Cardiac computed tomography (CT) has expanded far beyond its traditional role in stable coronary artery disease (CAD) over the past decades.1,2 Current applications include assessment of structural and valvular heart disease,3 quantification of coronary and valvular calcium burden,4,5 procedural planning for complex percutaneous interventions,6 and surveillance for cardiac allograft vasculopathy after cardiac transplantation.7 Despite these advances, the optimal role of coronary computed tomography angiography (CCTA) in acute coronary syndromes (ACSs) remains under active investigation.8,9
Invasive coronary angiography (ICA) is still the standard of care for high-risk non-ST-elevation acute coronary syndrome (NSTE-ACS). However, up to one-third of patients undergoing ICA show no significant obstructive CAD, underscoring the need for improved risk stratification and noninvasive diagnostic strategies such as CCTA.8,10,11 Recent guidelines suggest that CCTA is most effective in patients with a low-to-intermediate pretest likelihood of ACS, particularly when troponin and ECG findings are inconclusive.11,12
Randomized controlled trial (RCT) evidence indicates that CCTA can reduce ICA rates, but it has not consistently reduced major adverse cardiovascular events (MACE) or revascularization, and it may increase length of stay and costs.13 Therefore, routine CCTA use in all intermediaterisk patients is not supported. Instead, targeted research is needed to define subgroups based on troponin dynamics,14 clinical risk scores (GRACE score), and CT plaque phenotype, in whom CCTA may safely replace or defer ICA. Advanced CT biomarkers, including low-attenuation plaque burden and CT-derived fractional flow reserve, combined with high-sensitivity troponin algorithms, offer promising pathways to move beyond workflow efficiencies toward improved clinical outcomes.
This review critically appraises current evidence for CCTA in NSTE-ACS, with particular emphasis on the disconnect between excellent rule-out capability and the lack of consistent clinical benefit, and explores whether advanced plaque characterization can refine patient selection beyond traditional risk stratification.
To date, no large-scale RCT has directly compared a CCTA-guided management strategy with contemporary standard care specifically in patients with non-ST-elevation myocardial infarction (NSTEMI), for several practical and ethical reasons. First, NSTEMI management is grounded in early antithrombotic therapy and routine ICA to guide revascularization, as reflected in guidelines and entrenched clinical practice. This leaves limited evidence for randomizing higher-risk patients to a noninvasive pathway in which ICA might be omitted or delayed. Second, most CCTA trials in ACS have enrolled mixed populations, suspected ACS, unstable angina, and troponin-negative or indeterminate cases rather than exclusively NSTEMI. The major randomized trials evaluating CCTA in suspected or confirmed NSTE-ACS are summarized in Table 1.
Key randomized trials of CCTA in suspected NSTE-ACS
| Trial | Population | Primary outcome | Key findings |
|---|---|---|---|
| RAPID-CTCA15 | Suspected/provisional ACS; intermediate risk (n = 1,748) | 1-year death or type 1/4b MI | No difference in MACE; reduced ICA use; modest reduction in subsequent testing, no major LOS difference |
| VERDICT8 | Confirmed NSTE-ACS (n = 1,023) | Diagnostic performance vs. ICA | High NPV for ruling out ≥50% stenosis; accurately characterized CAD distribution |
| BEACON22 | ED suspected ACS, low-intermediate risk | MACE at follow-up | No difference in MACE; shorter LOS and lower costs in low-risk patients |
| PROTECCT21 | Intermediate hs-cTn ‘observe zone’ | 12-month MACE | No change in MACE; trend to fewer downstream tests |
ED, emergency department; hs-cTn, high-sensitivity cardiac troponin; LOS, length of stay; MI, myocardial infarction
The RAPID-CTCA trial enrolled intermediate- to high-risk patients but was not powered to determine whether a CCTA-guided strategy improves outcomes in the confirmed NSTEMI subgroup.15 In this landmark trial, early CCTA did not reduce the primary endpoint of 1-year all-cause death or recurrent type 1 or type 4b myocardial infarction (5.8% in the CCTA group vs. 6.1% with standard care; adjusted hazard ratio 0.91, 95% CI 0.62–1.35; P = 0.65).14 Early CCTA reduced ICA use (adjusted hazard ratio 0.81, 95% CI 0.72–0.92; p = 0.001), yet rates of revascularization, ACS therapies, and preventive treatments at discharge were similar between groups.15 Hospital length of stay increased modestly (median increase 0.21 days, 95% CI 0.05–0.40 days), and overall healthcare costs were higher with CCTA.14 Subgroup analyses further suggested that the effects of CCTA on outcomes and resource use were broadly consistent regardless of baseline troponin status.16,17
