Takotsubo syndrome is an acute cardiac syndrome characterized by typical regional wall motion abnormalities that reflect impairment of myocardial contractility, leading to acute heart failure in the absence of culprit epicardial coronary artery disease. [1] Since its first description in Japan in 1990, Takotsubo syndrome (stress cardiomyopathy/broken heart syndrome/apical ballooning syndrome) has emerged as an important form of acute reversible myocardial injury.[2] The European Society of Cardiology position statement from the task force on Takotsubo syndrome subclassifies this pathological entity into primary and secondary Takotsubo syndrome. In the primary form, the acute cardiac symptoms are the primary reason for seeking care, usually from emergency medical services, acute cardiac services, or the primary care physician, whereas the secondary form occurs in patients already hospitalized for another medical, surgical, anesthetic, obstetric, or psychiatric condition.[1] Complications, such as acute heart failure, left ventricular outflow tract obstruction (in the apical anatomical variant), and mitral regurgitation, leading to cardiogenic shock are not infrequent, and although this state is reversible, the syndrome is also recurrent, with an average recurrence rate reported as 2% to 4% per year.[2,3]
A 46-year-old female patient with no significant past medical history, admitted for hepatectomy, being a living hepatic transplant donor for her sister, is evaluated by the cardiology team in the ICU on the postoperative second day for sudden aggravated dyspnea. Her cardiologic preoperative evaluation and echocardiography were normal. Clinically, she was normotensive (BP 130/80 mmHg), with no need for hemodynamic support, tachycardic with a heart rate of 110 bpm, polypneic, dyspneic, with rales on both pulmonary areas. Blood workup: high hs-cTnI levels (2900-->2500 ug/l), NT proBNP 3900 pg/ml. ECG shows sinus tachycardia and new T wave inversion DI, aVL, V4-V6, with a QTc prolongation (QTc 516 ms) (Figure 1). Transthoracic point of care echocardiography (POCUS) (Figure 2): severe hypokinesia of 2/3 apical segments of anterior, inferior, and lateral left ventricular walls (“apical ballooning” aspect of the left ventricle), severe LV systolic dysfunction, LVEF 30%, no signs of left ventricle outflow tract obstruction (LVOTO), grade II mitral regurgitation, normal pericardium, normal size atria, and right ventricle, no intracardiac masses in transthoracic evaluation, normal right ventricle longitudinal function, bilateral lung B lines. The differential diagnosis of this acute LV systolic dysfunction in the postoperative period is mainly between acute myocardial infarction (with or without obstructive coronary arteries), Takotsubo syndrome, and myocarditis. The coronary angiography was postponed at this stage giving the high infectious risk in a post-hepatectomy patient and the lack of patient’s consent in the early postoperative days. The clinical context, the postoperative state (being a hepatic donor for her sister), the dysfunctional myocardium extending beyond the territory of a single coronary artery, the absence of cardiovascular risk factors, and the InterTAK score of 80 points, are all arguments for the high likelihood of Takotsubo syndrome in comparison with acute coronary syndrome. Initially the evolution was worsening, with low cardiac output and cardiogenic shock phenomena, with the need for vasopressor and inotropic support in medium doses, with endotracheal reintubation and invasive ventilation for 72 hours, and with parallel administration of loop diuretic treatment and mineralocorticoid receptor antagonist + low molecular weight heparin for venous thromboembolism prophylaxis. After weaning from inotropic and vasopressor support, treatment with iSGLT2 and If channel blocker was associated.
