The Pacific white shrimp, scientifically known as Penaeus vannamei (Boone, 1931) is a species of significant commercial importance and is farmed extensively across many countries and regions around the world. The rapid expansion of intensive shrimp aquaculture has unfortunately led to environmental damage and significant crop losses caused by disease outbreaks in many regions, primarily due to excessive water exchange and large amounts of effluent discharge (Xu et al., 2018; Khanjani et al., 2024 a, 2025 a; Khanjani and Mohammadi, 2025). In response, biofloc-based production systems that use minimal/zero water exchange have recently been developed for cultivating P. vannamei (Khanjani et al., 2025 b). These innovative systems help reduce environmental impact, enhance biosecurity, and boost overall production (Samocha et al., 2015; Emerenciano et al., 2025). In these systems, bioflocs serve a dual purpose: they maintain water quality by removing harmful nitrogen compounds like ammonia and nitrite (Khanjani et al., 2024 b, 2025 c), and they also enhance shrimp performance by improving feed utilization (Wasielesky et al., 2006), promoting growth (Khanjani et al., 2022 a), and boosting natural immune and antioxidant activities (Xu and Pan, 2013).
Pacific white shrimp and Nile tilapia have been widely used as benchmarks in biofloc aquaculture systems )Samocha et al., 2017, Khanjani and Alizadeh, 2024). However, the successful application of biofloc aquaculture systems has also been observed in other species such as butter catfish (Ompok bimaculatus) (Behera et al., 2025), freshwater prawns (Macrobrachium rosenbergii) (Negrini et al., 2017), black tiger shrimp (Penaeus monodon) (Promthale et al., 2019), banana shrimp (Fenneropenaeus merguiensis) (Khanjani and Sharifinia, 2022), brown shrimp (Penaeus aztecus) (Kaya, 2025), giant gourami (Osphronemus goramy) (Amriawati et al., 2021), common carp (Cyprinus carpio) (Minabi et al., 2020), and rohu (Labeo rohita) (Ahmed et al., 2019).
Shrimp aquaculture production can be enhanced by increasing both the cultivation area and the stocking density. Moreover, different stocking densities have been reported in biofloc aquaculture systems for species such as P. vannamei (Said et al., 2022; Irani et al., 2023), F. merguiensis (Khanjani et al., 2022 b), and Cryphiops caementarius (Mendez et al., 2024). Stocking density is a critical component of shrimp farming performance that must be meticulously optimized to secure both economic sustainability and shrimp welfare (Ghosh et al., 2013).
Contemporary shrimp farming is transitioning from extensive, low-yield operations in large ponds to more controlled, high-yield systems in smaller ponds and tanks under biofloc technology (BFT) conditions (Rodríguez-Olague et al., 2021). Over the past decade, significant advancements have been made in the production of P. vannamei using high density, biofloc-based systems that operate with zero water exchange (Prangnell et al., 2016; Samocha et al., 2017; Taw, 2010; Irani et al., 2023; Khanjani et al., 2025 b).
In shrimp farming systems that use BFT, different stocking densities affect water quality (Khanjani et al., 2022 b), growth performance (Said et al., 2024), survival rates (Tao et al., 2021), immune and antioxidant activities (Liu et al., 2017; Said et al., 2024), biochemical indices (Mendez et al., 2024), and microbial communities (Said et al., 2022). For example, Said et al. (2024) investigated how stocking density influences shrimp farming using a BFT system and found that it has a significant impact. Their study compared two BFT setups (one stocked at 50 shrimp/m2 and the other at 200 shrimp/m2) revealing that higher densities come with several disadvantages. Specifically, increased density resulted in reduced growth performance, a higher feed conversion ratio, and lower survival rates. Additionally, shrimp reared at the higher density showed diminished expression of genes linked to growth and immunity, alongside an elevated expression of stress-related genes.
There is limited information regarding the effect of stocking density on hemolymph biochemical and immune parameters, as well as on the antioxidant and metabolic activities of the hepatopancreas in Pacific white shrimp cultured in a biofloc system. This gap will be addressed in the present study.
The experiment was conducted for 35 days at the Persian Gulf marine aquaculture hatchery in Bandar Kolahi (Minab, Hormozgan Province, Iran). Juvenile Pacific white shrimp with an initial average weight of 3.85±0.34 g (M ± SD) were obtained from shrimp rearing ponds in Tiyab, Minab. For the present study, four experimental treatments were arranged in three replicates across 12 circular fiberglass tanks. Prior to stocking, the tanks were disinfected and rinsed, then each was filled with 150 L of sand-filtered water. The biofloc aquaculture system was evaluated under four distinct stocking densities: Treatment 1 at 0.308 g/L, Treatment 2 at 0.616 g/L, Treatment 3 at 0.924 g/L, and Treatment 4 at 1.23 g/L (Table 1).
Characteristics of the treatments used based on different stocking densities, in 35 days of the experiment
| Treatment 1 | Stocking density | Number of shrimp in the rearing tank (150 L) | No. | WE (daily) |
|---|---|---|---|---|
| 0.5–1% WE | SD1 | 12 | 0.308 g/L | Treatment 1 |
| 0.5–1% WE | SD2 | 24 | 0.616 g/L | Treatment 2 |
| 0.5–1% WE | SD3 | 36 | 0.924 g/L | Treatment 3 |
| 0.5–1% WE | SD4 | 48 | 1.23 g/L | Treatment 4 |
Abbreviations: number (No), water exchange (WE).
