Bacillus thuringiensis is a globally distributed gram-positive, soil-dwelling saprophytic organism known for its spore-forming ability, which enables its survival in unfavorable conditions (Palma et al. 2014). The discovery of B. thuringiensis is attributed to Shigetane Ishiwatari, who first discovered it in Japan in 1901 from the diseased larvae of the silkworm Bombyx mori (Ishiwata 1901). Subsequently, in 1911, Ernest Berliner isolated the same bacterium from dead Mediterranean flour moth larvae and named it B. thuringiensis 1911, after the nearby German province of Thuringia (Berliner 1911; DeLucca II et al. 1981; Monnerat et al. 2008). The species B. thuringiensis is characterized by the production of different insecticidal proteins, such as parasporal crystals, during the sporulation phase (Santos et al. 2022). These crystals predominantly comprise one or more crystal (Cry) and cytolytic (Cyt) proteins, which have been extensively explored as biopesticide agents for managing pests and targeting human cancer cells (Palma et al. 2014; Brasseur et al. 2015; Bel et al. 2020).
Cry proteins are parasporal inclusion Cry proteins encoded by the Cry gene, which exhibit toxicity against specific insect orders (Xu C et al. 2014; Efremenko et al. 2023). Moreover, Cry toxins target receptors on the midgut epithelia (Moazamian et al. 2018). Cry proteins are classified into 75 families with 800 different Cry genes based on the homology of their amino acid sequences (Sajid et al. 2018), whereas Cyt proteins are grouped into three families with 38 genes (Jouzani et al. 2017).
Cry proteins target parasitic agents, such as Trichomonas vaginalis (Kondo et al. 2002), Plasmodium berghei (Xu Z et al. 2004), Leishmania major (El-Sadawy et al. 2008), and nematodes (Zheng et al. 2018). Additionally, several Cry proteins have demonstrated efficacy against common insect orders, including Coleoptera (Donovan et al. 2006), Diptera (Shankar et al. 2019), and Lepidoptera (butterflies and moths) (Plata-Rueda et al. 2020). Numerous studies have shown that Cyt proteins are toxic to pathogenic bacteria (Tamegnon 2012), fungus (Jouzani et al. 2017), and cancer cells (Rezaei et al. 2023) (Figure 1).

Diagram illustrating the potential effects of Cry proteins from B. thuringiensis
In this review, we summarize the current knowledge on the mechanisms-of-action (MOA) of PS families produced by B. thuringiensis, highlight persistent research gaps, and discuss future perspectives for their development as potential targeted cancer therapeutics.
After decades of employing Cry and Cyt proteins as biopesticides, it was established that some parasporal Cry proteins parasporin (PS) are non-toxic to insects but exhibit high cytotoxicity against human cancer cells (Mizuki Eiichi et al. 2000). PS constitutes genetically diverse Cry proteins derived from non-insecticidal inclusion proteins of B. thuringiensis. They are categorized into six groups (PS1–PS6) with varying sizes and possess markedly different mechanisms of action against human cancer cells (Mizuki Eiichi et al. 2000). In this review, we summarize the mechanisms-of-action (MOA) of the PS families produced by B. thuringiensis as potential alternatives for cancer treatment.
Understanding the mechanism of classical Cry and Cyt toxins provides a valuable structural and mechanistic framework for elucidating parasporin function. While Cry and Cyt toxins are well-known for their insecticidal activity, primarily targeting receptors in the insect midgut (e.g., cadherin, aminopeptidase-N) and forming lytic pores that lead to osmotic lysis, parasporins represent a distinct subclass of non-insecticidal Cry proteins with unique mechanisms directed against mammalian cancer cells (Mizuki et al., 2000; Ohba et al., 2009). Unlike classical toxins, parasporins recognize cancer-specific surface markers such as GPI-anchored proteins or Beclin-1, and may induce apoptosis, necrosis, or pore formation in a cell type-dependent manner (Katayama et al., 2007; Kitada et al., 2009). A detailed comparison of PS-specific mechanisms is provided in Section 5. Cyt toxin is a key component that acts synergistically with different family members. A previous study demonstrated a synergistic effect between Cyt1Ab and Cyt2Ba against resistant Culex quinquefasciatus and Aedes aegypti larvae (Figure 2) (Wirth et al. 2001). It has been established that Cry11Aa is toxic upon binding to the transmembrane protein cadherin (CAD); however, in the absence of this receptor, Cry-4Ba can oligomerize following proteolytic activation (Rodríguez-Almazán et al. 2012). A CAD-based alternative model has been proposed. In this model, cytotoxicity is mediated through the interaction of CAD receptors, which activate an Mg2+-dependent signaling pathway, ultimately causing cell death (Zhang et al. 2005). Moreover, Cry toxin triggers the stimulation of G protein and adenylyl cyclase (AC), which increases intracellular cyclic adenosine monophosphate levels. This, in turn, activates protein kinase A, destabilizing the cytoskeleton and membrane-bound ion channels (Soberon et al. 2009). Defects in toxin-receptor interactions are the most common mechanisms of resistance to Cry toxins (Liu et al. 2021). At least four distinct protein receptors have been identified for Cry1A toxins (toxins specific to lepidopterans): a cadherin-like protein [CADR], a glycosylphosphatidyl-inositol (GPI)-anchored aminopeptidase-N (APN), a GPI-anchored alkaline phosphatase (ALP), and a 270 kDa glycoconjugat (Gómez et al. 2007).
