The urinary tract, traditionally considered sterile in the absence of infection, is now recognized as a complex ecosystem inhabited by diverse microbial communities. Known as the urinary tract microbiota (urobiota), these microbial communities play a pivotal role in maintaining urological health and preventing diseases. The discovery of the urobiota has revolutionized our understanding of urological health, revealing that the balance of bacteria, fungi, viruses, and archaea in the urinary tract is crucial for its normal functioning and defense against pathogens (Perez-Carrasco et al. 2021; Colella et al. 2023). The establishment of the urinary microbiota begins shortly after birth, influenced initially by maternal microbiota and later shaped by factors such as diet and antibiotic use (Storm et al. 2022). Advancements in next-generation sequencing (NGS) technologies have propelled the study of the urinary tract microbiota, allowing for the identification of microbial communities without the need for culture (Heidrich et al. 2022). These technologies have uncovered a complex and variable microbiota among individuals, underscoring the personalized nature of the urinary tract microbiota (Colella et al. 2023). Significant differences exist between male and female urinary microbiota, attributable to anatomical, physiological, hormonal, and lifestyle factors (Perez-Carrasco et al. 2021). The shorter female urethra’s proximity to the anus facilitates bacterial transfer, potentially leading to a more diverse microbiota and higher urinary tract infection (UTI) risk (Foxman 2002). Hormonal fluctuations in females, especially estrogen, influence the urinary microbiota by affecting Lactobacillus spp. abundance (Amabebe and Anumba 2018), whereas the male urinary tract’s stability is influenced by testosterone (Hold et al. 2014). Additionally, the secretions of male prostate can influence the microbial environment by providing nutrients to certain bacteria or by containing antimicrobial compounds that shape the microbial community (Nickel et al. 2008; Lewis et al. 2013). Lifestyle factors such as diet, personal hygiene, physical activity, and sexual activity significantly impact the urinary tract microbiota. Diet can influence microbial growth, while hygiene practices can prevent pathogenic overgrowth or may cause dysbiosis (Reid et al. 2003; Howell 2007; Nicole 2014). Regular physical activity benefits the microbiota by altering immune and hormonal responses (Monda et al. 2017). Sexual activity introduces external microorganisms, affecting the microbial balance and increasing UTI risk, particularly in women (Foxman 2002; Toh et al. 2023). The urobiota may provide valuable biomarkers for early detection and diagnosis of urological conditions, offering more nuanced insights than traditional methods that rely on symptoms or specific pathogens (De Maio et al. 2023; Jirillo et al. 2024). Changes in the urobiota could signal disease onset, such as UTI or interstitial cystitis, before symptoms appear. Analyzing the urinary microbiota may help understand the mechanisms behind conditions like bladder cancer and chronic pelvic pain syndrome, enabling personalized diagnostic approaches (De Maio et al. 2023; Jirillo et al. 2024). Therapeutically, research on the urobiota is exploring microbiota-based treatments, probiotics, and prebiotics to restore microbial balance and prevent or treat urological conditions (Reid et al. 2003). The potential for urobiota transplants, dietary interventions, and understanding drug-microbiota interactions could enhance treatment efficacy and reduce side effects, emphasizing the role of a healthy microbiota in urological health (Tariq et al. 2017; Magruder et al. 2019; Chen et al. 2022; Zhao et al. 2023). The aim of our study was to preliminarily characterize the urobiota of asymptomatic, presumably healthy young adult males in Poland. The justification for taking up the study problem is the very limited amount of data data describing the composition of the healthy urinary tract microbiota and the lack of a similar research in Polish population. The results of the pilot study will be used to design further experiments in the field of discovering the human urobiota and its impact on health and disease.