The VERDICT trials enrolled one of the most clinically relevant populations: confirmed NSTE-ACS patients with ischemic ECG changes and/or elevated biomarkers.8,18 CCTA achieved a per-patient negative predictive value (NPV) of 90.9% for excluding ≥50% coronary stenosis, with a sensitivity of 96.5% and specificity of 72.4%. Notably, NPV remained stable across clinical risk strata, and diagnostic performance was similar whether CCTA was performed 2–3 hours or 2–3 days after diagnosis. Approximately 33% of NSTE-ACS patients in VERDICT had non-obstructive CAD, highlighting marked heterogeneity within this syndrome.8,19 The VERDICT program, therefore, supports excellent diagnostic and prognostic performance of CCTA; however, because ICA was performed in all participants by design, CCTA did not function as a gatekeeper, and there is no direct evidence that CCTA-guided management reduces MACE in this high-risk population. A major limitation is the observational nature of CCTA within the trial: CCTA findings were blinded and did not influence clinical decisions, leaving unresolved whether a CCTA-first strategy would alter outcomes. Despite this, CCTA appeared comparable to ICA for predicting 4-year clinical outcomes.19,20
The PROTECCT trial addressed a different clinical question by targeting the diagnostic ‘gray zone’: patients with intermediate troponin concentrations (5–50 ng/L) and non-ischemic ECGs.21 In this setting, where diagnosis remains uncertain after initial troponin testing, CCTA did not reduce length of stay or overall costs, although patients with <25% stenosis had shorter admissions and 12-month MACE rates were similar between strategies.21 These neutral results likely reflect the difficulty of confidently identifying patients with ≥25% stenosis in this intermediate-risk cohort, leading to admissions and additional testing that attenuate any advantage of early CCTA.
BEACON and CARMENTA enrolled broader suspected ACS populations while excluding patients requiring urgent catheterization, thereby under-representing the highest risk ‘authentic NSTEMI’ cohort that defines the key evidence gap.22,23 BEACON included patients with acute chest pain or symptoms suggestive of ACS requiring further evaluation. The cohort had a higher CAD prevalence than earlier studies, partly due to the inclusion of individuals with mildly elevated high-sensitivity troponins, while very low-risk and non-cardiac presentations were excluded. Conducted in the high-sensitivity troponin era, BEACON reflects improved standard care that likely reduces the incremental diagnostic value of early CCTA.22 Revascularization rates were similar (9% with CCTA vs. 7% with standard care; p = 0.40), as were safety outcomes and MACE.22 Nonetheless, outpatient testing was less frequent and direct medical costs were lower in the CCTA arm,22 supporting the concept that evolving standard pathways may shift the value proposition of CCTA toward resource utilization rather than hard outcomes.
CARMENTA directly tested a CCTA-first strategy against standard care in NSTEMI.23 Consecutive patients with acute chest pain, normal or inconclusive ECG (no ST-T elevation or dynamic changes), and elevated high-sensitivity troponin T (>14 ng/L at baseline or 3 hours) were randomized; all were considered appropriate for ICA before inclusion. Key exclusions included severe ischemia requiring urgent angiography, symptoms suggestive of a non-myocardial infarction cause, previous CAD (myocardial infarction, revascularization, >50% stenosis, or positive noninvasive imaging), type II myocardial infarction, age >85 years, and contraindications to CCTA or cardiac magnetic resonance. Thus, CARMENTA focused on patients with intermediate-risk NSTEMI with biomarker elevation, no ongoing ischemia, and no previous CAD.23 A CCTA-first strategy reduced ICA by 34% versus usual care (p < 0.001) without increasing 1-year MACE or procedure-related adverse events (16% with CCTA-first vs. 23% with routine care; p = 0.28).23 These data suggest that, in carefully selected patients with NSTEMI, CCTA-first may safely reduce unnecessary invasive procedures.