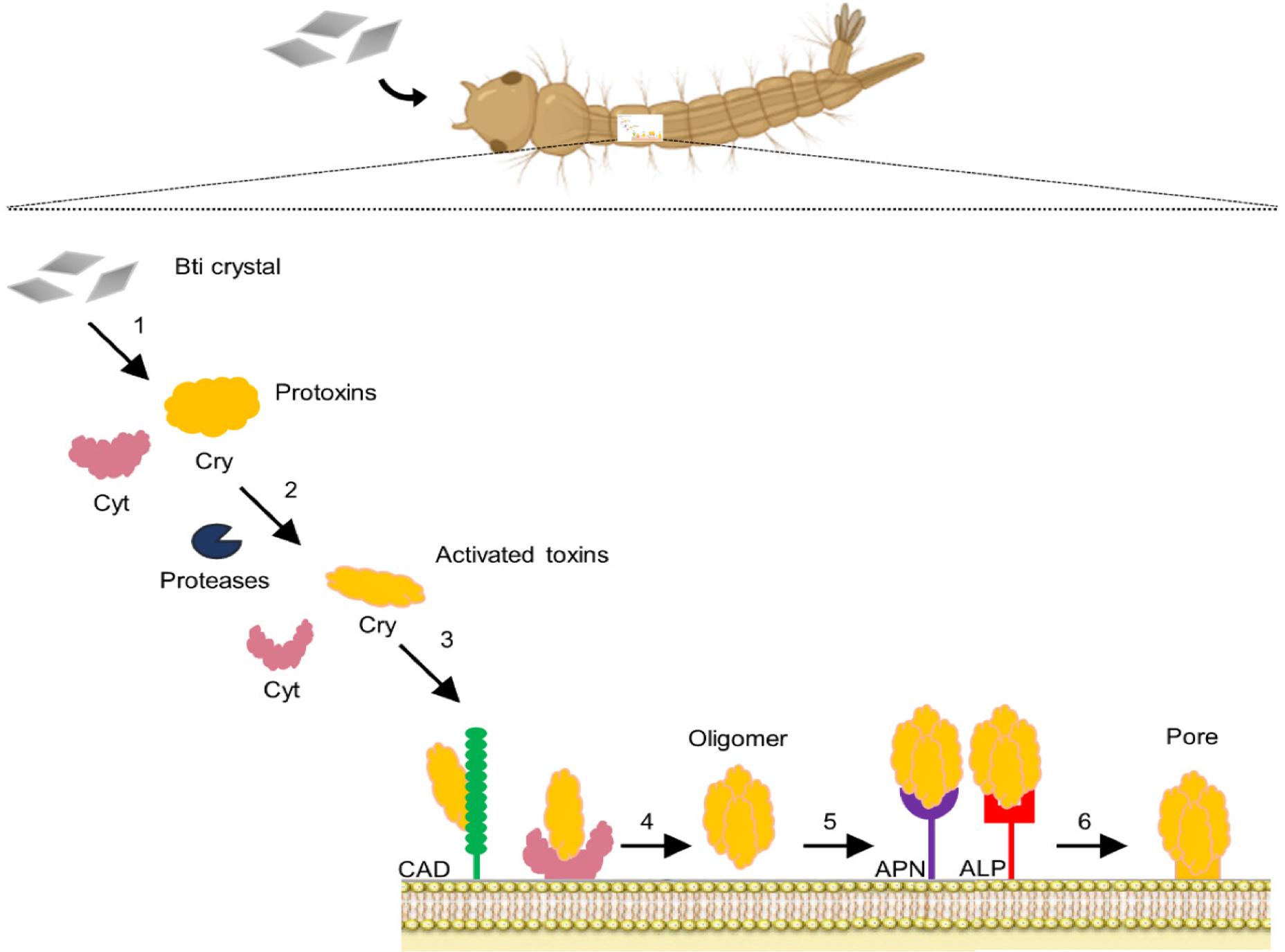
Diagram illustrating the synergistic effects between B. thuringiensis Cry and Cyt toxins on mosquito larvae.