The study group consisted of 40 young (aged 18–40) asymptomatic, presumably healthy men (no active infectious disease, no history of UTI, no chronic diseases, and no antimicrobial therapy in the six months before the study). Candidates were sought from among all interested male adults. Information about the possibility of participating in the study (along with basic information) was propagated via the Medical University of Lodz websites and social media. Participants were qualified by the physician at the individual meetings. Participants were provided with all the details of the course of the study and were provided with answers to any questions they may have. Then, after signing the written consent of participation, they were included in the study group. Participants had the option to opt out of the study at any stage without incurring adverse consequences. Participation in the study involved self-completing a lifestyle questionnaire and self-donating a urine sample, using the “mid-stream” method into a sterile container. Participants received detailed instructions with illustrations describing how to properly collect a male “mid-stream” urine sample. Samples were collected at the participants’ home from the first morning void and transported to the laboratory within 2 hours. Data obtained from participants were anonymized through unique randomly generated codes. The biological material was processed in a microbiology laboratory according to the methods described below. In order to ensure the good ethical practice of research in the project and to meet the legal requirements of the medical experiment, the approval of the Bioethical Commission at the Medical University of Lodz was obtained. Furthermore, it was ensured that all activities undertaken during this research project were consistent with the set of rules and guidelines “Good manners in science” (Pol. “Dobre obyczaje w nauce”), developed by the Ethics Committee in Science of the Polish Academy of Sciences.
The collected urine samples were analyzed microbiologically with expanded quantitative urine culture (EQUC), following the Hilt et al. (2014) protocol. The urine culture was done by quantitatively spreading 0.1 ml of urine onto Blood Agar plates, Chocolate Agar plates, Colistin & Nalidixic Acid Blood Agar plates, and Anaerobe Blood Agar plates (Thermo Fisher Scientific, USA) before incubation for 48 hours at various temperatures and atmospheric conditions (see Figure 1). The detection threshold was 10 colony-forming units (CFU)/ml, which corresponds to one colony of growth on any plate (Price et al. 2016; Price et al. 2020a). Each morphologically distinct colony type was isolated on a different plate of the same media to prepare a pure culture for organism identification. The matrix-assisted laser desorption ionization time-of-flight mass spectrometry was used with VITEK MS (bioMérieux, France) to identify the bacterial isolates.

Summary of plates and conditions that were used for culture (plates: BA – Blood Agar Plate, CHA – Chocolate Agar, CNA – Colistin & Nalidixid Acid Agar, ABA – Anaerobe Blood Agar).
Statistical analysis was performed using Statistica 13 software (TIBCO Software Inc., USA). The distribution of collected data was checked using the Shapiro–Wilk test. All variables were distributed non-normally. The correlations were checked using Spearman’s test. A p-value of 0.05 was considered the limit of statistical significance. Descriptive statistics were prepared using Microsoft Excel 2019 software (Microsoft Corporation, USA).
The characteristics of study participants are presented in Table 1. The raw data from cultures presented in this study is available in the Supplementary Material. Bacterial growth was observed in 88% (n = 35) of the samples collected, and 12% (n = 5) of participants’ samples were below the level of detection. The bacterial count in the positive samples ranged from 10 to 104 CFU/mL, and the diversity ranged from 2 to 16 various species identified per sample. The most common type of bacteria phylum was Firmicutes, and the most prevalent genera were Streptococcus, Staphylococcus, Corynebacterium, Micrococcus, Enterococcus, and Winkia. The collected data allowed us to define three distinctive urotypes (more than 50% bacteria belonged to one genus), in which the following genera predominated: Streptococcus, Staphylococcus, and Corynebacterium. We also identified samples with Actinotignum, Microbacterium, Aerococcus, Peptoniphilus, and Enterococcus urotypes, but they were only with one representative so we excluded them from group analyses. The compositions of positive test samples at the genus level are shown in Figure 2. Figures 3–7 present the results of examined group comparisons depending on sexual activity, sexual behaviors, and physical activity time. None of these comparisons reached statistical significance but visible trends suggest potential associations worth further exploration. The lack of significance may stem from the limited sample size within subgroups, which reduces the statistical power to detect subtle but biologically relevant differences.
