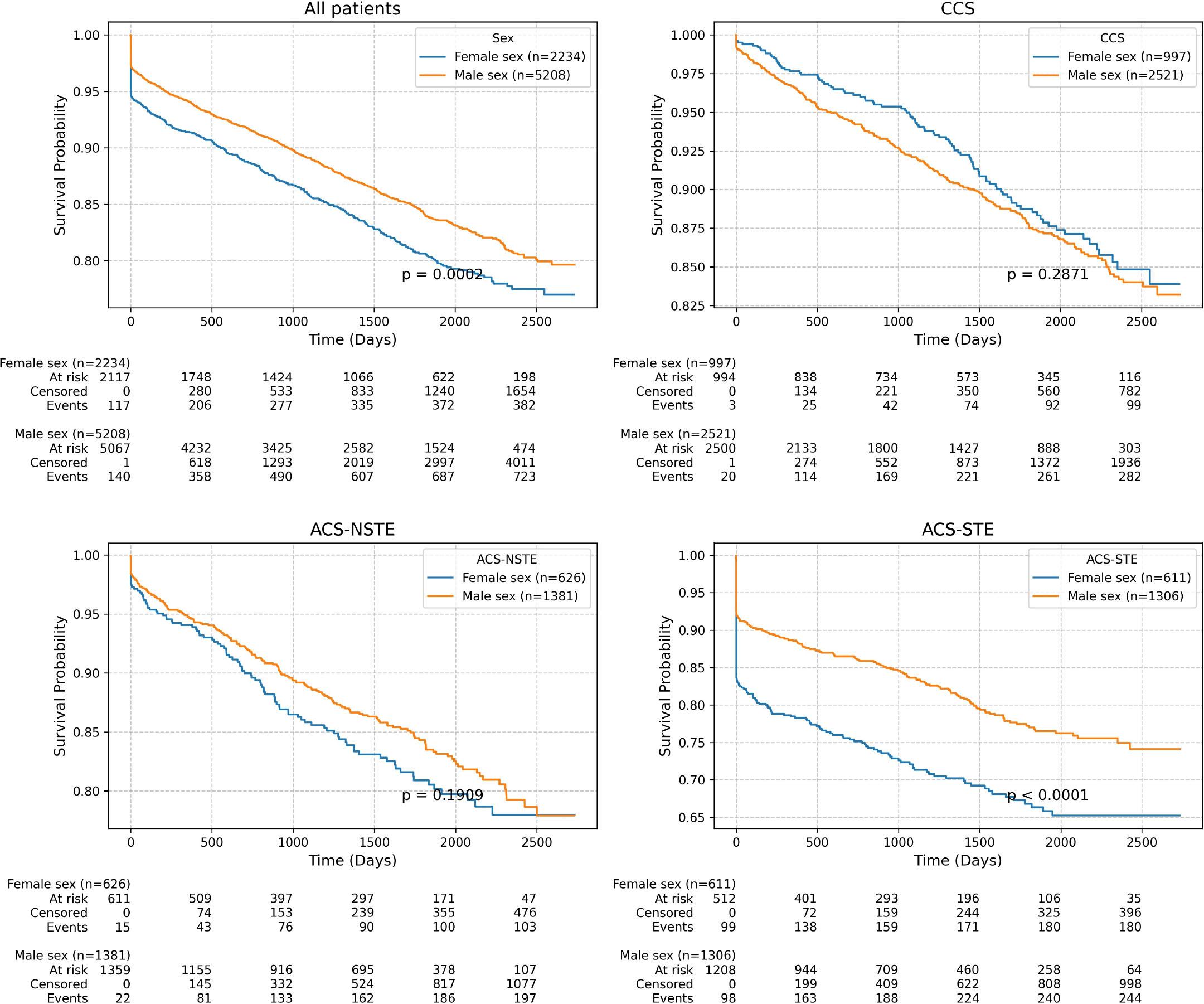
Cardiovascular-cause mortality among the studied population.