Overall, evidence for hard outcomes in high-risk NSTE-ACS/NSTEMI remains limited and largely indirect. The most robust studies consistently show that CCTA can be safely implemented in intermediate-risk ACS with meaningful effects on resource utilization, but without demonstrable superiority in clinical outcomes across risk categories. Elzanaty et al. and Kong et al. similarly report that, in RCTs enrolling suspected/confirmed NSTE-ACS, an initial CCTA strategy reduces ICA referral and improves the diagnostic yield of obstructive disease among those who proceed to ICA, without a clear difference in MACE.24,25
Economic analyses and meta-analyses report cost reductions of approximately 17–21% with CCTA in low-to-intermediate risk cohorts.26,27 In RAPID-CTCA and implementation studies, cost savings appear to derive mainly from earlier discharge and avoidance of subsequent non-invasive testing rather than from large absolute reductions in ICA volume.15 Accordingly, economic benefit is well established for low- and intermediate-risk ACS, while cost superiority in high-risk NSTE-ACS/NSTEMI remains unproven.
The high-sensitivity troponin era has also created a diagnostic paradox that CCTA may help address. Contemporary data indicate that 25–50% of patients with elevated high-sensitivity troponin have non-obstructive CAD at ICA.28,29 In this context, traditional risk tools (GRACE, TIMI, ESC risk scores) show limited ability to discriminate obstructive CAD among unstable patients. Many troponinpositive presentations reflect type 2 myocardial infarction or myocardial injury rather than type 1 myocardial infarction and therefore require different diagnostic pathways. Myocardial infarction with non-obstructive coronary arteries (MINOCA) represents a smaller but clinically important subgroup, accounting for approximately 5–15% of myocardial infarction cases.30
Culprit identification remains a major challenge in NSTEMI. Large registries and angiographic series demonstrate that 20–34% of patients with NSTEMI have coronary occlusions despite the absence of ST-segment elevation.28,29 In multivessel disease, common in NSTE-ACS, up to 30% of cases may have no clearly identifiable culprit on angiography, and more than 10% may have multiple plausible culprit lesions.31 The 2025 American College of Cardiology guidelines acknowledge that culprit-vessel identification in NSTE-ACS with multivessel disease is often difficult and that angiography may overestimate nonculprit lesion severity.32 At present, CCTA cannot reliably distinguish culprit from bystander lesions prospectively: it primarily characterizes stenosis and plaque morphology but cannot consistently identify the acutely thrombosed or ruptured plaque driving the event. This remains a critical evidence gap and a priority for development and validation of noninvasive culprit-identification strategies in NSTEMI and NSTE-ACS.
Beyond stenosis, CCTA can provide prognostic information through plaque characterization. A RAPID-CTCA substudy showed that low-attenuation plaque burden independently predicted 1-year mortality or recurrent myocardial infarction in patients with acute chest pain.33 Quantitative plaque analysis demonstrated that patients above the median low-attenuation plaque burden had an approximately eightfold higher risk of death or nonfatal myocardial infarction at 1 year (HR 7.80; 95% CI 2.33–26.0; p < 0.001), independent of GRACE score and obstructive CAD, and outperforming stenosis severity alone.33 This aligns with SCOT-HEART and related data suggesting that besides stenosis severity, plaque composition also influences outcomes.34
Guidelines have increasingly reflected these nuances. The 2023 ESC Guidelines for ACS management downgraded CCTA in NSTE-ACS from Class I (2020 NSTE-ACS guidelines) to Class IIa, particularly when high-sensitivity troponin results are ambiguous or when troponin is elevated without diagnostic ECG changes.11 Importantly, CCTA is contraindicated (Class III) in patients with clear high-risk features requiring prompt invasive management.15,22,35 Similarly, the 2025 ACC/AHA Guideline for ACS management recommends CCTA in selected patients with low-risk NSTE-ACS when the diagnosis is uncertain, as part of a selective invasive strategy to refine risk assessment and the need for ICA.
Taken together, this evolution highlights a central limitation: an approach focused solely on stenosis detection may be insufficient to guide management decisions that improve outcomes. This strengthens the rationale for evaluating whether advanced plaque characterization, such as quantitative plaque analysis and assessment of pericoronary inflammation, can improve risk prediction beyond conventional CCTA and enable better patient selection for invasive versus noninvasive strategies.
The evolution of this guideline highlights a critical issue: focusing entirely on stenosis detection seems inadequate for informing management decisions that enhance outcomes. This scenario necessitates exploration into whether advanced plaque characterization, such as quantitative plaque analysis and pericoronary inflammation assessment, can enhance risk prediction beyond the capabilities of conventional CCTA.