Characteristics of study participants.
| Male | n = 40 (100%) |
| Age | 20–36 years old (mean = 24) |
| Sexually active | n = 30 (75%) |
| Sexual behavior | |
| - vaginal | n = 19 (63%) |
| - oral | n = 27 (90%) |
| - anal | n = 11 (36%) |
| Physically active | n = 17 (43%) |
| Physical activity time | 20–120 min daily (mean =55) |
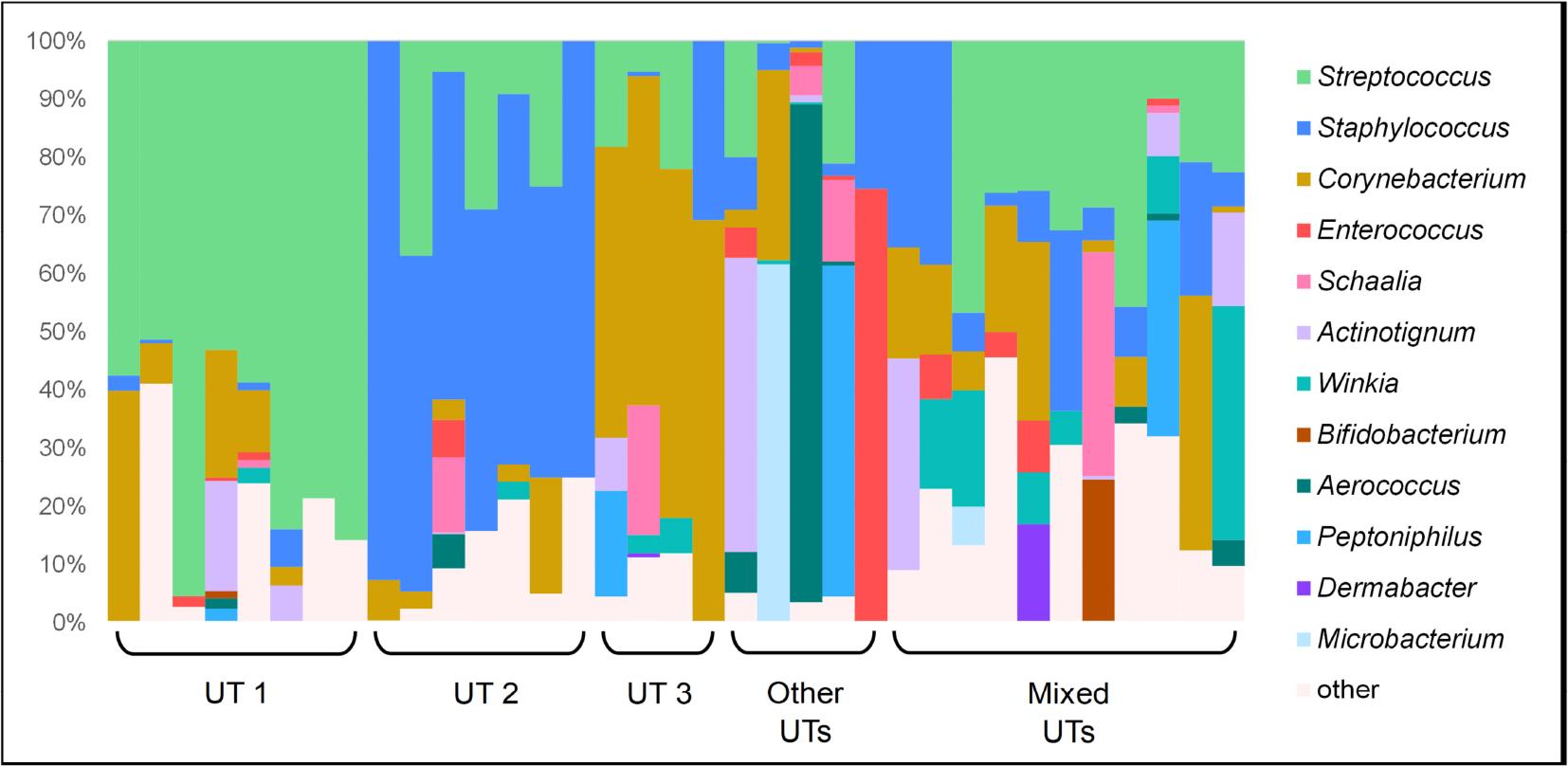
Graphical presentation of urinary microbiota assessed by expanded quantitative urine culture (EQUC) among examined males (n = 35). Microbiota profiles are shown as stacked bar graphs depicting the relative abundance of various genera. Defined urotypes (UTs) were: UT 1 – Streptococcus spp., UT 2 – Staphylococcus spp., UT 3 – Corynebacterium spp. Other UTs – other genus predominance observed in one sample only, Mixed UTs – no predominant genus detected.