Since the discovery of the first gene encoding a novel insecticidal protein from B. thuringiensis in 1981, the number of such genes has steadily increased (Schnepf and Whiteley 1981). Initially, the primary ranking of the relevant genes for B. thuringiensis pesticidal Cry and Cyt nomenclature was based on the insecticidal properties of crystal proteins (Höfte and Whiteley 1989). The CryI, CryII, CryIII, and CryIV genes encode proteins that are toxic to lepidopterans, both lepidopterans and dipterans, coleopterans, and only dipterans, respectively. Although classification based on this criterion does not inherently indicate similarity in protein structure, host range, or mode of action, the updated nomenclature, proposed by proposed by Crickmore and co-workers (Crickmore et al. 2021), employes a hierarchical clustering using amino acid sequences allows the grouping of closely related toxins. For instance, in the primary rank, Roman numerals have been replaced with Arabic numerals for proteins with less than 45% sequence identity (protein name and Arabic number, e.g., Vip1 and Vip2) (Palma et al. 2014). Since most of these novel proteins were grouped together as Cry proteins it became obvious that the present nomenclature had limits in characterizing the large variety of proteins that had been identified. The fundamental concepts of the 1998 edition are retained in this revised nomenclature, but specific mnemonics are added to illustrate different structural groups. Initially, 16 classes were specified, three of them (Cry, Cyt, and Vip3) remain unaltered from the former naming of B. thuringiensis toxins. All proteins that were previously classified as belonging to Cyt and Vip3 maintain their current names. Only proteins thought to have the traditional three-domain structure are currently included in the Cry class. This includes both proteins with and without an extended C-terminus, as well as variations with extra regions such beta-trefoil domains. The remaining classes are non-3-domain proteins that were previously identified by the Cry mnemonic and/or newly contributed sequences (from bacteria other than Bt). The specified pfam domains, recognized structures, and other information provide the foundation of these further classes. The three-letter mnemonics were selected to either reflect a historical name (Mcf, Mtx) or the type of pesticidal protein they represent (Mpp – Mtx2-like; Tpp – Toxin_10-like, etc.). Proteins for which there is not enough information to assign them to a particular class will fall under one class, Xpp, which has been designated as a holding class. The Xpp mnemonic need to be regarded as transient (Crickmore et al. 2021). In the revised B. thuringiensis toxin nomenclature system, parasporins are now classified under the Mpp (Mammalian Pore-forming Protein) designation (Crickmore et al., 2021). For consistency with the historical and prevalent literature cited herein, the term ‘parasporin’ (PS) will be retained throughout this review.
The discovery of the anticancer properties of parasporins was largely serendipitous. During routine screening of B. thuringiensis strains for novel insecticidal proteins, Mizuki et al. (1999, 2000) observed that certain parasporal inclusions from non-insecticidal, non-hemolytic strains exhibited selective cytocidal activity against human leukemic cells. This unexpected finding led to the hypothesis that these proteins might recognize surface markers preferentially expressed on cancer cells, prompting targeted research into their mechanism and specificity. Subsequent studies confirmed that PSs constitute a unique group of Cry toxins with preferential toxicity toward various human cancer cell lines, distinguishing them from classical insecticidal toxins (Mizuki et al., 2000; Ohba et al., 2009). PS proteins are a novel class of Cry toxins present in some strains of B. thuringiensis lack hemolytic properties but exhibit the ability to preferentially target cancerous cells (Mizuki Eiichi et al. 2000). This discovery has prompted exploration into potential alternative biological applications for these non-insecticidal Cry proteins. Subsequent studies have consistently demonstrated that PSs are distinct protein toxins that preferentially eliminate human cancer cells while being largely non-toxic to normal human cells (Mizuki E et al. 1999; Mizuki Eiichi et al. 2000).
PSs are classified based on the degree of pairwise amino acid identity, employing the Cry toxin-inspired quaternary rank nomenclature scheme. Initially, there were four first-rank categories (PS1–PS4) (Saitoh et al. 2006); however, subsequently, two more subgroups (PS5 and PS6) were added (Nagamatsu et al. 2010; Aboul-Soud et al. 2019).To date, 19 members of the six families of PSs and their cancer cell-killing mechanisms have been described and are presented in Table 1.
Overview of identified parasporins (PS/Mpp): B. thuringiensis strains, associated Cry genes, activation proteases, main cellular targets and observed cytotoxic outcomaes.