In the treatments, before stocking the shrimp, 0.5 ml of floc/L was added to each tank as the initial inoculum. Initially, shrimp were fed three times a day (at 8:00 a.m., 2:00 p.m., and 8:00 p.m.) at a rate equivalent to 5% of their body weight, using a diet containing 38% protein, 9% lipid, and 14% ash (Faradaneh Manufacturing Co., Shahrekord, Iran). As the shrimp grew, the feeding rate as a percentage of their body weight was gradually reduced.
The experiment was conducted in an indoor hall under natural lighting conditions. Water quality parameters including temperature (27–28°C), dissolved oxygen (5–6 mg/L), and salinity (31–32 ppt) were recorded across the different treatments. For aeration and oxygen supply, three air stones connected to an air source were installed at the bottom of the tanks. To promote the growth and development of biofloc in tanks with limited water exchange, a carbon source molasses (with 55.18% dry matter and 46.25% carbohydrates) was used to adjust the carbon-to-nitrogen ratio to 15:1 (Avnimelech, 2015).
To calculate and compare growth indices including weight gain and specific growth rate shrimp were weighed at the beginning of the experiment and then weekly throughout the rearing period. The numbers of shrimp stocked at the start and remaining at the end of the experiment were recorded to determine survival rates. Additionally, nutritional indices such as the feed conversion ratio were calculated based on the formulas provided below:
At the end of the experiment, shrimp were fasted for 24 hours prior to sampling. Fifteen shrimp from each replicate were randomly selected, anesthetized using clove oil at a dose of 100 ppm, and then humanely euthanized in accordance with ethical guidelines (Luedemana and Lightnera, 1992). The hepatopancreas was dissected on ice. The isolated tissue was rinsed with a cold saline solution (0.8%) and subsequently stored at –80°C for later analyses.
For the preparation of the enzyme extract, the hepatopancreas tissue was thawed and homogenized at 4°C using a hydrochloric acid–Tris buffer at a pH of 7.4. The resulting homogenate was then centrifuged at 4000 rpm for 10 minutes at 4°C, and the supernatant was collected for the determination of biochemical parameters (Yang et al., 2010).
The activities of the antioxidant enzymes, superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) were measured using commercial kits from ZellBio GmbH (Germany) according to the manufacturer's guidelines. Likewise, the metabolic enzyme activities, including alkaline phosphatase (ALP), aspartate aminotransferase (AST), and alanine aminotransferase (ALT), were determined using commercial kits from (Pars Azmun Co., Iran) as specified by the manufacturer.
At the end of the experiment, hemolymph samples were collected from shrimp reared under different treatments. Initially, after a 24-h fasting period, 10 shrimp were randomly selected from each tank and placed in an ice bath at 4°C for 15 minutes to minimize stress and reduce their activity. A sterile 1 mL insulin syringe equipped with a G26 needle preloaded with 0.4 mL of Alzor anticoagulant solution (composed of 27 mM sodium citrate, 336 mM sodium chloride, 9 mM EDTA, and 115 mM glucose, at pH 7) (Jones et al., 2010) was then used for hemolymph collection. Hemolymph was collected from the abdominal sinus region (the first and second swimming legs adjacent to the ventral nerve cord) (Xu and Pan, 2013). The tip of the syringe needle was gently inserted at a 45° oblique angle beneath the chitinous layer, and approximately 0.3 mL of hemolymph was withdrawn from each shrimp; afterwards, the sampling syringes were shaken to mix the hemolymph with the anticoagulant solution. The collected hemolymph samples were stored in separate microtubes in a freezer at –80°C until analysis. The microtubes containing the samples were thawed after being transferred from the –80°C freezer to the laboratory at room temperature (27°C), and then the samples were homogenized using a vortex mixer for 30 seconds prior to being used for subsequent analyses (Liu et al., 2019; Xu et al., 2019). For measuring the biochemical parameters of hemolymph, the samples were placed in a centrifuge at 12,000 rpm at 4°C for 15 minutes to separate the plasma. Then, the upper liquid was separated and used for the analysis of the parameters. Glucose, cholesterol, and triglycerides were measured using a spectrophotometer via a colorimetric method with commercial diagnostic kits (Pars Azmun Co., Iran) following the manufacturer's instructions.
Lysozyme activity was measured using a turbidity assay, with Micrococcus lysodeikticus as the substrate and egg white lysozyme as the standard (Ellis, 1990; Nayak et al., 2008). Phenoloxidase activity was determined using a turbidity assay method following the oxidation of the L-dopa (3,4-dihydroxy-L-phenylalanine) substrate (Sigma) (Söderhäll and Hall, 1984).
All statistical analyses were performed using SPSS version 23. Significant differences between groups were assessed by one-way analysis of variance (ANOVA). Mean differences were compared using Duncan's multiple range tests. For all statistical tests, differences were considered statistically significant at P<0.05. All graphs were drawn using Excel version 2019.