Demographic and clinical characteristics of the studied population.
| Parameter | All patients (n=7442) | Females (n=2234) | Males (n=5208) | p value |
|---|---|---|---|---|
| In hospital death | 256/7442 (3.44%) | 116/2234 (5.19%) | 140/5208 (2.69%) | 0,0005 |
| Age (years) | 65.04 (57.56 - 71.87) | 67.52 (60.75 - 74.22) | 64.03 (55.76 - 70.74) | <0.0001 |
| BMI (kg/m2) | 28.37 (25.81 - 31.44) | 28.36 (25.39 - 32.03) | 28.37 (25.95 - 31.24) | 0,54415 |
| Prior MI | 1574/7442 (21.15%) | 412/2234 (18.44%) | 1162/5208 (22.31%) | 0,0005 |
| Prior CABG | 201/7442 (2.7%) | 45/2234 (2.01%) | 156/5208 (3%) | 0,01799 |
| Prior PCI | 1902/7442 (25.56%) | 539/2234 (24.13%) | 1363/5208 (26.17%) | 0,07696 |
| Stroke/TIA | 515/7442 (6.92%) | 183/2234 (8.19%) | 332/5208 (6.37%) | 0,008 |
| Atrial fibrillation | 869/7442 (11.68%) | 293/2234 (13.12%) | 576/5208 (11.06%) | 0,0075 |
| Peripheral artery disease | 804/7442 (10.8%) | 200/2234 (8.95%) | 604/5208 (11.6%) | 0,0015 |
| Chronic kidney disease | 569/7442 (7.65%) | 204/2234 (9.13%) | 365/5208 (7.01%) | 0,001 |
| Valvular heart disease | 456/7442 (6.13%) | 173/2234 (7.74%) | 283/5208 (5.43%) | 0,0005 |
| Dilated cardiomyopathy | 628/7442 (8.44%) | 154/2234 (6.89%) | 474/5208 (9.1%) | 0,0015 |
| Chronic heart failure | 863/7442 (11.6%) | 210/2234 (9.4%) | 653/5208 (12.54%) | 0,0005 |
| COPD | 662/7442 (8.9%) | 165/2234 (7.39%) | 497/5208 (9.54%) | 0,0035 |
| Smoking status | 0 (0 - 1) | 0 (0 - 1) | 1 (0 - 1) | <0.0001 |
| Smokers | 3257/7442 (42.07%) | 596/2234 (26.68%) | 2661/5208 (51.09%) | <0.0001 |
| Diabets Mellitus | 1479/7442 (19.87%) | 460/2234 (20.59%) | 1019/5208 (19.57%) | 0,30535 |
| Hypertension | 3872/7442 (52.03%) | 1165/2234 (52.15%) | 2707/5208 (51.98%) | 0,90455 |
| Dyslipidemia | 3025/7442 (40.65%) | 871/2234 (38.99%) | 2154/5208 (41.36%) | 0,04998 |
| LVEF (%) | 50 (40 - 55) | 50 (40 - 55) | 50 (40 - 55) | <0.0001 |
| ICU stay | 346/7442 (4.65%) | 120/2234 (5.37%) | 226/5208 (4.34%) | 0,05497 |
| ACS-STE | 1917/7442 (25.76%) | 611/2234 (27.35%) | 1306/5208 (25.08%) | 0,04498 |
| ACS-NSTEMI | 621/7442 (8.34%) | 196/2234 (8.77%) | 425/5208 (8.16%) | 0,3958 |
| ACS-UA | 1386/7442 (18.62%) | 430/2234 (19.25%) | 956/5208 (18.36%) | 0,3903 |
| CCS | 3518/7442 (47.27%) | 997/2234 (44.63%) | 2521/5208 (48.41%) | 0,003 |
| Time from symptom onset to hospitalization (hours) | 9.69 (5.26 - 21.39) | 10.6 (5.78 - 24.02) | 9.07 (5.13 - 20.68) | 0,03767 |
| Time from hospitalization to PCI (hours) | 24.93 (3.1 - 56.7) | 24.52 (2.51 - 51.14) | 25.08 (3.27 - 62.27) | 0,01517 |
| Time from symptom onset to PCI hours | 17.06 (6.42 - 54.68) | 19.29 (7.8 - 60.31) | 16.16 (6.04 - 47.22) | 0,02729 |