| PS | B. thuringiensis Strain | Cry Gene | Protease Activation | Main Cellular Target | Observed Cellular Outcome | Reference |
|---|---|---|---|---|---|---|
| PS1Aa1 | A1190 | Cry31Aa1 | Trypsin | HeLa | Apoptosis | (Mizuki Eiichi et al. 2000) |
| PS1Aa2 | M15 | Cry31Aa2 | Trypsin | HepG2 | Not determined | (Jung et al. 2007) |
| PS1Aa3 | B195 | Cry31Aa3 | Trypsin | HeLa | Not determined | (Uemori et al. 2008) |
| PS1Aa4 | Bt79-25 | Cry31Aa4 | Proteinase K | Not determined | Not determined | (Uemori et al. 2008) |
| PS1Aa5 | Bt92-10 | Cry31Aa5 | Proteinase K | Not determined | Not determined | (Uemori et al. 2008) |
| PS1Aa6 | M019, 64-1-94 | Cry31Aa6 | Trypsin | HepG2 | Not determined | (Gonzalez et al. 2011) |
| PS1Ab1 | B195 | Cry31Ab1 | Trypsin | HeLa | Not determined | (Uemori et al. 2008) |
| PS1Ab2 | Bt31-5 | Cry31Ab2 | Proteinase K | Not determined | Not determined | (Uemori et al. 2008) |
| PS1Ac1 | Bt87-29 | Cry31Ac1 | Proteinase K | Not determined | Not determined | (Uemori et al. 2008) |
| PS1Ac2 | B0462 | Cry31Ac2 | Proteinase K | HeLa | Apoptosis | (Kuroda et al. 2013) |
| PS2Aa1 | A1547 | Cry46Aa1 | Proteinase K | HepG2 | Pore-forming | (Ito et al. 2004) |
| PS2Aa2 | A1470 | Cry31Aa2 | Proteinase K | MOLT-4 | Not determined | (Okumura et al. 2013) |
| PS2Ab1 | TK-E6 | Cry31Ab1 | Proteinase K | Jurkat | Not determined | (Hayakawa et al. 2007) |
| PS3Aa1 | A1462 | Cry41Aa1 | Proteinase K | HL60 | Not determined | (Yamashita et al. 2000) |
| PS3Ab1 | A1462 | Cry41Ab1 | Proteinase K | HL60 | Not determined | (Yamashita et al. 2000) |
| PS4Aa1 | A1470 | Cry45Aa1 | Proteinase K | CaCo2 | Pore-forming | (Okumura et al. 2006) |
| PS5Aa1 | A1100 | Cry64Aa1 | Proteinase K | TCS | Not determined | (Okumura et al. 2006; Ekino et al. 2014) |
| PS6Aa1 | M019, 64-1-94 | Cry63Aa1 | Trypsin | HepG2 | Not determined | (Gonzalez et al. 2011) |
During the first discovery of PS1 from the isolate A1190, its antitumor effect was demonstrated in cervical cancer (HeLa) (Mizuki Eiichi et al. 2000) and liver cancer (HepG2) cell lines (Jung et al. 2007) through binding to the Beclin-1 receptor in the cell membrane (Katayama et al. 2011). Beclin-1 is a noteworthy protein found intracellularly in all healthy cells, where it plays a role in the processes of autophagy and apoptosis. However, in susceptible cells with limited Beclin-1 expression, it is found extracellularly and functions as a PS1 receptor (Liang et al. 1999). This presents a unique and paradoxical mechanism: PS1 targets a receptor whose normal intracellular function is pro-survival (autophagy). The binding of PS1 to extracellular Beclin-1 subverts this function, triggering a pro-death signal via Ca2+ influx (Katayama et al. 2007). This mechanism suggests that PS1 may selectively target cancer cells that have mislocalized Beclin-1, a phenomenon reported in certain malignancies, thereby exploiting a cancer-specific vulnerability rather than merely a differentially expressed receptor. PS1 promotes Ca2+ influx from the extracellular buffer, an action that can be counteracted by G-protein inhibitor suramin. This ultimately leads to PS1-induced cytotoxicity and triggers cell death via an apoptotic pathway (Katayama et al. 2007). Ca2+ is essential for controlling a number of cell functions, including autophagy. Different organelles within cells act as Ca2+ storage chambers and regulate Ca2+ levels under different conditions. Through Ca2+ channels or other associated signaling proteins, Ca2+ in various compartments can influence autophagy. According to research, Ca2+ controls autophagy through different signal transduction pathways in both normal and stressed situations, which may contribute to the emergence and progression of neurodegenerative disorders (Hou et al. 2024).
PS1Aa1, a typical three-domain protein, exhibits a preference for cytotoxicity against several human cancer cell lines, including HepG2, HeLa, acute T-cell lymphoblastic leukemia (MOLT-4), and promyelocytic leukemia (HL60) (Okassov et al. 2015). HeLa cells, but not MOLT-4 cells, were found to be susceptible to the cytotoxic effects of PS1Ac2 (Kuroda et al. 2013). Furthermore, PS1Aa1 proteins from nine non-larvicidal and non-hemolytic B. thuringiensis strains native to Saudi Arabia demonstrated significant dose-dependent cytotoxicity against HeLa malignant cells but not HT-29 cells. The average half-maximal inhibitory concentration (IC50) value was 6.8 µg/mL, ranging from 3.2 (B. thuringiensis strain 1) to 14.2.8 µg/mL (B. thuringiensis strain 6) (Aboul-Soud et al. 2019).