The results of growth performance, shrimp SR, FCR, and FE are presented in Table 2. The results show that with increasing stocking density, BWG (3.16 g), SGR (1.7%/day), SR (88.19%), and FE (44.59%) decreased, with the lowest values observed in the SD4 treatment (with a density of 1.23 g/L), which was significantly different from the other treatments (P<0.05). The most suitable growth performance (BWG, SGR), SR, and FCR were observed up to a stocking density of 0.616 g/L (SD2).
Growth and nutritional performance of P. vannamei reared under different stocking densities in a biofloc system for 5 weeks
| Parameters | SD1 | SD2 | SD3 | SD4 |
|---|---|---|---|---|
| FBW (g) | 9.60±0.53 a | 9.34±0.44 a | 8.25±0.55 b | 7.01±0.66 c |
| BWG (g) | 5.75±0.53 a | 5.49±0.45 a | 4.40±0.55 b | 3.16±0.66 c |
| SGR (%/day) | 2.60±0.16 a | 2.53±0.13 a | 2.17±0.19 b | 1.70±0.27 c |
| FCR | 1.20±0.11 c | 1.27±0.09 c | 1.69±0.22 b | 2.34±0.55 a |
| FE (%) | 83.92±7.73 a | 78.99±6.50 a | 59.88±7.54 b | 44.59±9.29 c |
| SR (%) | 97.22±4.30 a | 95.83±3.72 a | 90.74±1.43 b | 88.19±2.84 c |
Abbreviations: final body weight (FBW), body weight gain (BWG), specific growth rate (SGR), feed conversion ratio (FCR), feed efficiency (FE), survival rate (SR).
Values are expressed as mean ± SD. Values in the same row with different letters are significantly different (P<0.05).
The results of the biochemical parameters (glucose, triglycerides, and cholesterol) are presented in Figure 1. The results show that with increasing stocking density, glucose levels increased (P<0.05), with the highest values being 41.16 mg/dL and 41.83 mg/dL observed in treatments SD3 and SD4, respectively. Stocking density did not have a significant effect on triglyceride and cholesterol levels (P>0.05). Triglyceride levels ranged from 150 to 152.33 mg/dL, while cholesterol levels ranged from 97.66 to 100.66 mg/dL.
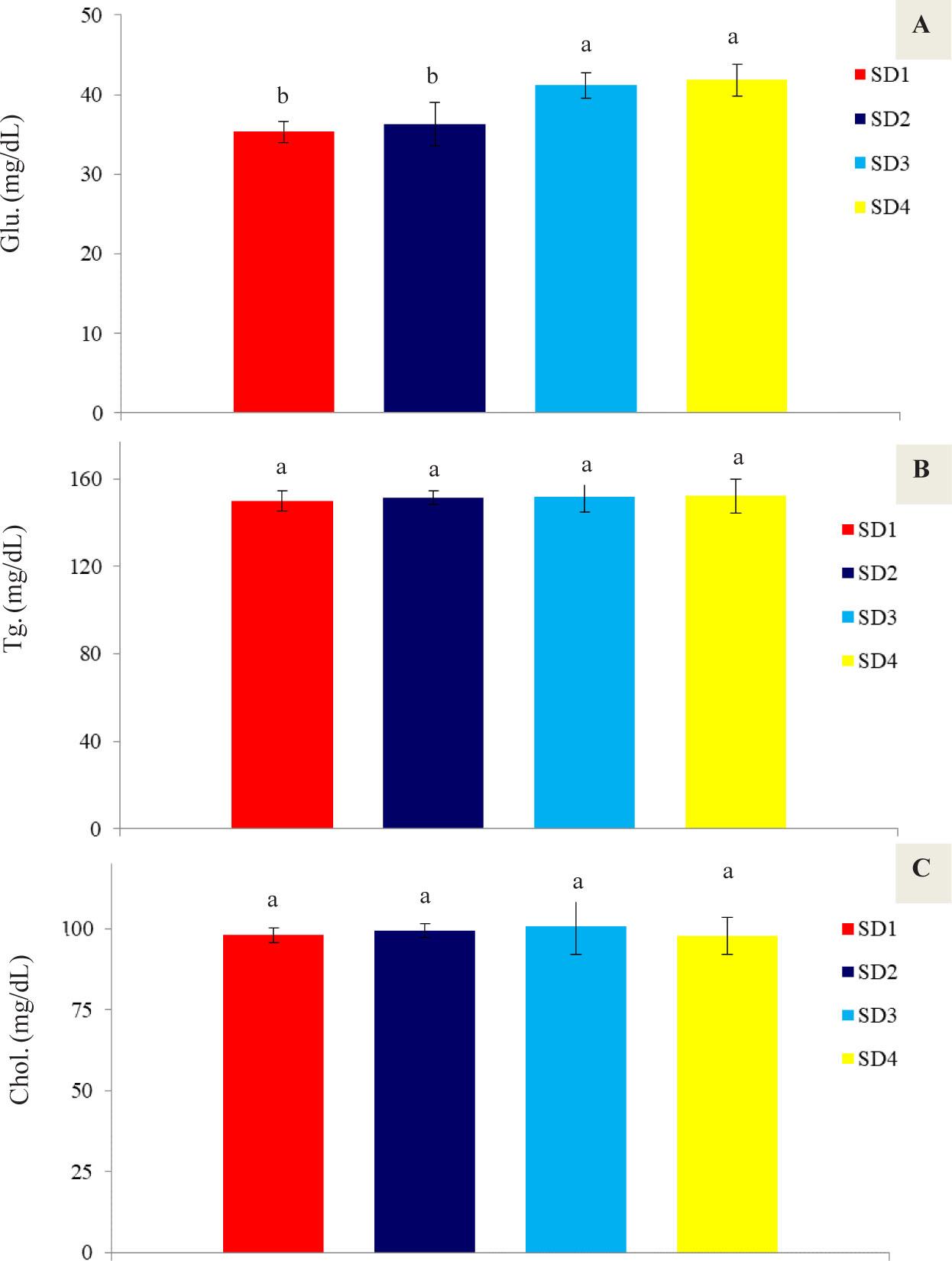
Biochemical activity of P. vannamei hemolymph including glucose (Glu. A), triglyceride (Tg. B), and cholesterol (Chol. C) under the influence of different stocking densities at the end of the experimental period (35 days)
The results of immune activities (lysozyme and phenoloxidase) in shrimp hemolymph are presented in Figure 2. The results show that with increasing stocking density in the biofloc system, immune activity decreased, with the lowest levels of lysozyme (18.33 U/mL/min) and phenoloxidase (0.55 U/mL) observed in treatment SD4 (P<0.05).