Our research aimed at exploring the microbial diversity within the urinary tract of asymptomatic, presumably healthy young men in Poland, provides new insights into the urobiota’s composition and its clinical implications. The results demonstrated a significant microbial diversity, contradicting the long-held belief of the urinary tract’s sterility in healthy individuals.
We found that 88% of the urinary samples showed bacterial growth, indicating a rich microbial presence even in the absence of symptoms. Our findings were consistent with the literature, where similar genera were frequently observed in the urobiota of healthy male individuals (Perez-Carrasco et al. 2021). The diversity observed, with some samples showing up to 16 different bacterial species, underscores the complexity of the urinary microbiome in men – however, it should be remembered that our results are based only on EQUC. This bacterial diversity is significant, as it may play a role in both maintaining urinary tract health and in the pathogenesis of diseases. For instance, Streptococcus and Staphylococcus genera are known for their role in both health and disease states, participating in biofilm formation and potentially inhibiting the growth of pathogenic bacteria (Perez-Carrasco et al. 2021; Colella et al. 2023).
Kogan et al. (2015) highlighted a similar diversity in their study, which noted the presence of both facultative aerobic and nonclostridial anaerobic bacteria in the urine of healthy individuals. They reported that the urinary tract harbors a complex mix of bacteria, which may contribute to local immunity and metabolic processes, suggesting a protective role against pathogens. Modena et al. (2017) further supported these findings by identifying significant changes in the urinary microbiome following kidney transplants, which correlated with clinical outcomes. Their study underscored the impact of microbiome alterations on the immune response and transplant rejection, suggesting that a stable urobiota might play a role in preventing immune-mediated tissue injuries.
Our results indicated variations in the urobiota linked to different sexual behaviors, which aligns with findings by Fouts et al. (2012), Gottschick et al. (2017), and Price et al. (2020b) who suggested that sexual activity can influence microbial communities in the urinary tract. These variations could potentially affect the host’s susceptibility to infections or other urological conditions.
Understanding the composition and role of urobiota in healthy individuals is crucial for developing new diagnostic and therapeutic strategies. The presence of a diverse and dynamic urobiota should challenge the current clinical approach to UTIs, highlighting the need for more nuanced diagnostics that differentiate between commensal and pathogenic bacteria. The presence of a diverse and dynamic urobiota might be important for maintaining urinary health. In a study by Bajic et al. (2020), men with lower urobiota diversity were more likely to report genitourinary symptoms, suggesting a potential protective role of commensal urinary microbes.
An interesting finding is the detection of Enterococcus spp. in 12 urine samples (34% of all positive samples) from asymptomatic young men. Enterococcus spp. are frequently identified in urine cultures, often leading to diagnostic challenges in clinical settings. While these bacteria are less commonly the etiological agents in UTIs, their presence can significantly affect urine culture results, potentially leading to false-positive interpretations of infection. Our recent epidemiological analysis (Bielec et al. 2023) has highlighted the prevalence and resistance patterns of Enterococcus spp. in UTIs, noting that their presence is often over-represented due to their resilience to common preservatives used in urine sample collection. This resilience can lead to overgrowth in stored samples, potentially masking or mimicking the presence of true pathogens – it can lead to unnecessary antibiotic treatment, particularly when it is not the cause of symptoms but rather a commensal.
Sexual behavior has been ignored in most human microbiome studies. There are limited data exploring what constitutes optimal urogenital microbiota in men. Similar to our data on bladder microbiota concerning the influence of sexual behavior could not be found in the available literature, so we decided to discuss the results with published data focused on the urethral microbiota.
Our study revealed that sexual activity tended to impact the composition of the bladder microbiota. We observed that sexually active individuals had more diverse microbiota compared to those who were less active or abstinent. This diversity was particularly noted in the presence of bacteria genera such as Streptococcus, Staphylococcus, and Corynebacterium, which were prevalent across different sexual behaviors including vaginal, oral, and anal intercourse.
Sexual activity appears to influence the diversity of urobiota. Toh et al. (2023) demonstrated that men engaging in vaginal and anal sex had significantly altered urobiota profiles, including higher abundance of Lactobacillus and Prevotella (traditionally associated with the female reproductive tract), compared to abstinent men.