Advanced plaque characterization with CCTA has shifted from a largely qualitative description of ‘high-risk’ features to a more quantitative assessment of plaque composition and pericoronary inflammation. High-risk plaque characteristics, including positive remodeling, the napkin-ring sign, spotty calcification, and low-attenuation plaque, have been independently validated as markers of plaque vulnerability (Figure 1).36 The SCOT-HEART study established low-attenuation noncalcified plaque as an independent predictor of myocardial infarction, while RAPID-CTCA confirmed that low-attenuation plaque burden predicts 1-year adverse outcomes in patients with suspected ACS. However, key evidence gaps remain: (i) the absence of standardized composite scoring systems specifically validated in acute settings, and (ii) the lack of prospective randomized evidence showing that identification of these features changes management and improves outcomes. Most studies have relied on binary or simple additive approaches, and quantitative/composite scores combining low-attenuation plaque, positive remodeling, napkin-ring sign, and spotty calcification are not yet validated for routine clinical use.37,38 Coronary Artery Disease-Reporting and Data System (CAD-RADS) 2.0 incorporates these plaque characteristics, but prospective validation and real-world applicability in acute scenarios remain limited.
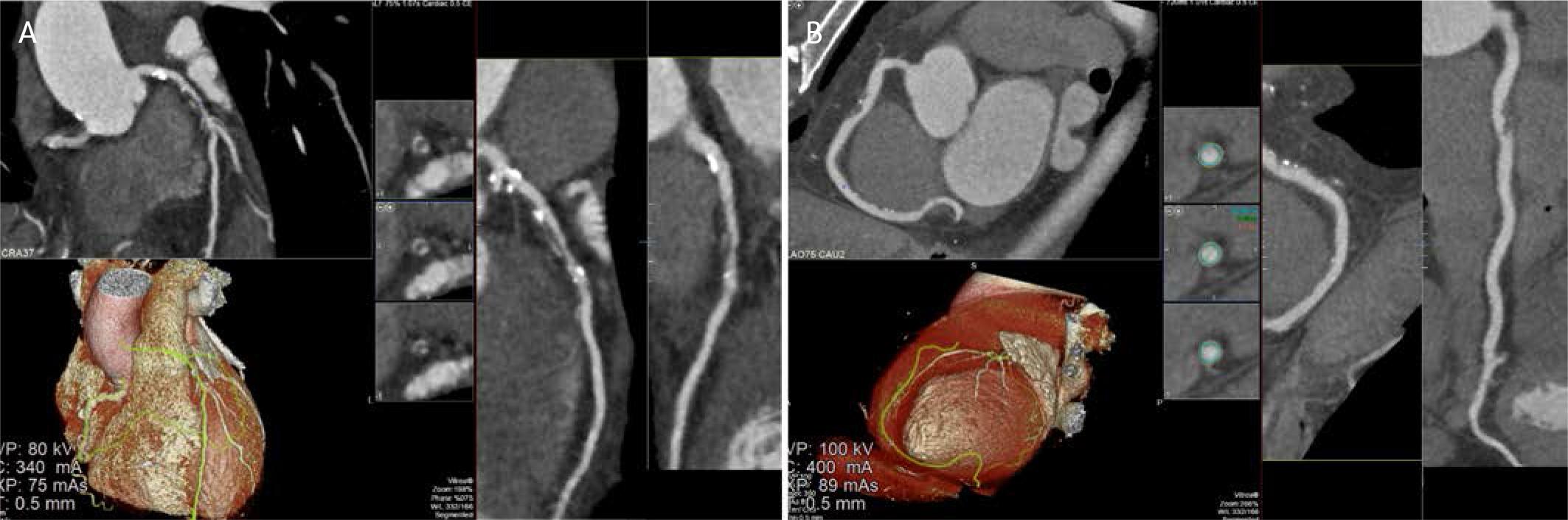
CCTA demonstrating high-risk atherosclerotic plaque morphology in the left anterior descending and right coronary arteries. A. Multiplanar reconstruction of the left anterior descending artery demonstrates an eccentric, predominantly non calcified, low attenuation plaque in the proximal–mid segment, associated with positive vessel remodeling and superimposed spotty calcifications. Volume rendered 3D reconstruction confirms the extent and distribution of the lesion along the LAD. B. CCTA images illustrating high-risk plaque characteristics. Axial cross-sectional images (upper right) demonstrate the typical features of vulnerable plaque, including the napkin-ring sign, spotty calcification, and low-attenuation plaque. The 3D volume rendered reconstruction (lower left) depicts the spatial relationship of the high-risk plaque within the coronary anatomy.