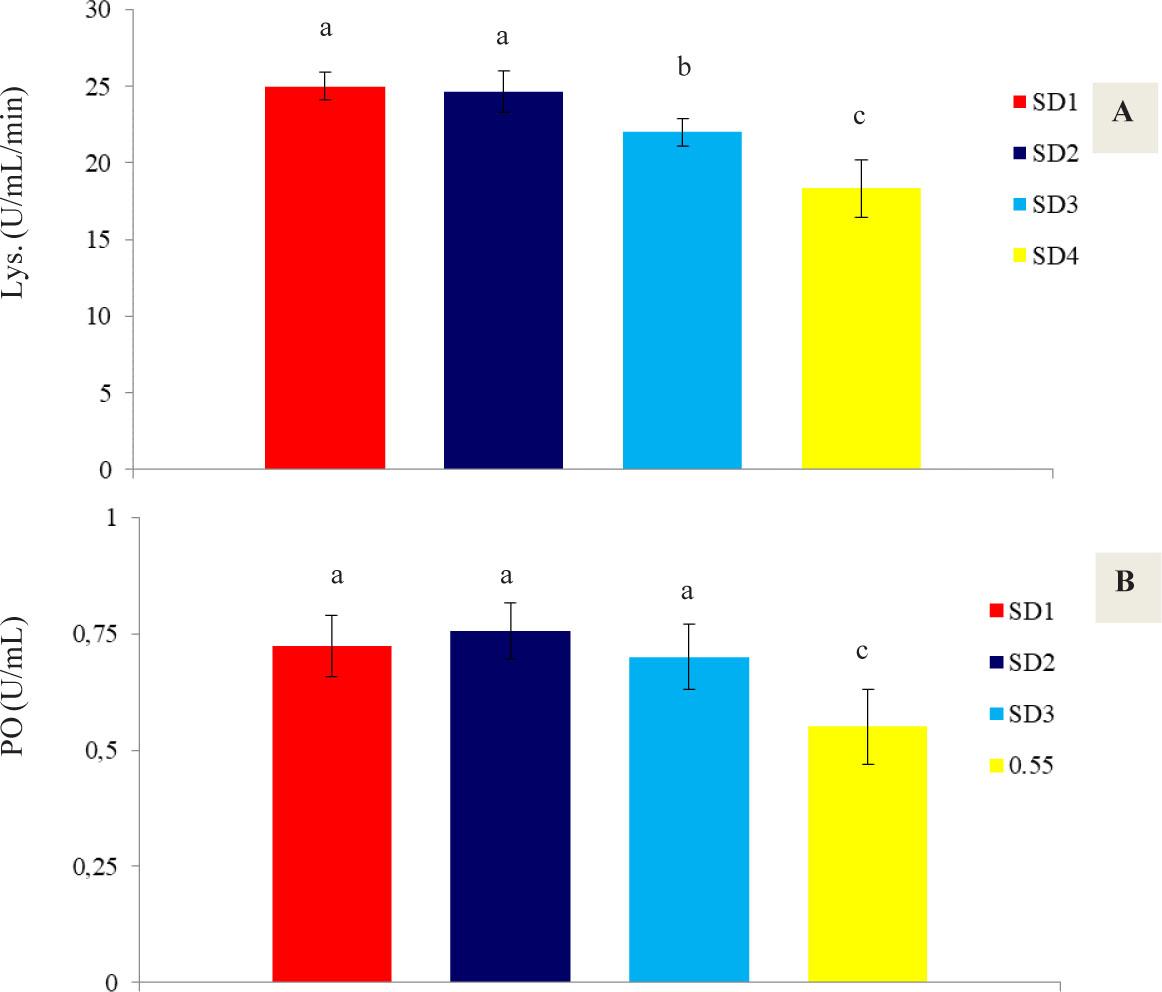
Immunity activities in the hemolymph of P. vannamei including lysozyme (Lys. A), and phenoloxidase (PO, B), under the influence of different stocking densities at the end of the experimental period (35 days)
The results of the metabolic enzymes of the shrimp hepatopancreas are presented in Figure 3. The results indicate that with increasing stocking density, ALP levels decrease, with the lowest value of 46 U/mg protein obtained in treatment SD4, which was significantly different from the other treatments (P<0.05).