| RCA angioplasty | 0 (0 - 1) | 0 (0 - 1) | 0 (0 - 1) | 0,75875 |
| LAD angioplasty | 0 (0 - 1) | 0 (0 - 1) | 0 (0 - 1) | 0,2436 |
| CXA angioplasty | 0 (0 - 1) | 0 (0 - 0) | 0 (0 - 1) | 0,00241 |
| LM angioplasty | 401/7126 (5.63%) | 130/2146 (6.06%) | 271/4980 (5.44%) | 0,33583 |
| Number of diseased coronary | 2 (1 - 3) | 2 (1 - 3) | 2 (1 - 3) | 0,07018 |
| Proximal segment angioplasty | 3198/7126 (44.88%) | 970/2146 (45.2%) | 2228/4980 (44.74%) | 0,73663 |
| Segments treated | 1 (1 - 2) | 1 (1 - 2) | 1 (1 - 2) | 0,14471 |
| TIMI pre PCI | 2 (0 - 3) | 2 (0 - 3) | 2 (0 - 3) | 0,49294 |
| TIMI post PCI | 3 (3 - 3) | 3 (3 - 3) | 3 (3 - 3) | 0,89929 |
| Complete revascularization | 2532/6950 (36.43%) | 800/2084 (38.39%) | 1732/4866 (35.59%) | 0,03298 |
| Radial vascular access | 5669/7442 (76.18%) | 1641/2234 (73.46%) | 4028/5208 (77.34%) | 0,0015 |
| Vascular closure device | 477/7442 (6.41%) | 178/2234 (7.97%) | 299/5208 (5.74%) | 0,001 |
| Cardiogenic shock | 171/7442 (2.3%) | 72/2234 (3.22%) | 99/5208 (1.9%) | 0,0015 |
| CPR during hospitalization | 183/7442 (2.46%) | 82/2234 (3.67%) | 101/5208 (1.94%) | 0,0005 |
| Hospitalization days | 5.3 (3.8 - 7.2) | 5.5 (3.8 - 7.5) | 5.2 (3.8 - 7.1) | 0,08437 |
| CK MB U L Max | 45 (23 - 165) | 52 (25 - 171.25) | 43 (23 - 161) | 0,00979 |
| Total cholesterol (mg/dL) | 166 (136 - 203) | 174 (144 - 211) | 163 (134 - 200) | <0.0001 |
| HDL cholesterol (mg/dL) | 36 (30 - 42) | 39 (32 - 45.62) | 34 (29 - 41) | <0.0001 |
| LDL cholesterol (mg/dL) | 97 (74 - 129) | 102 (79 - 136) | 96 (71 - 126) | <0.0001 |
| Triglyceride (mg/dL) | 127 (93 - 182) | 128 (95 - 181) | 127 (91.68 - 183) | 0,80268 |
| Creatinine (mg/dL) | 0.91 (0.79 - 1.1) | 0.82 (0.73 - 1.02) | 0.94 (0.83 - 1.11) | <0.0001 |
| CrCl (ml/min) | 77.04 (58.23 - 93.36) | 68.67 (45.44 - 81.32) | 82.97 (63.28 - 97.71) | <0.0001 |
| Hemoglobin (g/dL) | 13.5 (12.4 - 14.57) | 12.5 (11.45 - 13.4) | 14 (12.9 - 14.85) | <0.0001 |
| Platelets (×103/μL) | 224 (187 - 267.3) | 238 (196 - 281) | 219 (183 - 260) | <0.0001 |
| WBC (×103/μL) | 8.14 (6.73 - 9.94) | 8.04 (6.58 - 9.91) | 8.18 (6.79 - 9.95) | 0,01384 |
| LDH (U/L) | 204 (164 - 355.81) | 218 (174 - 379.16) | 200 (161 - 344.55) | <0.0001 |
| PCR (mg/dL) | 1.69 (0.39 - 8.1) | 1.72 (0.44 - 6.88) | 1.68 (0.37 - 8.64) | 0,95628 |
| Tronopine (ng/L) | 0.94 (0.05 - 92.9) | 2 (0.05 - 132.75) | 0.63 (0.05 - 58.83) | 0,28548 |
| NTproBNP (pg/ml) | 963 (212 - 2897) | 1100 (327 - 3615) | 876.5 (181.25 - 2574) | 0,04279 |
| SYNTAX score | 16 (9 - 25) | 16 (9 - 25.5) | 16 (9 - 24.5) | 0,70737 |