PS2, a member of B. thuringiensis serovar dakota (H15), was first identified in B. thuringiensis A1547 (Wong 2010). PS2 induces damage in hepatoma cells by reacting intensively with human receptor-like proteins (Ito et al. 2004). PS2 is concentrated in lipid rafts and can potentially bind specifically to cholesterol in the membranes of human cancer cells. A previous study demonstrated that the efficiency of oligomerization is affected by the absence of cholesterol, suggesting that PS2 induces pore formation in the plasma membranes of cells (Abe et al. 2008). This cholesterol-dependent mechanism aligns PS2 with a broader family of bacterial cholesterol-dependent cytolysins (CDCs), such as perfringolysin O. However, unlike many CDCs that bind cholesterol directly via a conserved undecapeptide motif, PS2’s primary interaction appears to be with glycosylphosphatidylinositol (GPI)-anchored proteins concentrated in lipid rafts, with cholesterol acting as a critical membrane component for stabilizing the oligomeric pore complex (Kitada et al., 2009). Accordingly, a multistep process model was proposed. Initially, PS2 would bind to the GPI-anchored receptors, primarily found in the proteins or cholesterol and sphingolipid-rich lipid rafts of the membrane. As the toxin becomes concentrated and undergoes oligomerization, transmembrane pores are formed, ultimately leading to cell death (Kitada et al. 2009).
The PS2 family comprise three members: PS2Aa1, PS2Aa2, and PS2Ab2 (Akiba and Okumura 2017). Crystal structures of representative PS1Aa1 and PS2Aa1 members have been determined and compared to those of well-researched toxins. In contrast to PS2Aa1, PS2Aa1 likely initiates its action by recognizing and binding to GPI-anchored proteins in the membranes of susceptible cancer cells. Subsequently, PS2 undergoes oligomerization, culminating in cell death (Ohba et al. 2009) (Figure 3).
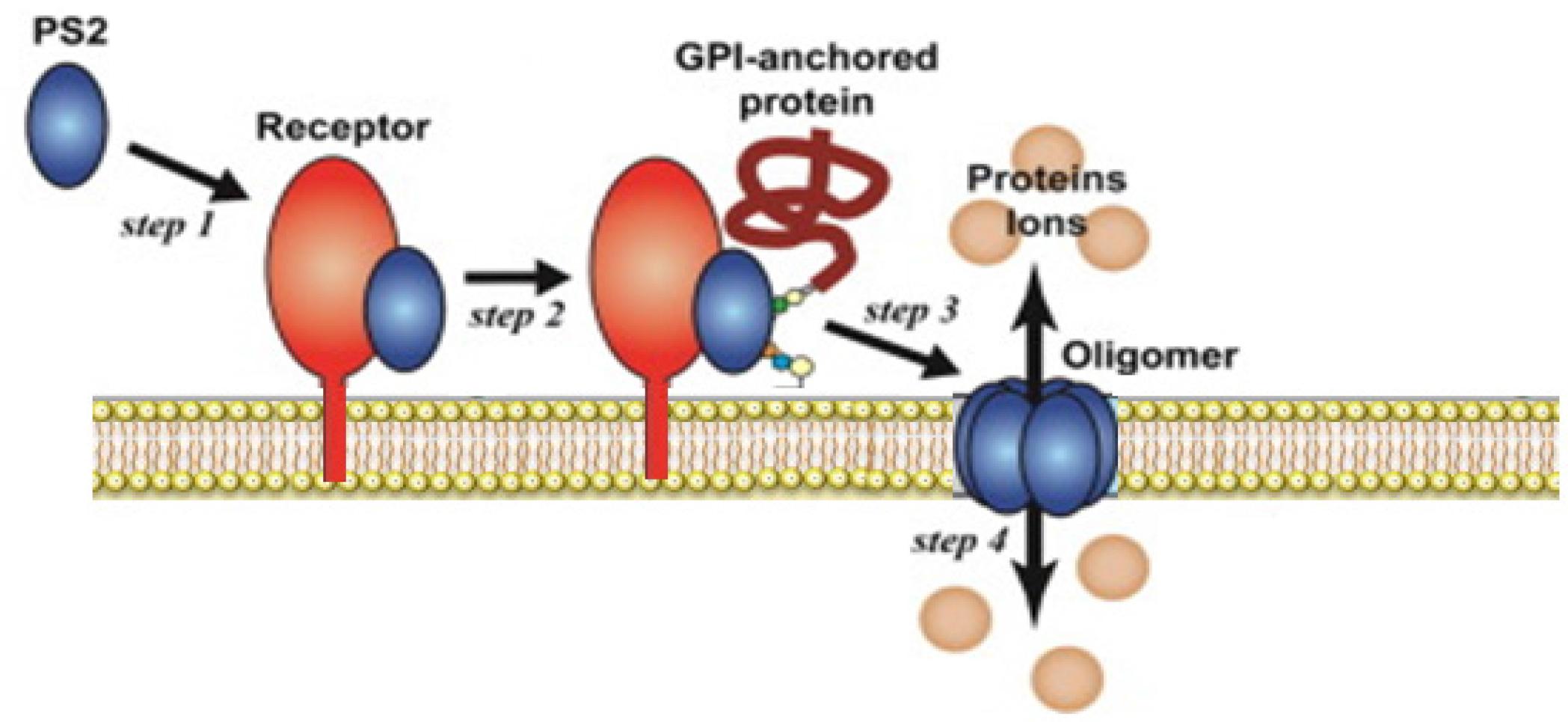
Generalized mechanism-of-action of parasporin 2 (PS2). PS2 binds to GPI membrane receptors in susceptible cells, which induces PS2 oligomerization, ultimately causing cell death.