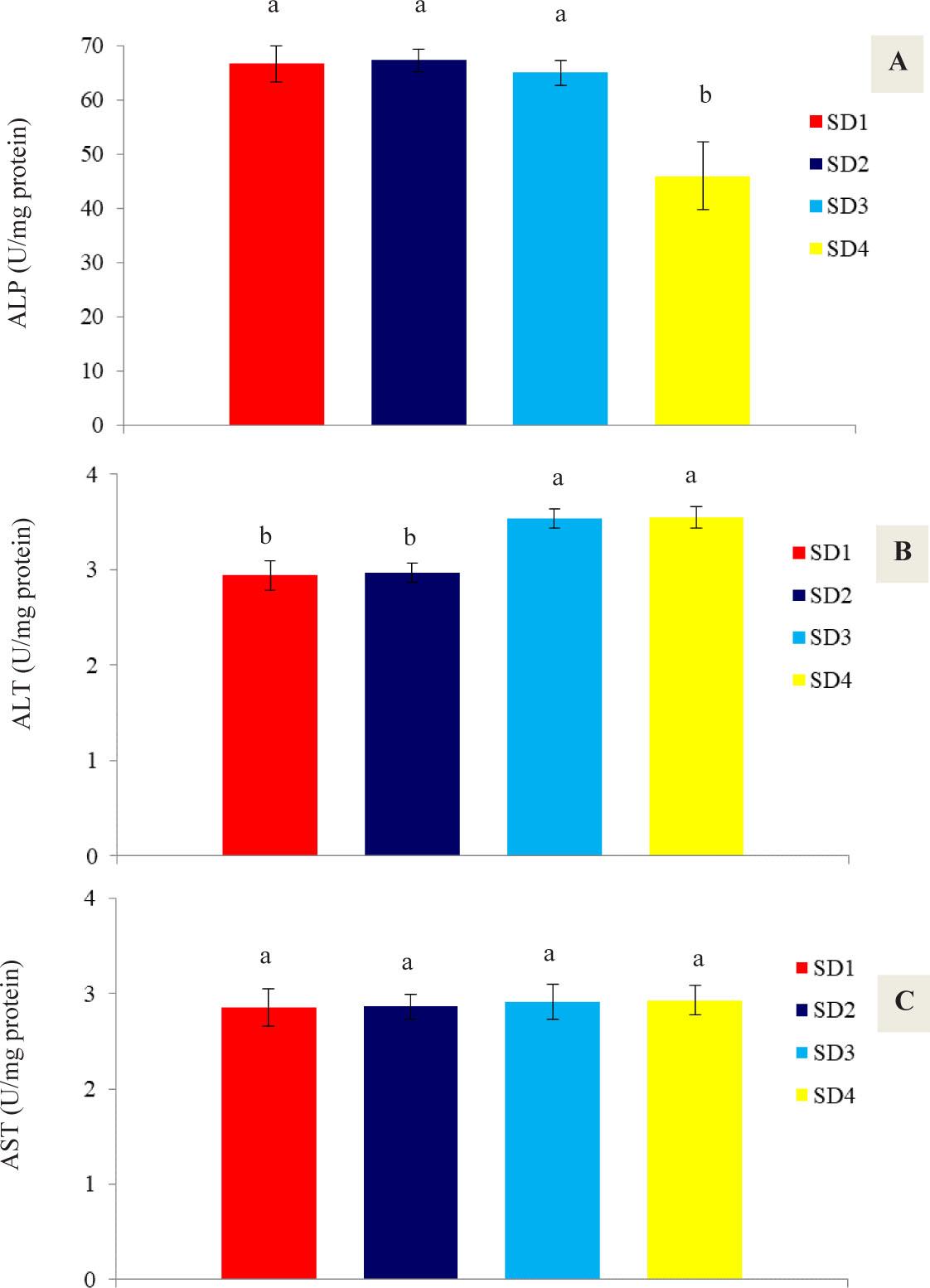
Metabolic enzyme activities (A: ALP, B: ALT, C: AST) in the hepatopancreas of P. vannamei at the end of the experimental period under the influence of different stocking densities at the end of the experimental period (35 days)
ALT levels increased with increasing stocking density, such that the lowest values observed in treatments SD1 and SD2 were 2.94 and 2.97 U/mg protein, respectively, which were significantly different from the other two treatments (P<0.05). No significant differences were observed in AST levels among the treatments, with values ranging from 2.85 U/mg protein (SD1) to 2.93 U/mg protein (SD4) (P>0.05).
The results of the antioxidant enzyme activities (SOD, CAT, GPx) of the shrimp hepatopancreas are presented in Figure 4. The results show that with increasing stocking density in the BFT system, the activity of antioxidant enzymes decreases, with the lowest activities of CAT, GPx, and SOD recorded at 0.4, 3.35, and 3.52 U/mg protein, respectively, in treatment SD4, which was significantly different from the other treatments (P<0.05).