Moreover, studies by Mores et al. (2021) and Wetmore et al. (2011) discuss how demographic and behavioral factors correlate with the presence of certain pathogens in the urethra, supporting our observations that sexual behavior influences microbial diversity and pathogen presence in the urinary tract. These studies collectively underline the significant impact of sexual behavior on the male urogenital microbiota, both in the bladder and urethra. Due to the small study group and lack of statistical significance, these results require additional confirmation.
The findings highlight the importance of considering sexual history and practices in the clinical assessment of urinary tract health and microbiota-related studies. The relationship between sexual activity and the diversity of the urobiota opens new possibilities for understanding the etiology of UTIs and other urogenital diseases, potentially leading to more targeted therapy that consider individual behavioral profile. While the differences between subgroups did not reach statistical significance, graphical trends observed in Figures 3–7 may indicate underlying patterns in urobiota composition associated with lifestyle factors such as sexual activity and physical activity. Given the small number of individuals in each behavioral subgroup, these findings should be interpreted with caution. However, they may inform hypotheses for future research and guide the design of larger-scale studies. If confirmed in broader cohorts, such exploratory results could eventually contribute to personalized approaches in urinary health management.
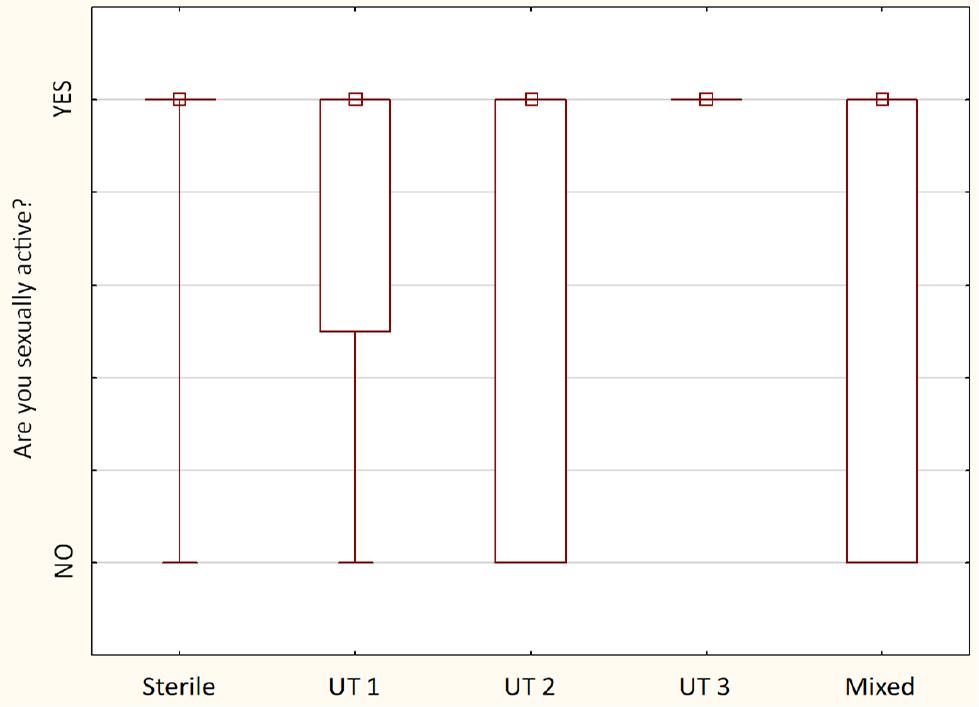
Comparison of sexual activity between detected urotypes among examined males (n = 40) – the squares show medians; the boxes show interquartile ranges; and the whiskers show total ranges. Legend: Sterile – no bacteria cultured, UT 1 – Streptococcus spp. predominance, UT 2 – Staphylococcus spp. predominance, UT 3 – Corynebacterium spp. predominance, Mixed – no predominant genus detected. Differences between groups were not statistically significant.
