FIGURE 1.
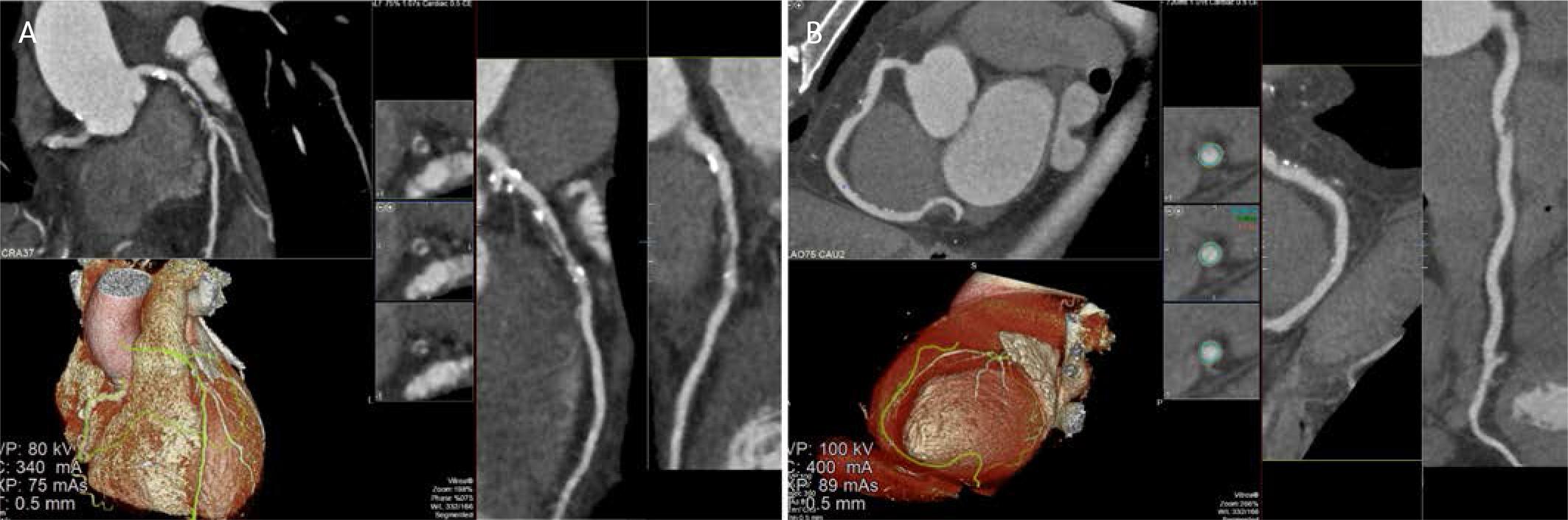
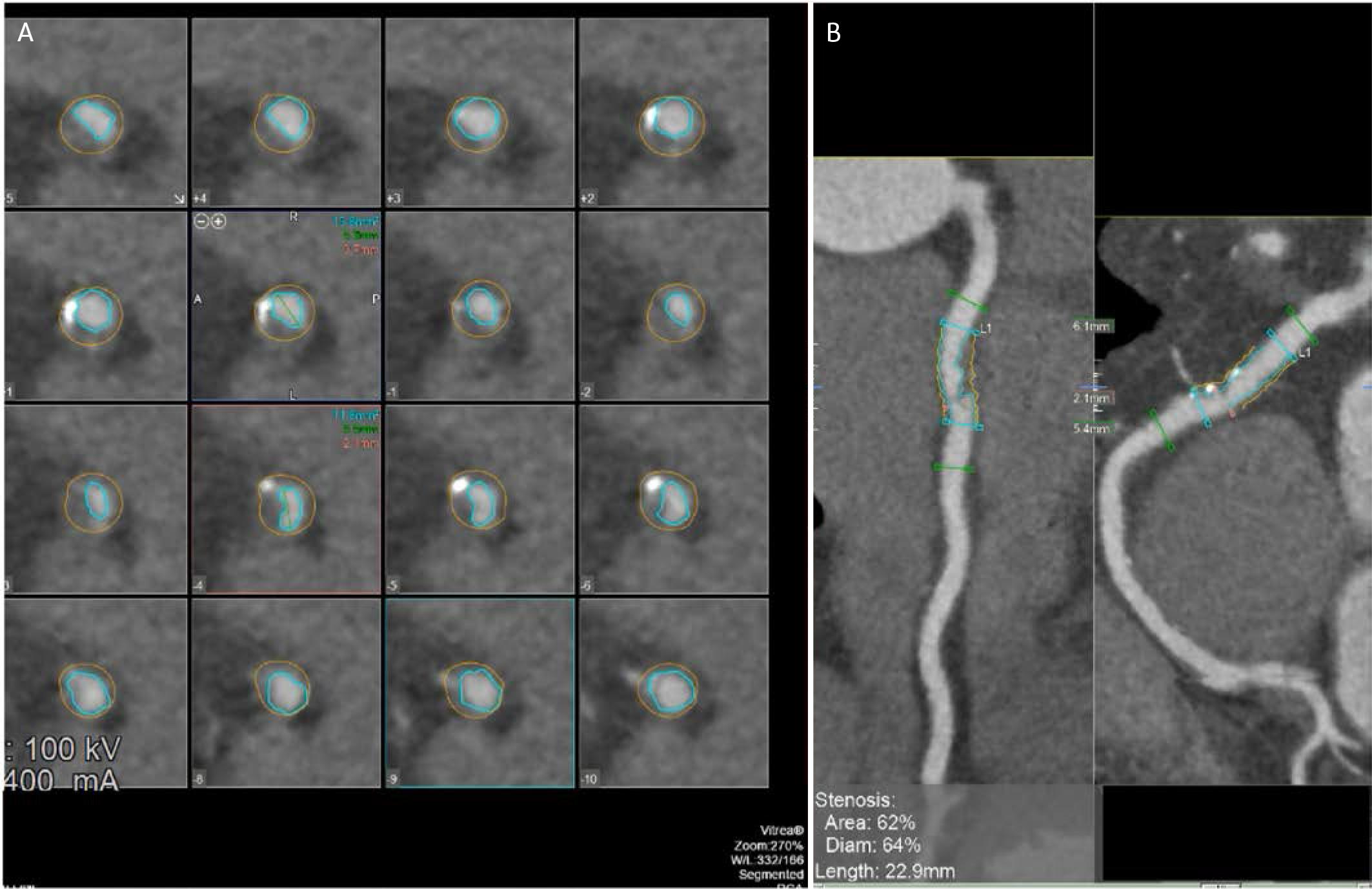
FIGURE 2.
Key randomized trials of CCTA in suspected NSTE-ACS
| Trial | Population | Primary outcome | Key findings |
|---|---|---|---|
| RAPID-CTCA15 | Suspected/provisional ACS; intermediate risk (n = 1,748) | 1-year death or type 1/4b MI | No difference in MACE; reduced ICA use; modest reduction in subsequent testing, no major LOS difference |
| VERDICT8 | Confirmed NSTE-ACS (n = 1,023) | Diagnostic performance vs. ICA | High NPV for ruling out ≥50% stenosis; accurately characterized CAD distribution |
| BEACON22 | ED suspected ACS, low-intermediate risk | MACE at follow-up | No difference in MACE; shorter LOS and lower costs in low-risk patients |
| PROTECCT21 | Intermediate hs-cTn ‘observe zone’ | 12-month MACE | No change in MACE; trend to fewer downstream tests |