The ROMICAT II trial showed that CCTA in patients with suspected NSTE-ACS reduced emergency department length of stay and increased direct discharge compared with routine evaluation.39 In ROMICAT II substudies, classic high-risk plaque features detected by CCTA provided independent and incremental prediction of ACS beyond obstructive stenosis.40 Quantitative scoring approaches (the ROMICAT score) that integrate plaque features improved diagnostic performance, achieving AUC values up to 0.91 when combined with clinical variables and stenosis severity.40 Nevertheless, despite improvements in diagnostic accuracy and process metrics, ROMICAT II did not demonstrate reductions in MACEs or downstream resource utilization compared with routine care.35
Assessment of pericoronary adipose tissue (PCAT) reflects a broader transition from purely anatomic imaging to functional–inflammatory phenotyping. The fat attenuation index (FAI), measured in Hounsfield units (HU) as PCAT attenuation, has emerged as a noninvasive biomarker of coronary inflammation.36,41 In NSTE-ACS, lesions with FAI values > -70.1 HU have been associated with a higher prevalence of spotty (patchy) calcification and low-attenuation plaque.42 Elevated FAI values have also been linked to more pro-inflammatory intracellular cytokine profiles in NSTE-ACS lesions, supporting FAI as an imaging correlate of inflammatory activity and plaque vulnerability in ACS. However, focused data in exclusive NSTEMI or unstable angina cohorts remain limited. In chronic/stable CAD, the same -70.1 HU threshold has been validated as a marker of residual inflammatory risk and predicts cardiac mortality and MACE independently of traditional risk factors, coronary calcium, and high-risk plaque features.43,44 Overall, PCAT/FAI appears most informative when integrated with quantitative plaque analysis and conventional risk assessment, rather than used as stand-alone markers, and recent studies suggest that CCTA-derived scores may correlate with pericoronary inflammation, as quantified by the FAI.45
Radiomics and texture analysis represent the leading edge of PCAT evaluation, with encouraging but still early evidence in NSTE-ACS. In a study involving 216 patients (108 NSTEMI, 108 unstable angina), Wang et al. validated PCAT radiomics; a combined three-vessel PCAT radiomics model achieved AUCs of 0.871–0.876 across training and two internal validation cohorts, and consistently outperformed both single-vessel models and an epicardial adipose tissue model (AUC 0.684–0.694).46 More recently, Lu et al. reported that PCAT radiomics could discriminate NSTEMI from unstable angina: in a cohort of 204 patients (102 NSTEMI, 102 unstable angina), radiomic features selected through a multi-step pipeline achieved an AUC of 0.898 in the test cohort.47 Importantly, this study applied contemporary high-sensitivity troponin-based diagnostic criteria, directly addressing the evolving classification of ACS.47 External validation and demonstration of incremental clinical utility in prospective workflows remain essential before large-scale implementation.
CCTA has been extensively validated in low- to intermediate-risk chest pain pathways and performs well for excluding CAD. In parallel, CT-derived fractional flow reserve (FFR-CT) has been widely studied in stable CAD as a non-invasive method to assess the functional significance of intermediate stenoses, reducing the need for invasive physiological assessment.48 More recently, CCTA, particularly when complemented by FFR-CT, has been proposed to reduce unnecessary ICA in selected patients with suspected NSTEMI. This interest has intensified in the high-sensitivity troponin era: while detection of myocardial injury has improved, specificity for type 1 myocardial infarction has declined, increasing referrals to ICA among patients with noncoronary causes of troponin elevation. Consequently, up to half of cases labeled as NSTEMI who undergo ICA do not ultimately receive coronary revascularization.49,50
In troponin-positive NSTEMI cohorts, Meier et al. reported that FFR-CT provided higher diagnostic accuracy than CCTA alone and improved the ability to rule out hemodynamically significant stenoses, using ICA as the reference standard.51 In a smaller cohort, Warren et al. found a strong correlation between FFR-CT and invasive FFR (r = 0.78); in per-vessel analyses, FFR-CT showed excellent diagnostic accuracy and outperformed anatomic assessment by CCTA alone.52
Collectively, these findings suggest that CCTA, alone or combined with FFR-CT, may refine NSTEMI management by improving the selection of patients most likely to benefit from ICA. Notably, even though all participants in the study of Meier et al. met NSTEMI diagnostic criteria (including troponin elevation), a substantial proportion had no significant obstructive CAD.51 This highlights the limitations of contemporary pathways: despite meeting guideline criteria for an early invasive strategy, more than one-third of patients underwent ICA without identification of an actionable culprit lesion. Beyond clinical implications, this exposes patients to avoidable procedural risk and increases healthcare utilization and costs. Prospective trials are therefore needed to demonstrate that an FFR-CT-guided strategy can safely reduce unnecessary ICA in NSTEMI, and to clarify its incremental value for nonculprit lesion assessment, where functional significance, timing, and the effects of acute microvascular dysfunction may differ markedly from stable settings.