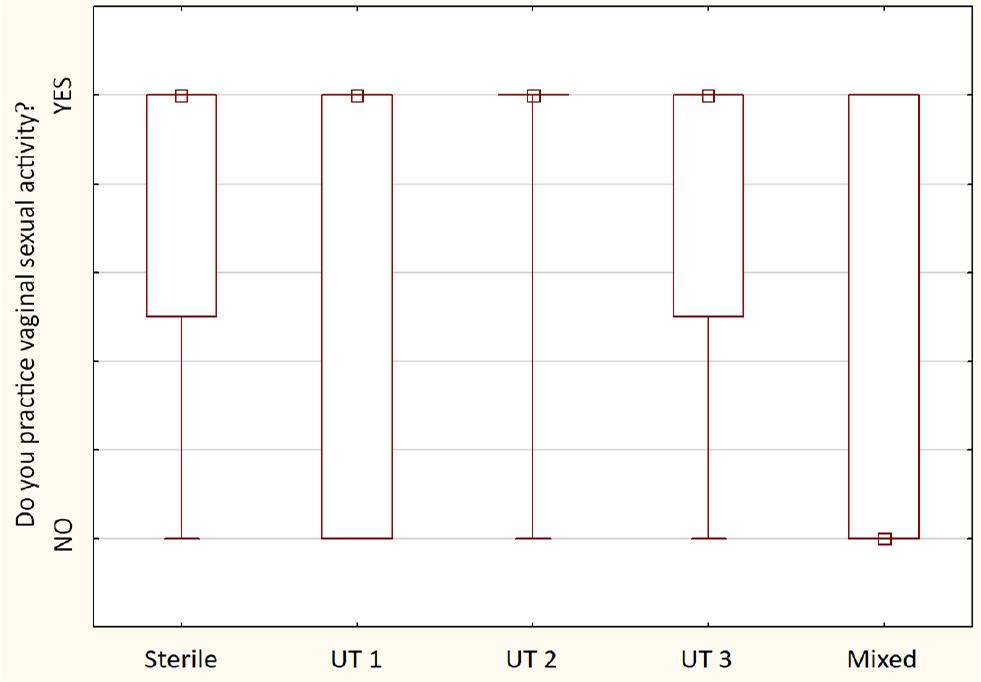
Comparison of vaginal sexual behavior between detected urotypes between examined sexually active males (n = 30) – the squares show medians; the boxes show interquartile ranges; and the whiskers show total ranges. Legend: Sterile – no bacteria cultured, UT 1 – Streptococcus spp. predominance, UT 2 – Staphylococcus spp. predominance, UT 3 – Corynebacterium spp. predominance, Mixed – no predominant genus detected. Differences between groups were not statistically significant.
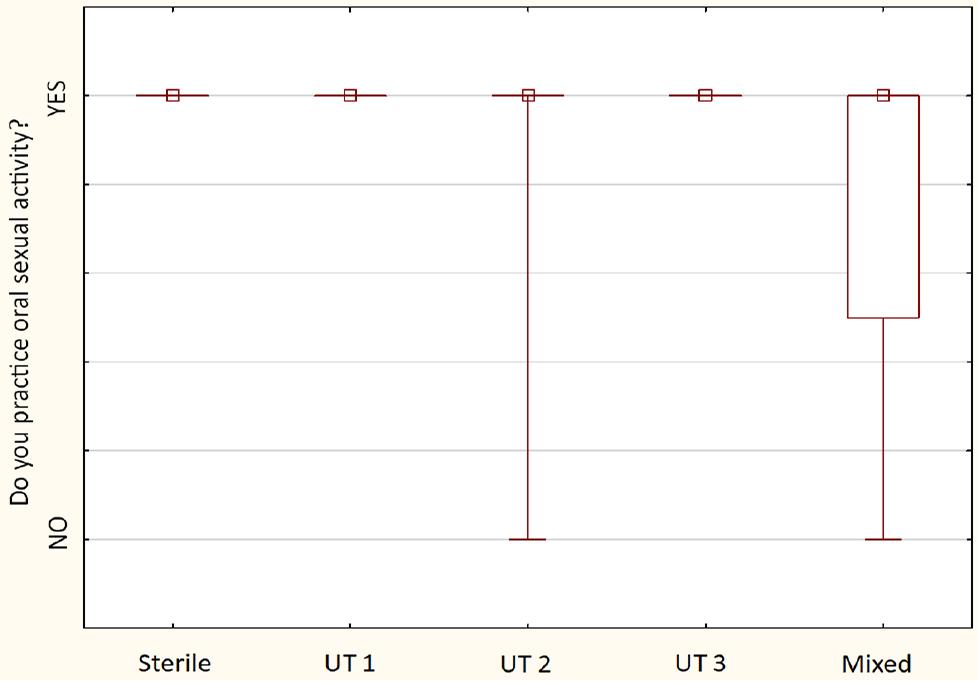
Comparison of oral sexual behavior between detected urotypes between examined sexually active males (n = 30) – the squares show medians; the boxes show interquartile ranges; and the whiskers show total ranges. Legend: Sterile – no bacteria cultured, UT 1 – Streptococcus spp. predominance, UT 2 – Staphylococcus spp. predominance, UT 3 – Corynebacterium spp. predominance, Mixed – no predominant genus detected. Differences between groups were not statistically significant.