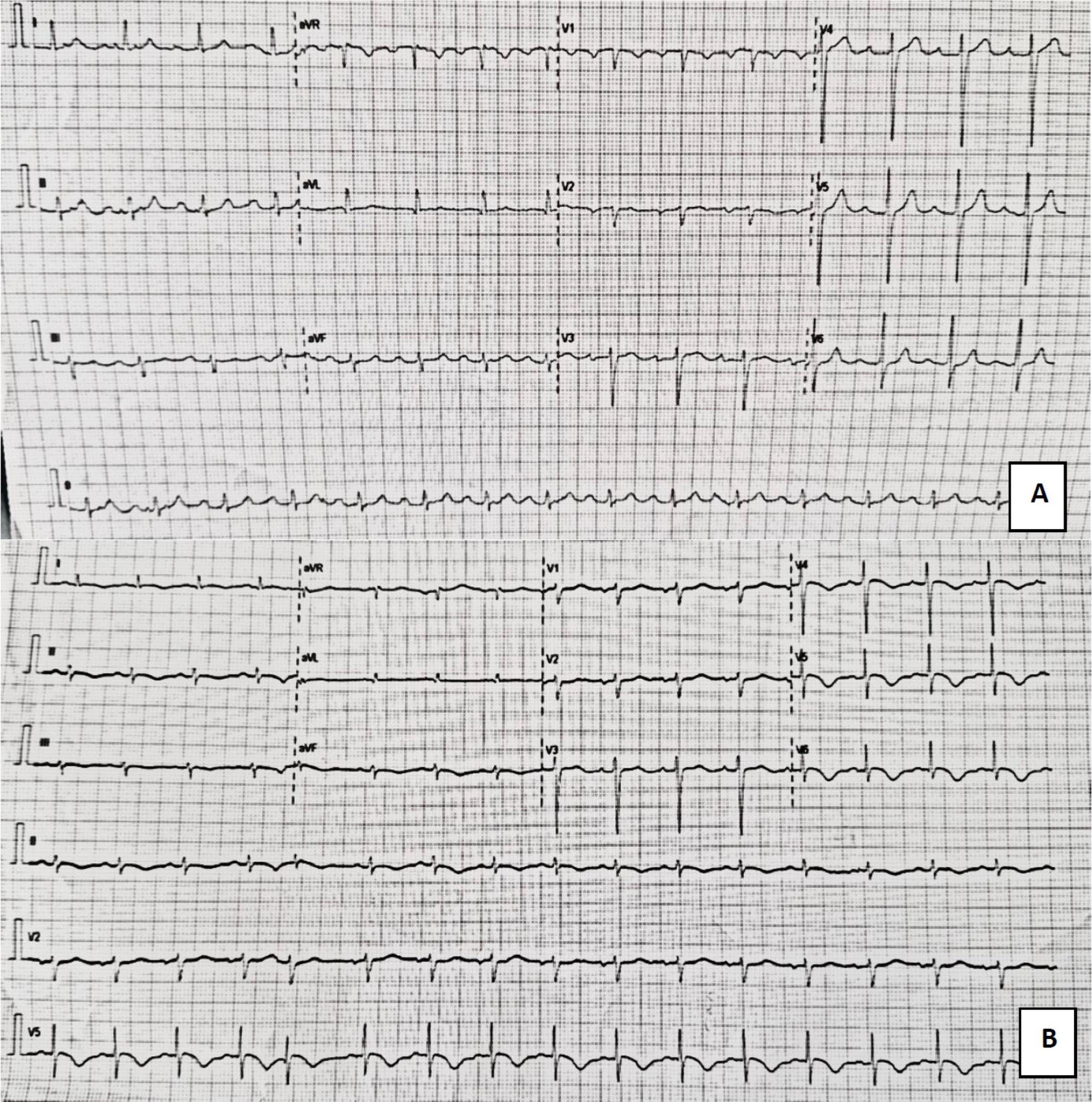
Normal preoperative ECG; B. Day 2 postoperative ECG findings showing sinus tachycardia 105 bpm, new T wave inversion DI, aVL, V4-V6, QTc prolongation (QTc 516 ms)
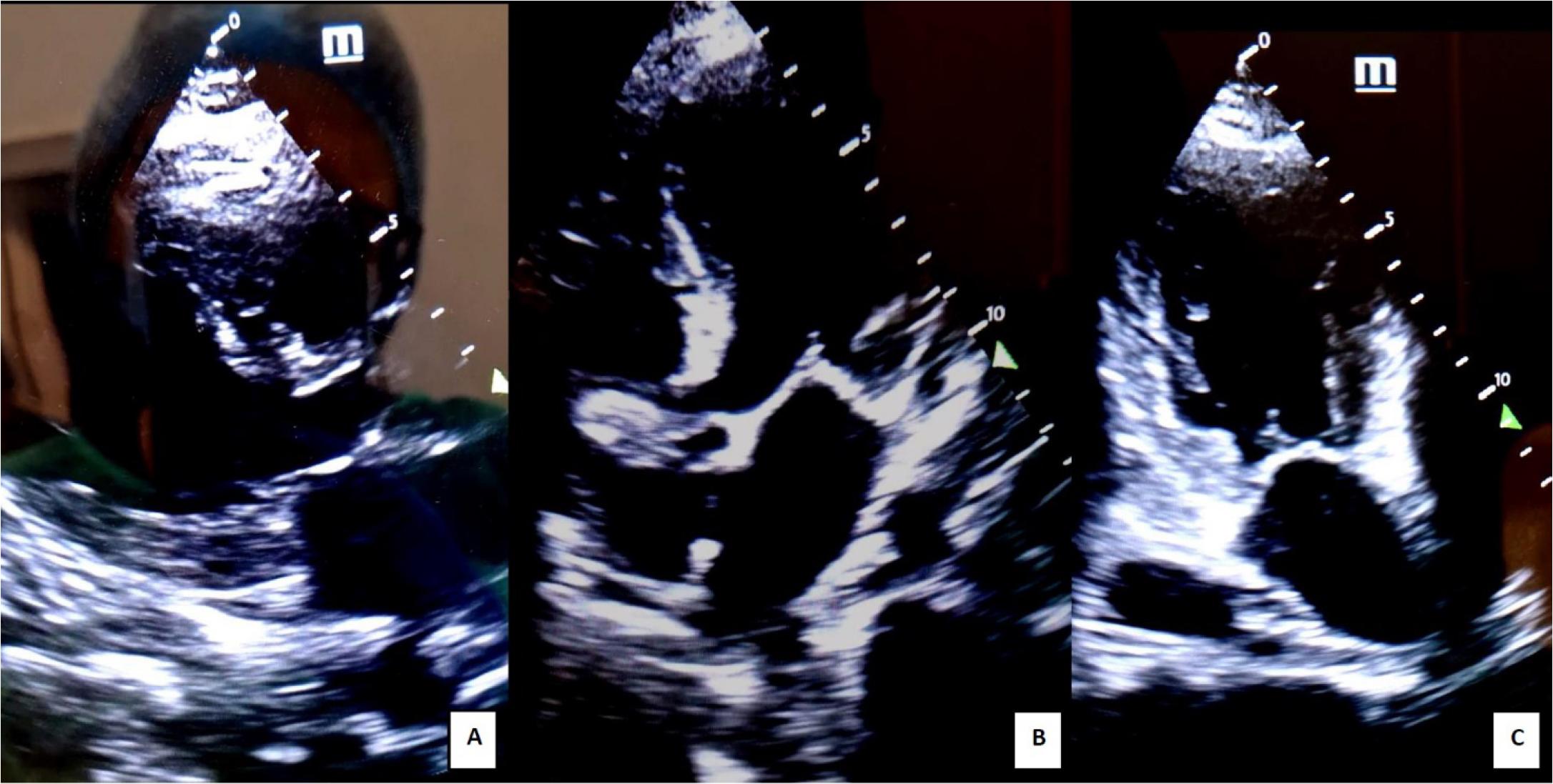
Transthoracic POCUS echocardiography 2nd day post-op: A. parasternal long axis view; B. apical four chamber view; C. apical 2 chamber view. Severe hypokinesia of 2/3 apical segments of anterior, inferior, and lateral walls (“apical ballooning” aspect of the left ventricle - dysfunctional myocardium extending beyond the territory of a single coronary artery); severe LV systolic dysfunction
Subsequently, the evolution was favorable; she was extubated, with a decreasing need for oxygen therapy, without dyspnea or chest pain, being evaluated through daily follow-up transthoracic echocardiography. The transthoracic echocardiography on the 9th postoperative day showed improved LV systolic function, with an LVEF of 46%, minimal hypokinesia of apical LV walls, normal LV filling pressures, stroke volume of 87.5 ml, cardiac output of 7l/ min, and normal RV systolic function (Figure 3). The persistence of T wave inversion is noted on the ECG. The transient character of regional left ventricle wall motion abnormalities is another argument for the high probability of Takotsubo syndrome. She was discharged in a good clinical state and redirected to perform a cardiac magnetic resonance.