| ACEF score | 1.35 (1.13 - 1.68) | 1.38 (1.16 - 1.72) | 1.33 (1.12 - 1.66) | <0.0001 |
| GRACE score | 86.41 (72.97 - 101.09) | 90.18 (78.1 - 105.6) | 84.72 (70.65 - 98.9) | <0.0001 |
Values are presented as absolute frequencies (%) in the case of binary variables. Non-parametric continuous variables and discrete variables were expressed as median (interquartile range). ACEF – age, creatinine, ejection fraction score; ACS – acute coronary syndrome; BMI – body mass index; CABG – coronary artery bypass graft; CCS – chronic coronary syndrome; CrCl – creatinine clearance; COPD – chronic obstructive pulmonary disease; CPR – cardiopulmonary resuscitation; CXA – circumflex artery; GRACE - Global Registry of Acute Coronary Events; HDL – high density lipoprotein; HR – hazard ratio; ICU – intensive care unit; LAD – left anterior descending artery; LDL – low density lipoprotein; LM – left main artery; LVEF – left ventricular ejection fraction; MI – myocardial infarction; PCI – percutaneous coronary intervention; RCA – right coronary artery; TIMI – Thrombolysis In Myocardial Infarction; UA – unstable angina; WBC – white blood cells.
Additionally, time from symptom onset to hospitalization, time to PCI, and time from hospitalization to PCI were significantly longer for females ([10.6h vs. 9h, p=0.037], [24.5h vs. 25h, p=0.01], [17h vs. 16.1h, p=0.0002]) in case of ACS. In terms of laboratory data, female patients presented with higher levels of cardiac biomarkers, including CK-MB (55 vs. 43 U/L, p = 0.009), Troponin (2 vs. 0.63 ng/L, p = 0.02), and NT-proBNP (1100 vs. 876 ng/mL, p = 0.042). They also exhibited higher levels of non-specific inflammatory markers, such as fibrinogen (375 vs. 356 mg/dl, p=0.46), LDH (214 vs. 197 U/L, p<0.0001), and platelet count (240 vs. 221 × 103/μL, p<0.0001), alongside lower levels of hemoglobin (12.5 vs. 14 g/dl, p<0.0001). Furthermore, women had significantly higher levels of total cholesterol (173 vs. 161 mg/dL, p < 0.0001) and LDL-cholesterol (103 vs. 96 mg/dL, p < 0.0001) compared to male patients.
They also exhibited significantly higher rates of cardiogenic shock (3.22% vs. 1.9%, p = 0.001), cardiopulmonary resuscitation (CPR) during hospitalization (3.67% vs. 1.94%, p = 0.0005), and in-hospital mortality (5.19% vs. 2.7%, p = 0.0005) compared to men. Despite similar complexity of coronary artery disease (CAD) between groups, reflected by similar SYNTAX scores, radial vascular access was used less frequently in the female group. PCI procedure complexity (complete revascularization rates, segments treated, and number of implanted stents) and TIMI flow before and after PCI did not differ between groups.