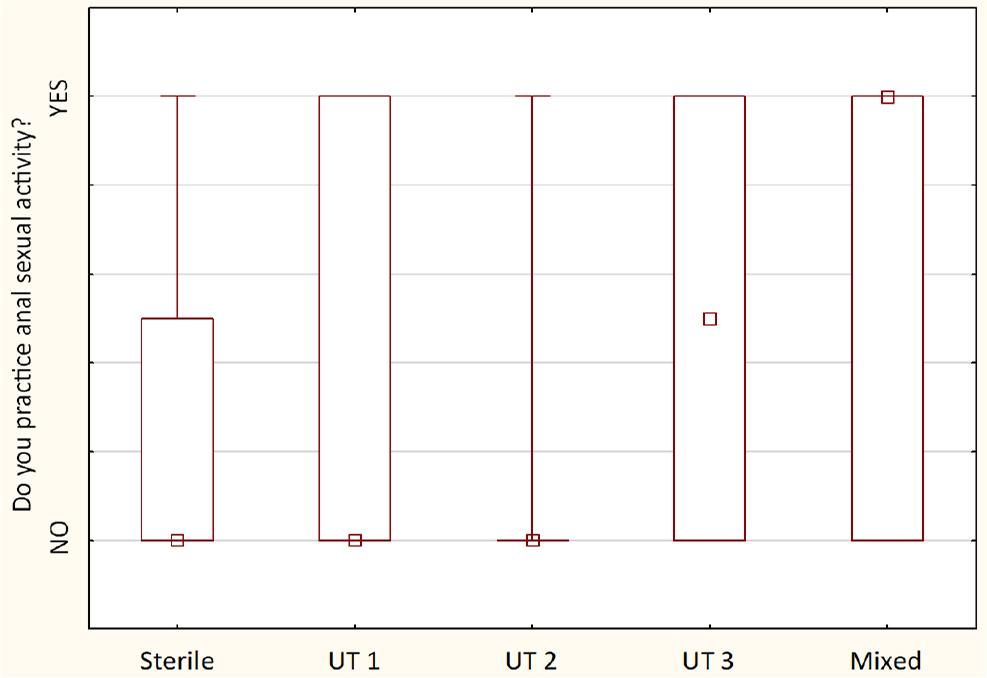
Comparison of anal sexual behavior between detected urotypes between examined sexually active males (n = 30) – the squares show medians; the boxes show interquartile ranges; and the whiskers show total ranges. Legend: Sterile – no bacteria cultured, UT 1 – Streptococcus spp. predominance, UT 2 – Staphylococcus spp. predominance, UT 3 – Corynebacterium spp. predominance, Mixed – no predominant genus detected. Differences between groups were not statistically significant.