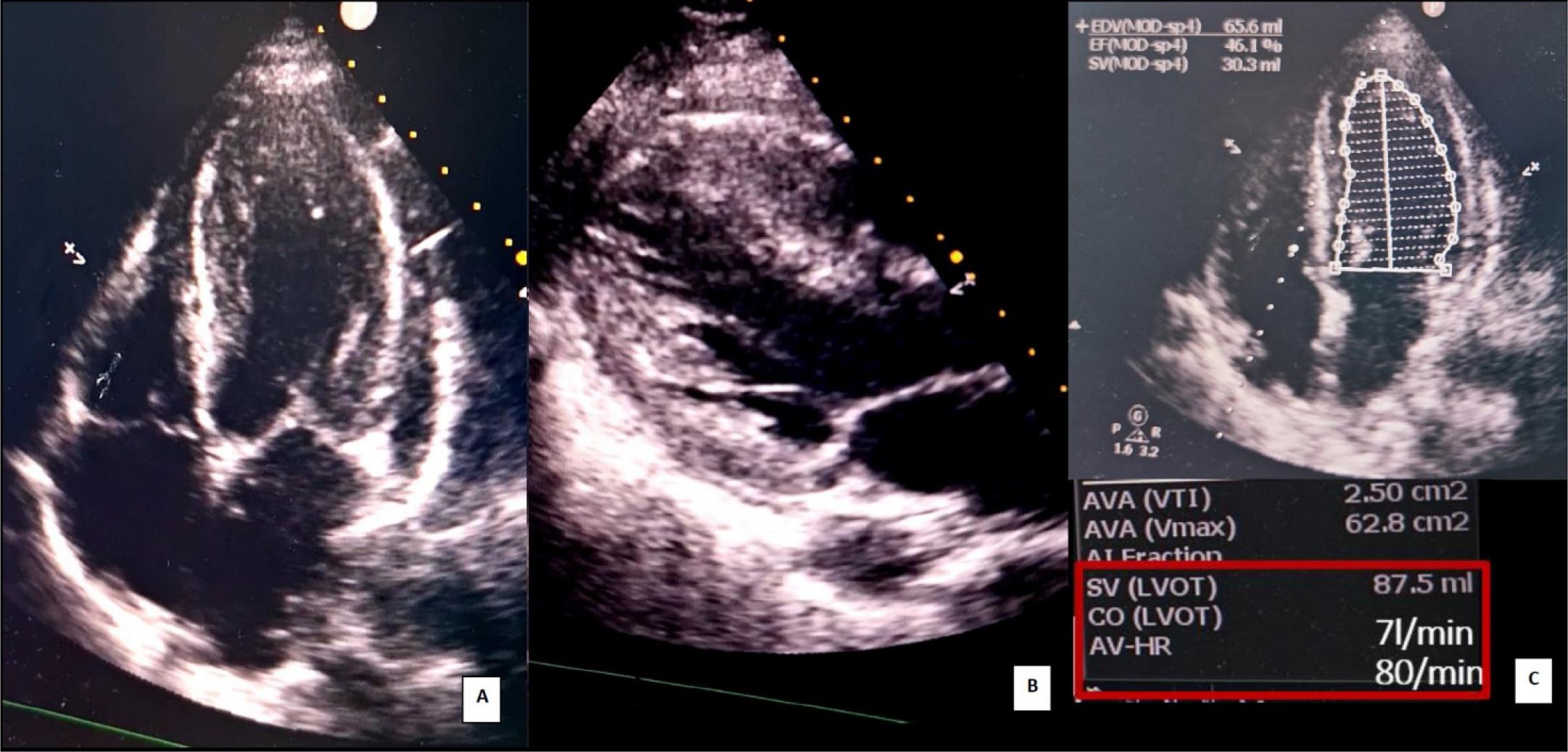
Transthoracic POCUS echocardiography 9th day post-op: A. apical four chamber view; B. parasternal long axis view; improved left ventricular systolic function, minimal hypokinesia of apical left ventricle walls; C. Left ventricular ejection fraction (LVEF) 46% (Simpson monoplane); Stroke volume (SV) and cardiac output (CO) determination using the continuity equation
Perioperative myocardial injury is defined as an acute cardiomyocyte injury (post-operative high-sensitivity cardiac troponin release) with or without accompanying symptoms, ECG changes, or imaging evidence of acute myocardial ischemia. It is reliably and rapidly detected using hs-cTn T/I measurements before and serially after surgery. To identify the underlying pathophysiology and define causal therapy, it is of major importance to perform systematic work up and early differentiation of primarily non-cardiac causes (e.g. severe sepsis, pulmonary embolism) versus the different cardiac causes (type 1/2 myocardial infarct, tachyarrhythmia, and acute heart failure). Increased awareness in the non-operative setting has led to a substantial increase in the detection of Takotsubo syndrome, and the use of transthoracic echocardiography in the work-up of peri-operative myocardial injury is strongly encouraged. Anecdotal evidence suggests that it is likely that the incidence is also higher than expected in the perioperative setting.[4]
The only lifesaving and definitive treatment for end-stage liver disease, acute liver failure, some metabolic diseases, and some liver tumors is currently liver transplantation. Despite remarkable recipient outcomes, a significant number of people die on the waiting list. One strategy used to counter-balance organ shortage has been the utilization of living donor liver transplantation, which is the only option in a region with insufficient deceased donor support. Studies consistently demonstrate that this strategy is equivalent to deceased donor liver transplantation in terms of patient and allograft survival.[5]Figure 1 A. Normal preoperative ECG; B. Day 2 postoperative ECG findings showing sinus tachycardia 105 bpm, new T wave inversion DI, aVL, V4-V6, QTc prolongation (QTc 516 ms)