PS3Aa1, also known as Cry41Aa1, is the only known member of the PS3 family (Hayakawa et al. 2007). PS3Aa1 has been shown to inhibit cell proliferation in several cancer cell lines, including HL60 and HepG2, through a pore-forming toxin (PFT) mechanism, resulting in ATP imbalance, cell enlargement, and increased cellular permeability (Krishnan et al. 2017). Although PS3 shares structural similarities with Cry proteins, it lacks the five conserved blocks characteristic of Cry proteins (Suárez-Barrera et al. 2021). PS3 causes a rapid and significant decrease in metabolic activity, accompanied by increased lactate dehydrogenase (LDH) production, distinguishing it from PS2, which primarily affects HL60 and HepG2 cancer cell lines (Ohba et al. 2009; Krishnan et al. 2017). The differential sensitivity of cell lines to PS3 may be attributed to variable expression of its specific, yet still unidentified, membrane receptor(s). Furthermore, its distinct PFT mechanism—resulting in rapid ATP depletion and necrosis-like death—might be particularly effective against cancer cells with high metabolic rates or compromised membrane repair mechanisms, such as certain leukemic (HL60) and hepatoma (HepG2) cells. This contrasts with the more apoptosis-prone pathway induced by PS1.
The first isolation of PS4 occurred in the soil isolate B. thuringiensis A1470 from serovar shandongiensis. This isolate yielded protein devoid of non-insecticidal and non-hemolytic properties, which subsequently formed irregularly shaped PS inclusions upon crystallization (Lee et al. 2001). The active protein is approximately 27 kDa in size and exhibits strong cytotoxicity against human uterine cervical cancer (TCS), MOLT-4, HL60, U937, HepG2, Sawano, leukemia (DE-4), and human colon cancer (CACO-2) cell lines (Inouye et al. 2008; Okumura et al. 2008). PS4 forms an oligomeric pore complex via nonspecific attachment to the plasma membrane of target cells. It exhibits cytotoxic activity throughout this process, in contrast to PS2, which is cholesterol-dependent for this activity. The first steps in the MOA of PS4 in promoting necrosis, which culminates in cell death, are the release of LDH and the entry of dextrans with different molecular weights into the cell (Inouye et al. 2008; Okumura et al. 2008; Okumura et al. 2011; Krishnan et al. 2017). However, according to PSI-BLAST search findings, PS4 shares sequence homology with Cry15Aa at 24%, a-toxin at 15%, aerolysin at 10%, and ε-toxin at 21%. In addition, a CD spectrum of PS4 showed 51% β-structure. Consequently, PS4 may represent a distinct β-PFT with a cholesterol-independent activity. Nonetheless, the determination of a particular receptor and its transmembrane mechanism remains unclear (Xu C et al. 2014). This cholesterol independence is a significant mechanistic divergence from PS2 and many other PFTs. It implies that PS4 could exert cytotoxic activity against a wider range of cancer cell types regardless of their membrane cholesterol composition or lipid raft integrity. Its structural homology to aerolysin-like β-PFTs suggests it may form small, protein-lined β-barrel pores, which could lead to a more selective ion permeability and a different death phenotype compared to the larger pores formed by some cholesterol-dependent toxins.
The PS5 family comprises only PS5Aa1, also known as Cry64Aa1, as its sole member. This protein was isolated from soil B. thuringiensis strain A1100, and it exhibits in vitro cytocidal activity against MOLT-4. The active form of PS5Aa1, which has an estimated molecular weight of 30 kDa and is specifically toxic to MOLT-4, shares 56.4% sequence similarity with the three-domain Cry toxins. Further sequence analysis of PS5Aa1 revealed that it is an epsilon protein that exhibits significant interaction with the drug methotrexate (Nagamatsu et al. 2010). PS5 exhibits cytotoxicity against different cancer cells, such as MOLT-4 and HepG2, and also against healthy cells, including normal lung (MRC-5) and uterus (UtSMCs) cells (Ekino et al. 2014). However, its MOA remains unclear (Ekino et al. 2014). Given its significant sequence similarity (56.4%) to three-domain Cry toxins and its interaction with methotrexate, one plausible hypothesis is that PS5Aa1 may act as a molecular mimic, potentially interfering with folate metabolism or other critical pathways in susceptible cells. Its activity against both cancer and normal cells suggests it may target a ubiquitously expressed receptor or a fundamental cellular process, highlighting the need for further research to identify its precise target and engineer tumor-selective variants.