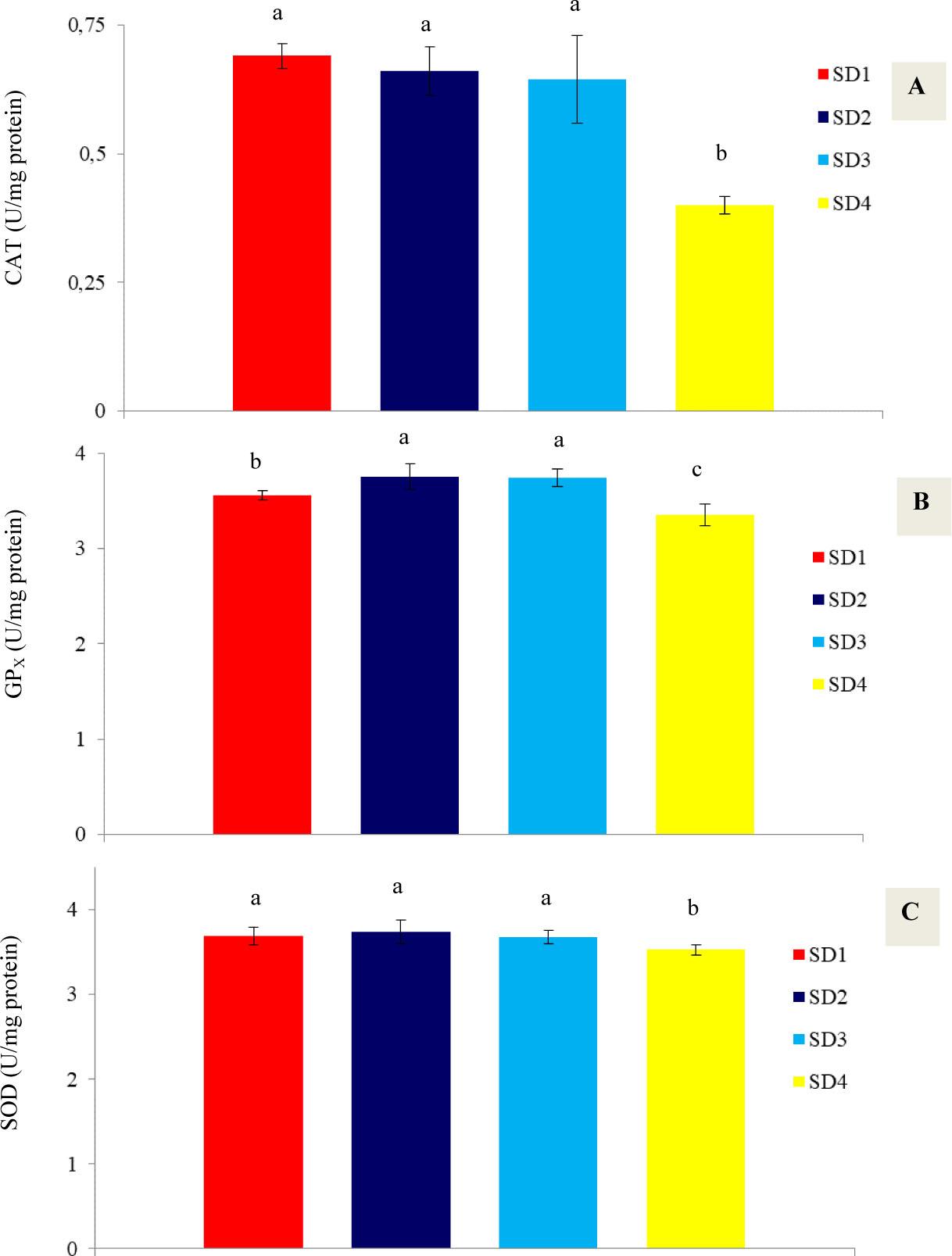
Antioxidant enzyme activities (A: CAT, B: GPx, C: SOD) in the hepatopancreas of P. vannamei at the end of the experimental period under the influence of different stocking densities at the end of the experimental period (35 days)
A reduction in growth is widely regarded as a reliable indicator of chronic stress, and in some species, high density itself acts as a persistent stressor. In the present study, significant differences in growth performance and SR were observed among different densities, with the lowest BWG (3.16), SR (88.19%) and a markedly reduced SGR (1.7%/day) obtained in treatment SD4.
The current results show that higher stocking densities result in lower growth performance and reduced SR compared to lower densities. Previous studies have documented an inverse relationship between stocking density and shrimp growth (Arambul-Muñoz et al., 2019; Khanjani et al., 2022 b; Said et al., 2024). This effect may be attributed to the extra energy expended on food competition, living space, and the stress induced by a larger number of animals (Santos et al., 2021; Adineh et al., 2019; Khanjani et al., 2022 b). Fleckenstein et al. (2020) reported that shrimp in low density treatments (100 shrimp/m3) achieved higher final weights and faster growth rates compared with those in high density treatments (200 shrimp/m3), with the low density group also showing slightly higher SR (91.9–97.2% vs. 90.6–91.8%). Said et al. (2024) reported that a higher SGR was achieved in low density treatments (50 shrimp/m2) compared with high density treatments (200 shrimp/m2). Additionally, the low density group exhibited a slightly higher SR (98.78%) compared to the high density group (96.75%)
Irani et al. (2023) investigated the effects of different stocking densities (100, 200, 300, and 400 shrimp/m3) of Pacific white shrimp in a BFT system. Their findings consistently showed that the highest FBW of 17.42 g was achieved at a density of 100 shrimp/m3, while the lowest SR of 87.92% was recorded at 400 shrimp/m3.