Prescription rates for guideline-recommended treatments included 1,919 patients (29.21%) who received ticagrelor as a potent P2Y12 receptor inhibitor and 4,829 patients (73.24%) who received clopidogrel. High prescription rates were observed for other treatment classes: aspirin (94.55% of patients), statins (95.52%), beta-blockers (83.93%), and angiotensin-converting enzyme inhibitors or angiotensin receptor blockers (82.9%). Lower prescription rates were observed for other therapeutic classes, including SGLT2 inhibitors (1.92%), NOACs (6.02%), VKAs (8.56%), and fibrates (6.08%). No differences were observed between groups in the prescription of beta-blockers, aldosterone antagonists, direct oral anticoagulants (DOACs), or sodium-glucose cotransporter 2 (SGLT2) inhibitors. In contrast, in the female group, prescription rates were significantly lower for ticagrelor (22.53% vs. 32.1%, p=0.0005), aspirin (93.7% vs. 96.3%, p=0.0005), statins (93.7% vs. 96.3%, p=0.0005), angiotensin-converting enzyme inhibitors (65.4% vs. 71.3%, p=0.0005), and fibrates (5.05% vs. 6.5%, p=0.02), despite presenting more frequently with ACS-STE and having higher LDL-cholesterol levels compared to male patients.