CT myocardial perfusion imaging (CT-MPI) is established in chronic coronary syndromes but remains less adopted in NSTEMI evaluation.53 The CALAMARI study (Ct Angiography/perfusion evaluation of non-st-eLevAtion MyocARdial Infarction) was a single-center prospective diagnostic cross-sectional study that assessed the sensitivity and specificity of combined CCTA and CT-MPI against ICA with FFR in NSTEMI.50 In this small cohort (n = 19), an integrated CCTA/CT-MPI protocol was technically feasible, provided clinically meaningful diagnostic information, and helped identify significant stenoses while distinguishing patients with nonobstructive CAD who might safely defer ICA.54 These early data support potential utility for triage and, possibly, culprit identification, but major gaps remain before broad implementation. First, robust head-to-head comparisons with established stress imaging modalities (stress CMR, PET/SPECT, stress echocardiography) are limited, preventing a clear definition of incremental diagnostic value, downstream management impact, and outcomes. Second, optimal timing of stress CT-MPI after presentation remains uncertain, as ACS involves dynamic coronary physiology and microvascular dysfunction that may influence perfusion measurements, particularly early after symptom onset or biomarker rise.
Health-economic evidence is also limited and highly system-dependent. Most cost/resource analyses of FFR-CT have been performed in stable chest pain pathways, and cost-effectiveness varies by payer structure, reimbursement, and downstream testing patterns. Real-world analyses suggest that value depends strongly on targeting (intermediate stenoses) and local workflow integration.55
Artificial intelligence (AI)-enhanced risk stratification in CAD has expanded rapidly, with many models trained and validated in stable or suspected stable CAD cohorts, often leveraging CCTA-derived plaque and stenosis quantification and providing incremental prognostic value for future events (Figure 2).56 In contrast, NSTEMI-specific AI tools remain comparatively underdeveloped. Although machine-learning-based scores for ACS exist (GRACE 3.0 for NSTE-ACS risk assessment), much of the supporting evidence comes from retrospective/registry datasets and external validation rather than models trained and tested within contemporary, unselected troponin-positive NSTEMI pathways.57 This limitation is clinically important because guideline-directed practice still relies on traditional risk scores (GRACE/TIMI) to guide early risk stratification and invasive timing decisions.11 A critical next step is prospective head-to-head validation in NSTEMI comparing AI models against GRACE/TIMI (discrimination, calibration, net reclassification), and, most importantly, testing whether AI-guided strategies can improve decisions and reduce events without delaying care in truly high-risk patients.