In this study, growth performance was optimal in the low density treatments (SD1 and SD2). In contrast, the medium (SD3) and high density (SD4) treatments exhibited lower FBWs (8.25±0.55 g and 7.01±0.66 g, respectively), indicating that increased stocking density impaired the growth of Pacific white shrimp. Additionally, variations in growth performance can be attributed to differences in the sources of the same species and the experimental conditions (Xu and Pan, 2012; Liu et al., 2017).
The cultivation of Pacific white shrimp in freshwater at three different stocking densities (90, 130, and 180 individuals/m2) showed that both the SGR and SR of the shrimp decreased with increasing density (Araneda et al., 2008). Similar findings were obtained in studies on the culture of the crab Eriocheir sinensis (Li et al., 2007) and the shrimp Palaemonetes sinensis (Dong et al., 2018), with both SGR and SR declining significantly as density increased. The findings of the present study further confirmed these results, showing that the FBW of the shrimp decreased with increasing stocking density and reached its lowest value in the treatment at 1.23 g/L.
In the present study, with increasing stocking density in the biofloc aquaculture system, the FCR increased, with the highest value (2.34) observed in treatment SD4. In the study by Bajracharya et al. (2025), increasing shrimp stocking density in an outdoor recirculating mixotrophic system led to an increased FCR, with values of 0.8, 0.9, 1.1, 1.1, and 1.5 obtained at densities of 50, 100, 200, 300, and 400 shrimp/m3, respectively.
Fleckenstein et al. (2020) observed that biofloc treatments at a low (2.3 kg/m3) and high (4.0 kg/m3) densities of exhibited FCRs between 1.1 and 1.3. In our study, FE was notably higher in the low density treatments (SD1 and SD2) compared to the high density ones (SD3 and SD4). This improvement in feed utilization may be due to the biofloc communities boosting digestive enzyme activity, thereby enhancing nutrient absorption and reducing FCR values (Becerra-Dórame et al., 2012). Nguyen et al. (2019), Vungarala et al. (2021), Said et al. (2024), and Bajracharya et al. (2025) found that higher stocking densities of P. vannamei are associated with increased FCR values. Moreover, improved feed utilization in lower density systems suggests that at elevated densities, shrimp may have a diminished capacity to graze on the bioflocs (Wasielesky et al., 2013).
In the present study, no significant differences in triglyceride levels were observed across different stocking densities. Guemez-Sorhouet et al. (2019) reported that hemolymph triglyceride levels in biofloc treatments did not significantly differ from those in the control groups. In the biofloc treatments, triglyceride levels of 36.1, 36.6, and 32.8 mg/dL were observed at stocking densities of 300, 600, and 900 PL/m3, respectively. Studies have shown that increased stress leads to reduced hemolymph triglyceride levels in Pacific white shrimp (Mercier et al., 2006).
Cholesterol in shrimp forms an integral part of the cell membranes and intracellular structures, serving as a precursor for steroid hormones and molting hormones, such as ecdysone. Cholesterol concentration is considered a key indicator of lipid metabolism in shrimp, as lipids are transported via the hemolymph (Shan et al., 2019). In the present study, no significant differences in cholesterol levels were observed among the different stocking density treatments in the biofloc aquaculture system. Martinez-Porchas et al. (2020) reported a significant decrease in hemolymph cholesterol in Pacific white shrimp reared in biofloc systems, whereas Hussain et al. (2021) found a significant increase. Glucose is released into the plasma via glycogenolysis, a process that primarily takes place in the hepatopancreas, muscles, and blood cells. Variations in hemolymph glucose levels are typically observed when shrimp experience stress (Yu et al., 2024). In the present study, an increasing trend in hemolymph glucose levels was observed in Pacific white shrimp as stocking density in the BFT system increased.
In many studies, the biofloc environment under various conditions did not show significant changes in shrimp glucose levels; however, in some cases, significant increases or decreases were observed (Martinez-Porchas et al., 2020; Guemez-Sorhouet et al., 2019; Kumar et al., 2017). Consequently, it can be concluded that increasing stocking density in aquaculture systems up to a certain threshold may alleviate rearing stress in Pacific white shrimp by promoting group behaviors (Bardera et al., 2019).
Shrimp, like other crustaceans, lack a specialized immune system and rely on various mechanisms associated with innate immunity to protect themselves from adverse environmental conditions (Bateman, 2021). In recent years, several researchers have demonstrated that BFT systems with limited water exchange are capable of enhancing the innate immunity of Pacific white shrimp (Kim et al., 2014; Liu et al., 2017; Khanjani et al., 2023).