PS6 is a polytopic membrane protein and is the most recently discovered member of the PS family. It is closely linked to PS2 and has a conserved sequence of 50 amino acids constituting three domains. Although several action modalities remain unknown, previous studies have reported that the cancer cell-killing activity of PS6 against HepG2 and HeLa cells is associated with its pore-forming activity; however, its modes of action remain undetermined (Nagamatsu et al. 2010; Mendoza-Almanza et al. 2020). As a polytopic membrane protein with conserved three-domain architecture and close linkage to PS2, PS6 likely shares mechanistic features with other parasporins. Its reported pore-forming activity suggests a PFT mechanism. Its specific cytotoxicity profile (HepG2 and HeLa) points towards the expression of a specific receptor complex. Structural modeling and domain-swapping experiments with PS2 could be fruitful strategies to elucidate whether its selectivity is governed by its receptor-binding domain (Domain II), akin to the specificity seen in Cry toxin variants.
All members of the PS family share a conserved structure (Figure 4). The structural comparison of PSs can be categorized based on their molecular masses, with higher and lower ranges. Higher molecular mass molecules like PS1, PS3, and PS6 initially present as nascent forms, weighing approximately 80 kDa. They then undergo processing to become active forms with a molecular mass of 60–70 kDa. Conversely, PS2, PS4, and PS5 start as inactive precursors with smaller molecular masses. They then undergo processing to transform into active forms with a molecular mass of 27–30 kDa (Suárez-Barrera et al. 2021).
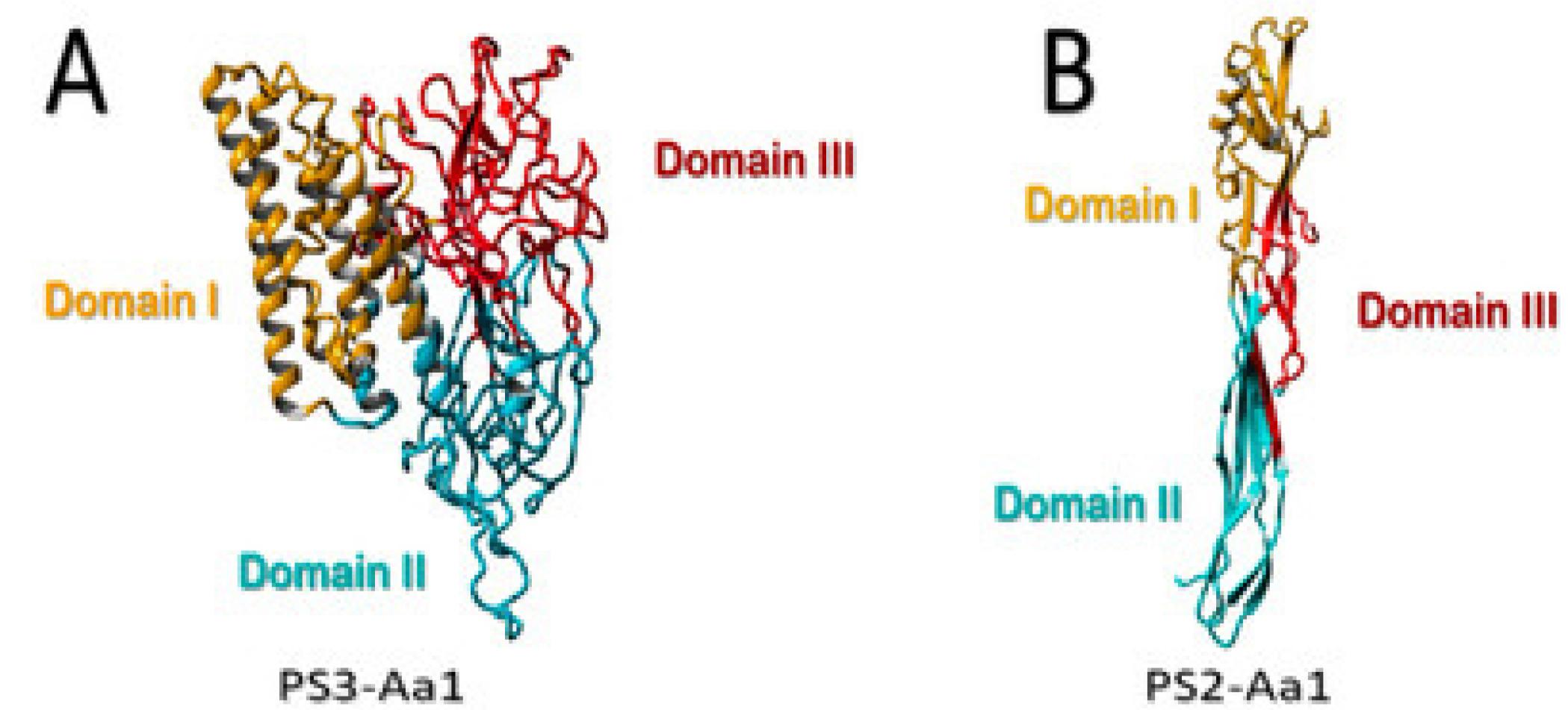
(A) Three-domain structural model of higher-molecular-weight PS3Aa1. (B) Structural model of low-molecular-weight PS2Aa1. Mechanism of cancer cell-killing action of PSs (Suárez-Barrera et al. 2021).