Multivariate Cox-proportional hazard regression models excluded an independent effect of gender on all-cause and cardiovascular-cause mortality, either after PCI for acute or chronic coronary syndromes (Figure 3). Although most independent determinants of mortality were common in both male and female patients (such as advanced age, low left ventricular ejection fraction, high creatinine levels, or the need for mechanical ventilation), there were also some notable differences (Figure 4). Accordingly, beta-blocker and ticagrelor prescriptions were independent predictors of all-cause mortality only in males, while lymphocyte count and the TIMI-flow observed post-PCI were independent determinants only in females. Regarding cardiovascular mortality, elevated heart rate and a higher number of diseased coronary arteries were independent predictors only in males. At the same time, the presence of COPD was an independent predictor only in females. Of note, the absence of betablocker and ticagrelor prescription was an independent predictor of cardiovascular-cause mortality in both sexes.
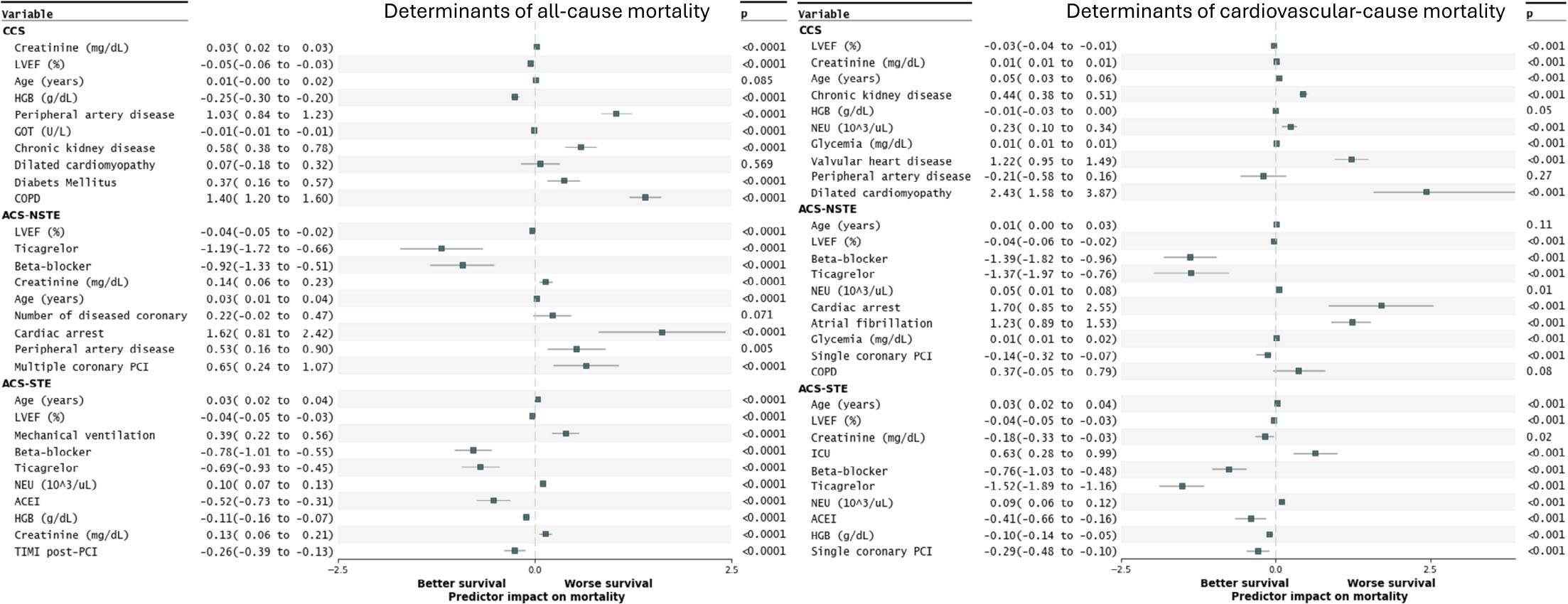
Predictors of survival in multivariate Cox proportional hazards models in different clinical forms of ischemic heart disease.
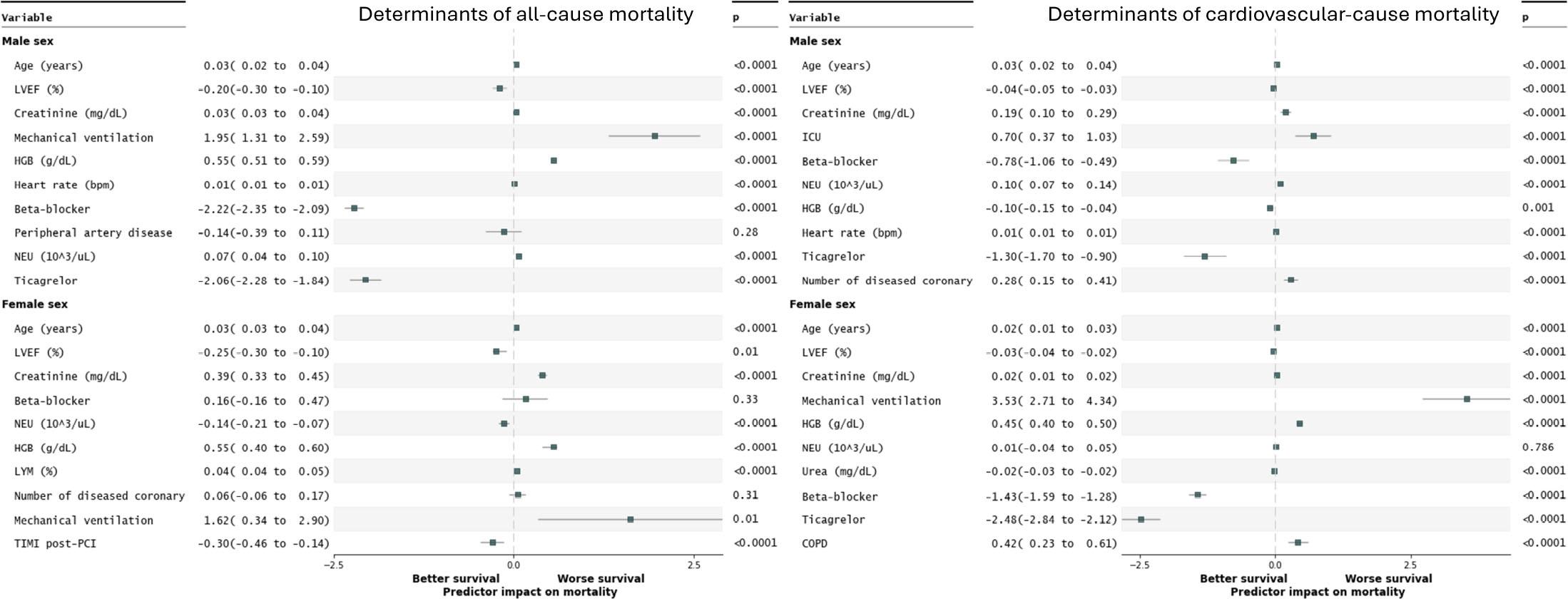
Predictors of survival in multivariate Cox proportional hazards models in males and females.