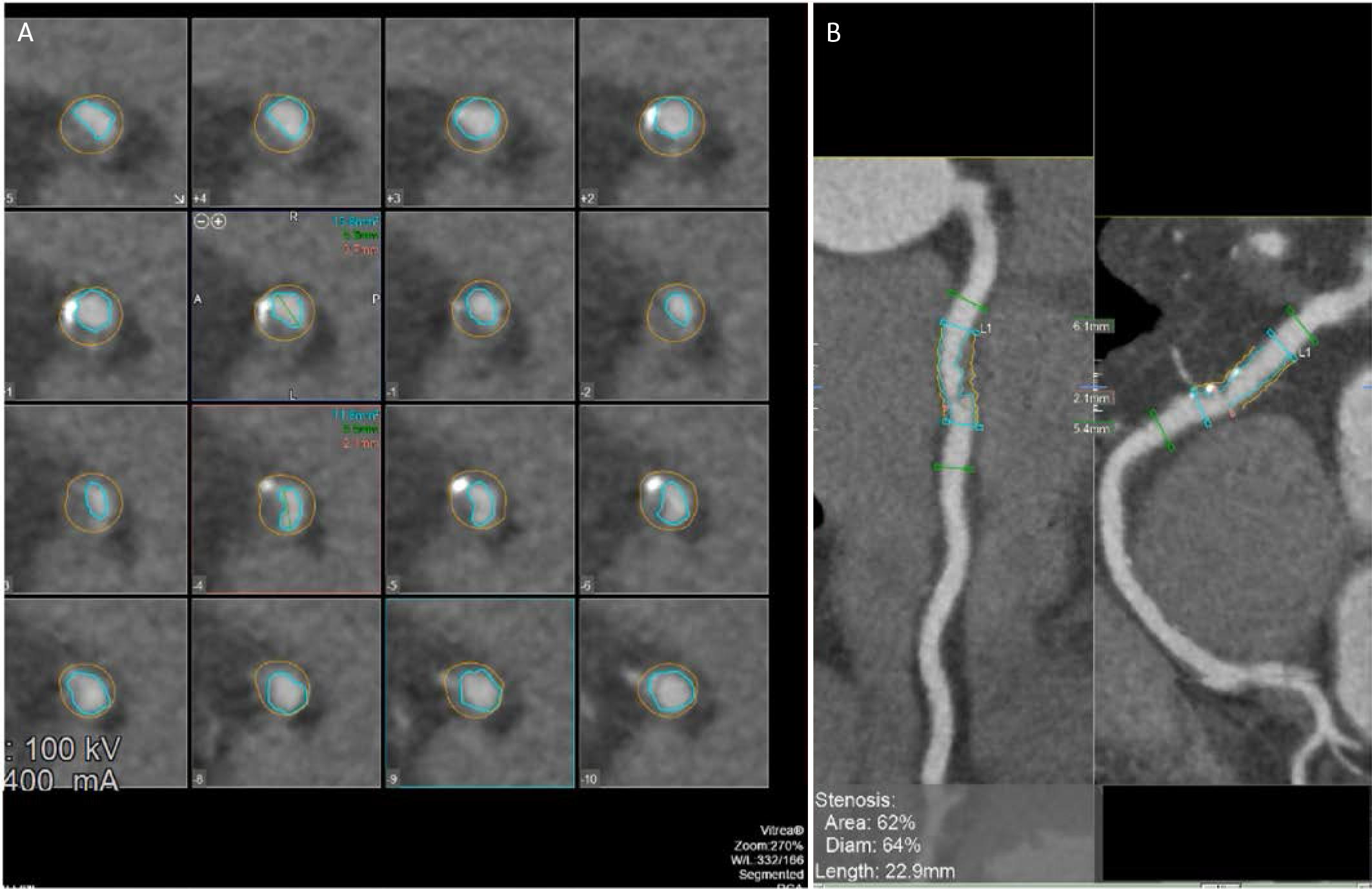
Automated coronary plaque quantification and stenosis assessment using CCTA post-processing software. A. Serial crosssectional images along the lesion, with automated luminal (inner contour) and vessel (outer contour) segmentation enabling precise assessment of plaque burden and vessel remodeling. B. Multiplanar reconstruction views of the affected coronary segment with software-derived measurements indicating a 62% area stenosis and 64% diameter stenosis over a lesion length of 22.9 mm, consistent with hemodynamically significant obstructive CAD as defined by contemporary CCTA criteria.
Automated plaque analysis from CCTA has matured sub - stantially. Multiple deep-learning pipelines can now segment coronary arteries and quantify stenosis and plaque burden/composition with good reproducibility and agreement against expert readers and invasive reference standards. A recent American consensus states that, among patients with visually evident plaque on CCTA, adding quantitative plaque analysis may be useful to enhance risk assessment and guide the intensity of preventive therapies.58 In the multicenter international CONFIRM2 registry, AI-guided quantitative CCTA driven mainly by noncalcified plaque burden and diameter stenosis improved 3-year MACE risk stratification compared with standard clinical CT metrics (including CAD-RADS, coronary calcium score, and the modified Duke Index), with higher discrimination and significant net reclassification.59
In a large multicenter study, Lin et al. developed and externally validated a deep-learning system that rapidly generates plaque and stenosis measurements from CCTA and demonstrated prognostic value for myocardial infarction.56 Similarly, the prospective multicenter REVEAL-PLAQUE study showed strong agreement between Al-derived plaque quantification and intravascular ultrasound for total and component plaque volumes.60 Real-world validations further support the generalizability of fully automated plaque/stenosis quantification across heterogeneous datasets.61 To standardize reporting and facilitate adoption, the Society of Cardiovascular Computed Tomography has published recommendations for quantitative CCTA assessment, including definitions and reporting standards for plaque metrics.62
Despite technical validation, real-time deployment in acute-care workflows remains a major translational gap. Although on-premises AI solutions can generate clinically relevant outputs within minutes in research settings,63 evidence for reliable integration into time-critical emergency decision-making is still limited.