Lysozyme is integral to the host's defense system, acting as a key element of innate immunity by neutralizing invading pathogens. It effectively eliminates both gram-positive and gram-negative bacteria (Shailesh and Sahoo, 2008).
In this study, phenoloxidase and lysozyme activities were reduced in the high density treatment compared to the low density treatment (Figure 2 A, B). Similar reductions in lysozyme activity have been reported in common carp (Cyprinus carpio) (Yin et al., 1995) and in P. vannamei (Liu et al., 2017) under overcrowding stress.
Phenoloxidase is a key enzyme in the shrimp immune defense system. It is activated through a stepwise process when an invading pathogen is detected, ultimately leading to the encapsulation of the pathogen via melanogenesis (Fan et al., 2011). Phenoloxidase also plays a pivotal role in controlling the hemolymph bacterial load (Fagutao et al., 2009), recognizing non-self agents, and defending against pathogenic bacteria (Amparyup et al., 2009). Phenoloxidase activity can serve as a health indicator since changes in this parameter are directly linked to disease status and environmental alterations. In the present study, the lowest level of phenoloxidase activity was observed in the highest stocking density treatment, which likely indicates that the shrimp in this group are more sensitive to disease, environmental changes, etc. compared to those in other treatments.
In the study by Lin et al. (2015), white shrimp maintained at high densities displayed a marked reduction in their ability to resist pathogens. This compromised immunity was evidenced by a significant decline in several key immune parameters, lower hemocyte counts, reduced phenoloxidase activity, a diminished respiratory burst, decreased activities of SOD, and lysozyme. These findings suggest that high stocking densities can severely impair the immune defenses of white shrimp, making them more vulnerable to disease challenges.
Metabolic enzymes (ALP, ALT, and AST) are generally regarded as indicators of hepatopancreatic function or damage in shrimp, thereby reflecting the overall health and immune status of aquatic organisms (Gyan et al., 2020). In shrimp, ALT and AST contribute to amino acid metabolism and are frequently employed as diagnostic markers for hepatic injury (Yu et al., 2024). ALP, a vital detoxification enzyme in many aquatic species including shrimp, plays a critical role in calcium absorption and metabolism, chitin secretion and synthesis, and also serves as an indicator of lysosomal enzyme activity (Yu et al., 2024).
In the present study, the levels of metabolic enzymes ALP, ALT, and AST were affected by an increase in stocking density, with ALP decreasing, ALT increasing, and AST remaining unchanged. Some studies have shown that the ALP enzyme is associated with the immune activities of cultured aquatic organisms (Khanjani et al., 2023), which may indicate decreased immune function at higher stocking densities.
In the study by Hussain et al. (2021), a significant reduction in ALT in the hemolymph of P. vannamei cultured in a BFT system was reported, and they argued that this decrease indicated improved hepatopancreatic performance in shrimp reared in the BFT system. In the present study, shrimp reared at higher stocking densities exhibited elevated hemolymph levels of ALT and AST, indicating that increased stocking density leads to damage in the hepatopancreatic tissue of shrimp and adversely affects their health status.
Antioxidants, such as superoxide dismutase (SOD), are essential enzymes that protect organisms from oxidative stress, and an organism's overall antioxidant capacity is closely linked to its health (Parrilla-Taylor and Zenteno-Savín, 2011). Enzymes like SOD and GPx form the primary enzymatic defense against free radicals. When their levels decrease, free radical concentrations rise, leading to impaired cellular function (Khanjani et al., 2023).
In the present study, the activity of the hepatopancreatic antioxidant enzymes (GPX, SOD, and CAT) decreased with increasing stocking density, with the lowest levels of CAT (0.4), SOD (3.52), and GPX (3.53 U/mg protein) recorded in the SD4 treatment.
In BFT systems, conditions positively enhance the antioxidant activity in shrimp (Khanjani et al., 2023). Liu et al. (2017) reported that both immunological response parameters and antioxidants including SOD, GPx, and malondialdehyde declined under higher density conditions (300, 400, and 500 shrimp/m3). According to Said et al. (2024), the levels of key antioxidant enzymes, including CAT, GPX, and SOD, in the hemolymph of P. vannamei were reduced when cultured at high density (200 shrimp/m2). These results suggest that high stocking density conditions in BFT aquaculture promote a weaker immunological and antioxidant defense.
Determining the optimal stocking density is crucial for maximizing shrimp production efficiency, and our study provides valuable insights that can directly influence commercial shrimp aquaculture practices. The findings of the present research demonstrated that increasing the density of juvenile Pacific white shrimp in a BFT system significantly affected growth, SR, FCR, hemolymph immune indices, as well as the metabolic and antioxidant enzymes of the shrimp hepatopancreas. Increased stocking density resulted in reduced FBW, SR, and FE, with the highest FCR observed at a density of 1.23 g/L. Furthermore, higher stocking density led to decreased immune activities (lysozyme, phenoloxidase) in the hemolymph and lower antioxidant enzyme activities (SOD, CAT, GPX) in the shrimp hepatopancreas. Under our experimental conditions, it is recommended that a stocking density of juvenile shrimp up to 0.616 g/L in the BFT system results in increased FBW, improved SR, and enhanced immune and antioxidant activities.