The findings of our study can be summarized as follows: (1) High long-term cardiovascular and all-cause mortality rates were observed for the entire included population. Women demonstrated lower survival probabilities for all-cause and cardiovascular mortality in ACS-STE, while no differences were observed in ACS-NSTE, and male patients had worse survival probabilities for all-cause mortality in chronic coronary syndromes. However, these gender-related differences in mortality were attributed to the distinct clinical characteristics of the male and female populations. Accordingly, gender per se was not associated independently with higher all cause or cardiovascular cause mortality. (2) Female patients were older and exhibited higher rates of stroke, a trial fibrillation, chronic kidney disease, and a lower rate of smoking, COPD, heart failure, a previous coronary disease, and peripheral artery disease compared to male patients. (3) Women presented more frequently with ACS-STE, experienced longer delays in treatment, and had worse conditions at admission compared to males, as reflected by higher GRACE and ACEF scores. (4) Female patients faced significantly higher rates of complications during hospitalization, including cardiogenic shock, need for CPR, and in-hospital mortality. (5) Female patients were less likely to receive guideline-recommended therapies, such as ticagrelor, aspirin, statins, and angiotensinconverting enzyme inhibitors, despite presenting with more severe disease. (6) Radial vascular access during PCI was less frequently used in females, although PCI procedure complexity and TIMI flow outcomes were comparable between sexes. (7) Most of the independent determinants of mortality were common in males and females. However, some of the comorbidities had different impacts: COPD was associated with worse cardiovascular-cause outcomes in females, while multivessel disease was especially associated with worse outcomes in males.
The highest mortality was observed in our study when compared with contemporary PCI cohorts [1]. A Western PCI population reported 10% mortality over a mean follow-up of 4.1 years [10], while in our registry, a 22% mortality was observed at 4 years. Overall, 5-year allcause mortality in the CCS subgroup in our study was 19.3% (Figure 1), which is higher than the 5-year all-cause mortality of 14.2% in the ISCHEMIA trial and the 5-year all-cause mortality of 11.2% in the SYNTAX study [11, 12]. Although Romania is a European region with a very high risk for cardiovascular mortality [13], this low survival rate, even with guideline-recommended treatment strategies, including myocardial revascularization, is especially disappointing. It suggests that clinical follow-up and secondary prevention strategies might need improvement in our country.
Consistent with international registry data [14–16]. women were affected by CAD in lower proportions compared to men. However, the women included in our study were older and had more non-cardiac comorbidities, findings that align with results from the Holtzman study [17] and a systematic review and meta-analysis conducted in 2018, which included 1,032,828 patients (258,713 women) across 49 studies examining PCI outcomes and sex-based differences [18]. In contrast, male patients were younger and had a higher prevalence of smoking, cardiac conditions, and atherosclerotic disease, consistent with known demographic and pathophysiological variations between sexes in CAD presentation [16, 19–21].