Solid organ transplant is at the top of the conditions in which physical and emotional stress is at the highest level, and an increasing number of articles have reported on secondary Takotsubo syndrome occurring in recipients during or after solid organ transplantation (0.3% of recipients).[6,7] Liver transplantation surgery itself provokes a significant amount of stress on the cardiovascular system due to hemodynamic instability, massive blood transfusions to compensate for the intraoperative blood loss, major vascular manipulations such as vessel clamping, and the long procedure time. Liver transplant recipients are particularly vulnerable to developing Takotsubo syndrome due to the impaired stress response in the inflammatory milieu of hepatic cirrhosis, in addition to the perioperative period stress.[7,8]
Living donor hepatectomy is a major operation, with the potential for morbidity in an otherwise healthy donor. All donor candidates should be well informed not only about the details of the early and late risks of living liver donation but also about possible outcomes for the recipient. Detailed physical evaluation and preoperative psychosocial evaluation is mandatory.[8] Most (95%) of the complications following donor hepatectomy are minor, lending support to the continued practice of live liver donation in the context of rigorous donor screening and transparent patient counselling. The remaining 5% represent major postoperative complications, graded Clavien III or higher, such as respiratory complications (pneumonitis, empyema, pleural effusion, pneumothorax, pulmonary collapse), bleeding, gastrointestinal complications (gastritis, gastroparesis, obstruction, peritonitis, pancreatitis), infectious, hepatobiliary complications (portal vein thrombosis, liver disorder, jaundice, ascites, fistula of bile duct and biliary anomaly) and venous thromboembolism.[9]
All donors want to save a loved one’s life, but the risk of donor morbidity and mortality is a major concern. Surgeon and center experience and higher annual case volume are associated with lower rates of postoperative complications, but live liver donor hepatectomy is not a complication-free procedure.[10] An important concern is the donor’s psychological state because acute or chronic mental health conditions, such as depression and anxiety, are present in one-third of patients with Takotsubo syndrome, making preoperative psychosocial evaluation mandatory.[11]
This presented case is particular because such cases of secondary Takotsubo syndrome in living liver donors are not currently reported in the literature.