Integrated multimodal AI aims to combine CCTA biomarkers with clinical, ECG, and laboratory variables to produce individualized diagnostic and prognostic outputs. Xu et al. developed a Random Forest model using CCTA features that identified ACS culprit lesions within 7 days (AUC 0.85) and stratified subsequent ACS risk, with strongest predictive performance within 2 years.64 These data support integrating anatomical and physiological CT features for more refined risk prediction.64
Beyond atherosclerotic plaque rupture, NSTE-ACS presentations can reflect alternative mechanisms such as spontaneous coronary artery dissection (SCAD), coronary embolism, vasospasm, and myocarditis.11 MINOCA, reported in approximately 5–25% of troponin-positive NSTEMI presentations, remains diagnostically and therapeutically challenging.11’32’65 Whether CCTA can reliably identify nonatherosclerotic etiologies or add value beyond ICA for phenotyping MINOCA remains an important but underexplored frontier.
For SCAD, CCTA demonstrates high specificity (up to 97%) but limited sensitivity (approximately 52–58% overall). Sensitivity is higher in proximal segments and lower in distal vessels, where SCAD is often most prevalent.66–68 CCTA can depict features such as abrupt or tapered luminal narrowing, intramural hematoma, and, less commonly, a visible dissection flap; however, spatial resolution remains inferior to ICA, and CCTA should not be considered a substitute for ICA in the acute setting. Its main advantage lies in noninvasive follow-up and in screening for extracoronary arteriopathies (e.g., fibromuscular dysplasia), as well as documenting healing in proximal or larger-caliber vessels.66’67’69
Myocarditis is not reliably diagnosed by CCTA, cardiac MRI remaining the preferred modality for tissue characterization and detection of myocardial inflammation/edema.70 Emerging data suggest potential utility of cardiac CT-derived extracellular volume quantification, but most evidence comes from valvular heart disease cohorts71 rather than ACS populations, and diagnostic performance in NSTE-ACS remains to be established.
Event-driven, prospective RCTs directly comparing CC-TA-guided strategies with standard invasive pathways, using MACE reduction as the primary endpoint, remain a critical unmet need. Current evidence supports that CCTA can reduce ICA utilization in selected NSTEMI cohorts, but it has not demonstrated consistent reductions in MACE. Large-scale trials, including TRACTION, are underway to address this gap.72
In parallel, AI implementation must progress beyond algorithm development to robust multicenter validation across diverse and multi-ethnic cohorts to improve generalizability and reduce bias. Standardized frameworks for plaque characterization and reporting, including ACS-adapted implementations of CAD-RADS 2.0 and ACS-specific validation of FAI thresholds, should be prioritized. Finally, direct comparisons of CT-FFR and invasive FFR in NSTEMI are needed: although correlation and diagnostic accuracy appear promising, further validation is required before CT-FFR can be confidently used to guide management in acute settings.52
CCTA provides strong diagnostic performance in NSTE-ACS, offering high negative predictive value alongside detailed assessment of plaque phenotype and pericoronary inflammation. However, these technical strengths have not yet translated into outcome benefits sufficient to justify routine adoption. A persistent paradox remains: excellent anatomic visualization does not consistently reduce mortality, myocardial infarction, or overall costs. Progress will require shifting the focus from whether CCTA can detect disease to identifying which patients benefit from early anatomical (and functional–inflammatory) information before invasive testing. Emerging approaches, quantitative plaque analysis, PCAT radiomics, and CT-FFR may improve patient selection, but they require prospective validation demonstrating that imaging-guided management changes are clinically meaningful and improve outcomes.