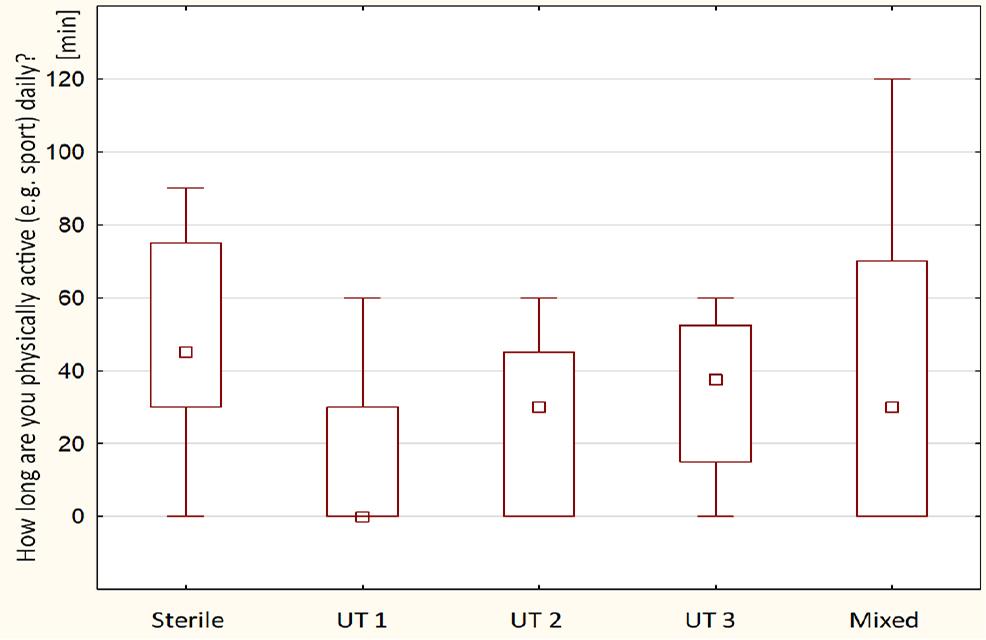
Comparison of physical activity time between detected urotypes among examined males (n = 40) – the squares show medians; the boxes show interquartile ranges; and the whiskers show total ranges. Legend: Sterile – no bacteria cultured, UT 1 – Streptococcus spp. predominance, UT 2 – Staphylococcus spp. predominance, UT 3 – Corynebacterium spp. predominance, Mixed – no predominant genus detected. Differences between groups were not statistically significant.
The obvious weakness of our study is the sampling method. The participants’ self-collection of “midstream” urine may have raised doubts as to whether the cultured bacteria came from the bladder or whether they were contamination from urethra, meatus or skin, rinsed off during urination. The most reliable method of collecting a urine sample for microbiological testing is suprapubic puncture (Perez-Carrasco et al. 2021). However, we abandoned this method for two reasons – (1) it would be doubtful to obtain the consent of the Bioethics Committee for a cognitive examination carrying the risk of an invasive procedure for participants; (2) it would be very difficult to recruit volunteers to participate in such a study. We minimized the risk of contamination by properly educating participants on how to properly collect the sample.
Another important methodological limitation of our study is exclusive reliance on the EQUC technique. Although EQUC represents a significant improvement over standard culture methods by enabling the detection of fastidious and anaerobic bacteria through diverse media and incubation conditions (Hilt et al. 2014), it remains inherently culture-dependent. As such, it fails to capture the full spectrum of microbial diversity, particularly taxa that are unculturable or require highly specific growth conditions. Recent studies employing NGS have consistently demonstrated a much broader microbial landscape in the urinary tract (Gottschick et al. 2017; Heidrich et al. 2022). Nevertheless, the choice of EQUC in this pilot study was justified by several pragmatic and clinical considerations. EQUC is cost-effective, technically accessible, and more easily transferable to routine microbiological diagnostics in clinical settings. Furthermore, it allows for direct recovery of viable organisms, which is essential for future phenotypic testing, antibiotic resistance profiling, or potential probiotic development. Despite its limitations, EQUC provides a relevant and practical tool for establishing a baseline understanding of cultivable urinary tract microbiota, especially in resource-constrained environments. Future research should aim to integrate culture-dependent and culture-independent methods to achieve a more comprehensive characterization of the urobiota.
This study contributes significantly to the understanding of the healthy male urobiota, showing that the urinary tract is a complex ecosystem with substantial microbial diversity. These findings pave the way for further research into how these microorganisms contribute to health and disease, with the potential to revolutionize the management of urinary tract conditions – perhaps by recommending changes in sexual behavior as a preventive measure.
Our study demonstrated the need for further research regarding the importance of urobiota for clinical practice. It should be borne in mind that even in a patient with symptoms of urinary tract disease, the detection of significant bacteriuria in a microbiological test may be a false positive result due to poor transport of the urine sample.