Multimodality imaging plays a key role in establishing the diagnosis, guiding therapy, and stratifying prognosis in both the acute and post-acute phases of Takotsubo syndrome. Transthoracic echocardiography is the first-line imaging tool due to its widespread availability and feasibility, especially in the acute care setting, and the ability to rapidly monitor changes in cardiac function over time.[12]
Patients can be screened by using the InterTAK score. A score of more than 70 points and echocardiographic findings compatible with apical or mid-ventricular ballooning with circumferential pattern will orient the diagnosis towards Takotsubo. In this case, epicardial coronary anatomy may be assessed invasively by coronary angiography or non-invasively by coronary computed tomography angiography.[12]
Cardiac magnetic resonance (CMR) can be useful in the acute phase, but its use is conditioned by the more limited availability compared with echocardiography and the difficulties in imaging unstable patients. It provides more detailed information about LV morphology and function, RV involvement, intraventricular thrombi, and pericardial effusion. CMR is crucial during the post-acute phase to rule out other common pathologies (acute coronary syndrome and myocarditis).[13]
A limitation in the final diagnosis of secondary Takotsubo syndrome in this presented case is the lack of angiographic confirmation of coronary status, but the clinical and psychological context (being a hepatic donor for her sister), the post-major surgery state, the dysfunctional myocardium extending beyond the territory of a single coronary artery, the absence of cardiovascular risk factors, the high InterTAK score, and the transient character of regional left ventricle wall motion abnormalities are all arguments for the high likelihood of secondary Takotsubo syndrome.
There are no randomized clinical trials to support specific treatment recommendations in Takotsubo syndrome. The main objective of in-hospital treatment should be supportive care to sustain life and to minimize complications during recovery because the key feature of Takotsubo syndrome is the reversibility to normal cardiac function. In patients with pulmonary congestion and hemodynamic stability, treatment is directed to relieve congestion with diuretics and vasodilators. Arterial vasodilators and beta-adrenergic blockers are useful especially if hypertension is present. For patients with hemodynamic instability, treatment depends on the presence of left ventricular outflow tract obstruction (LVOTO). If there are no signs of LVOTO, inotropic agents, and if not sufficient, mechanical left ventricular assist devices (such as intra-aortic balloon pump or Impella) should be considered.[14,15] Intravenous fluids and low doses of short-acting beta-adrenergic blockers may be used with caution to reduce the LVOTO or peripherally active vasopressor drugs to maintain adequate perfusion pressure as a temporizing solution to LVOTO resolution or mechanical support.[16] In refractory cases of Takotsubo syndrome with LVOTO or biventricular failure, extracorporeal membrane oxygenation may be considered as a mechanical cardiac assist device.[17]
A risk of recurrence exists — 2% to 4% per year and up to 20% at 10 years; it is common even after recovery of left ventricle systolic function, and in contrast to previous perception, patients who have experienced Takotsubo syndrome may describes symptoms such as fatigue (74%), shortness of breath (43%), chest pain (8%), palpitations (8%), and exercise intolerance in comparison with control subjects.[18]
Currently, there is no evidence to guide the long-term management of these cases, including a meta-analysis showing a lack of efficacy of common heart failure therapies.[19] Nevertheless, many experts advocate for the use of ß-blockers in patients with increased sympathetic tone, ongoing cardiac symptoms, persistent anxiety, or recurrent episodes.[1] In the presented case, selective beta-adrenergic blockers were started after hemodynamic stability. Given the high frequency of mental health comorbidities, attention should also be given to assessment and referral to treatment for psychiatric problems that may persist and contribute to risk of recurrence and long-term outcomes. In addition, cardiac structural abnormalities (e.g., impaired LV strain patterns) and metabolic alterations have been described; a cardiac magnetic resonance was recommended after discharge of this patient for the evaluation of myocardial involvement and potential structural changes after this acute event.
Takotsubo syndrome is an acute reversible heart failure syndrome; it is a form of stress-induced cardiomyopathy characterized by transitory left ventricular wall motion abnormalities. Several pathophysiologic mechanisms have been proposed, such as the contributions of the autonomic sympathetic nervous system, circulating catecholamines, or endothelial dysfunction; however, the exact pathophysiology remains unknown. Also, there is little data regarding the management and long-term treatment of these patients; major recommendations are based on expert opinion. Takotsubo syndrome is a complication occurring in liver transplant recipients, with a rising number of cases published. Secondary Takotsubo syndrome occurring in a living liver donor is in itself a particularity due to the fact that such cases are not currently reported in literature. We report this case to raise awareness regarding this possible outcome in living donors for solid organ transplantation and to underline the importance of pre-operative screening in these patients, including psychological evaluation and support.