Our study revealed significant sex-based differences depending on the form of presentation. The proportion of females was higher among patients with ACS-STE (27.3% vs. 25.6%, p=0.04), similar to findings from Berger et al.’s meta-analysis of 136,247 patients with STEMI [22] or from the Vienna STEMI registry [23]. Conversely, males predominated in the chronic coronary syndrome category, consistent with findings from Ferrari et al.’s study [24]. For our population, the symptom onset-to-treatment time, door-to-balloon time, and reperfusion time were longer for female patients undergoing PCI compared to male patients. This phenomenon, also noted by Guo Y. in their systematic review [18] on gender differences in PCI outcomes, is attributed to sex-based variations in CAD pathophysiology, a higher frequency of atypical symptoms in women, and the tendency of medical professionals to associate CAD with male patients, as highlighted by Leonard et al. [20]. At hospitalization, women presented with higher levels of myocardial necrosis markers, inflammatory markers, and procoagulant status, as reflected by higher GRACE and ACEF scores. These differences, as explained by Rao et al. in their meta-analysis [25], are attributed to anatomical and physiological variations, as well as longer delays from symptom onset to hospitalization and PCI in women.
In terms of severity of coronary artery disease, lesion- and procedure complexity, our study did not show a significant difference between the two sexes, correlated with similar SYNTAX score for the two groups of patients, contrary to Taqueti R.’s review that showed a more severe coronary disease in male patients [26]. Furthermore, the radial approach for PCI was less frequently used in women, consistent with findings from Savage et al., who reported higher rates of initial radial artery access failure in women (11.3% vs. 3.1%; p < 0.001) [27]. These differences were attributed to anatomical particularities, hemodynamic instability, and the need for additional procedures such as temporary pacing or mechanical circulatory support [27–29]. These disparities may explain the higher rates of complications among women, including cardiogenic shock (3.22% vs. 1.9%, p=0.001), the need for resuscitation, and increased inhospital mortality. Guo et al.’s meta-analysis[18] similarly found that male patients had a lower incidence of cardiogenic shock (OR 0.78, 95% CI 0.65–0.92, p <.001) and in-hospital mortality (OR 0.58, 95% CI 0.52–0.63, P < .001, I2 = 66%),%), aligning with our results.
Although our study did not identify differences in PCI procedure complexity or coronary disease severity between sexes, significant disparities were observed in medication prescriptions. Women were less likely to receive guideline-recommended therapies despite presenting with more severe conditions, a phenomenon frequently noted in prior studies [20, 30–33]. This trend reflects a tendency among physicians to avoid more aggressive treatment in critically ill patients, despite the potential benefits. The observed differences in presentation and treatment strategies are reflected in survival outcomes. Women had higher in-hospital mortality regardless of whether they presented with acute or chronic coronary syndrome. In line with previous findings [34] this could be explained by the different demographic and clinical characteristics of the two sexes. In the long term, women with ACS-STE exhibited higher mortality from both cardiovascular and all-cause events. However, there were no sex differences in survival among patients with ACS-NSTE, while men with CCS had a higher all-cause mortality rate than women. Notably, cardiovascular-cause mortality after PCI for stable CAD was not different in males and females. Although gender was not an independent determinant of all-cause or cardiovascular cause mortality, these findings emphasize the importance of addressing sex-based disparities in CAD management.
The main limitation of our study is that it reflects findings from a single center. However, we analyzed data from a relatively large number of patients, with geographical origin from the whole territory of Romania [35]. Another potential limitation is that unmeasured confounders, like detailed socioeconomic factors, might influence the analyzed outcomes. While internally validated, the multivariable prediction models could benefit from external validation. Accordingly, future research in larger, nationwide registries is essential to confirm these findings and drive systemic improvements.
The observed high long-term mortality after PCI suggests that follow-up and secondary prevention strategies might need improvement in Romanian patients suffering from CAD. Although gender per se was not an independent determinant of survival, we identified significant gender-related differences in clinical characteristics and treatment patterns of CAD. Efforts should prioritize reducing delays in care, ensuring equitable access to evidence-based therapies, and tailoring interventions to the specific needs of male and female patients. Our findings should be confirmed in larger, nationwide registries.