The crystal structures of PS1Aa1 and PS2Aa1 have been identified and compared with those of well-studied toxins. PS1Aa1, which corresponds to Cry31Aa1, contains 723 amino acids and has a calculated molecular weight of 81 kDa. It also contains three domains of insecticidal Cry toxins, the structures of which differ from those of the Cry proteins, although it is further broken down into two polypeptides.
The PS2Aa1 is composed of three domains: I, II and III. While domain I is responsible for membrane penetration and pore formation, domain II mediates specific binding to GPI-anchored proteins on cancer cell membranes. Stabilization of the oligomeric pore complex is conferred by domain III (Abe et al. 2008). The PS2Aa1 molecules create a sandwich structure that includes a β-hairpin (S6 and S7 for the nontoxic protein; S8 and S9 for PS2Aa) along with an anti-parallel five-stranded β-sheet (S3, S9, S12, S5, and S8 for the nontoxic protein; S5, S6, S7, S10, S11, and S13 in PS2Aa). Furthermore, three out of the five strands of the β-sheet reorganize with the S1 strand to create a four-stranded β-sheet adjacent to the boundary of domain I, linked to the helices of domain I via hydrophobic interactions. Site-directed mutagenesis was conducted on the amphipathic β-strand of an epsilon toxin. Domain III also participates in the formation of pores and contains a loop that traverses the membrane. It is crucial for the interaction among the individual monomers within the oligomer. In PS2, the five-stranded β-sheet is transformed into a three-stranded β-sheet along with two anti-parallel strands (S6, S11, S13/S14, S7, and S10) (Suárez-Barrera et al. 2021). The crystal structure of PS3Aa1 (Cry41Aa1) reveals a three-domain architecture typical of many Cry proteins, comprising a receptor-binding domain (Domain II), a pore-forming domain (Domain I), and a carbohydrate-binding domain (Domain III). This structure facilitates membrane insertion and oligomerization (Kitada et al. 2009). Unlike PS2Aa1, PS4Aa1 has three domains comprising 338 amino acids and possesses a computed molecular weight of 37 kDa. Its structure is distinct from that of Cry proteins (Katayama et al. 2007; Suárez-Barrera et al. 2021).
The MOA of PSs against target cancer cells is poorly understood; however, existing evidence has demonstrated that parasporins exhibit numerous mechanisms of action to kill cancer cells. Although it is generally known that the specificity of Cry toxins depends on the detection of cell membrane receptors (cadherin, aminopeptidase-N, and alkaline phosphatase), these proteins function similarly to Cry toxins due to their strong cell type selectivity. However, research on the interaction of PS proteins with cell membrane receptors is currently ongoing. Several compounds that function as PS receptors have been documented and patented. In this context, Beclin-1 has been reported to act as PSAa1 receptor on the surface of susceptible cancer cells (Mendoza-Almanza et al. 2020). In the natural environment, several PS proteins can induce cell death in mammalian cells through pore-forming toxins (PFTs) and detergent models and/or apoptosis activation (Mendoza-Almanza et al. 2020). PFTs represent the most prevalent cytotoxic agent produced and secreted by various bacteria in human infections. Specifically for PS proteins, evidence supports that PS2Aa1 and PS4Aa1 function as PFTs by binding to GPI-anchored proteins or cholesterol-rich lipid rafts, oligomerizing, and forming transmembrane pores that lead to ion imbalance and cell death (Kitada et al. 2009; Okumura et al., 2011). PS1Aa1 induces apoptosis through Ca2+ influx and caspase activation (Katayama et al. 2007). They can be induced in two distinct ways: the first involves a direct cell disruption that can ultimately lead to cell death (Figure 5), whereas the second entails indirect damage caused by PFT-induced inflammatory responses such as interleukin (IL)-1, IL-6, and tumor necrosis factor-α (Los et al. 2013). PFTs can perforate both the plasma membrane and intracellular organelle membranes, diffuse toward the target cell, and bind to specific receptors, ultimately leading to cell death (Ulhuq and Mariano 2022). PFTs recognize various host cell proteinaceous receptors, including GPI-anchored proteins and cholesterol, to detect target cells (Los et al. 2013). PFTs are a unique and significant area of study in the development of novel and widely applicable antimicrobial prophylactics and treatments owing to their near-ubiquitous presence in bacterial infections (Li et al. 2021). PFTs can attach to the host cell surface and initiate their translocation across the plasma membrane. They then undergo a series of conformational changes to reach their cytosolic targets, consequently exposing or generating new hydrophobic surfaces capable of penetrating the core of cell membranes (Wanderley et al. 2020).
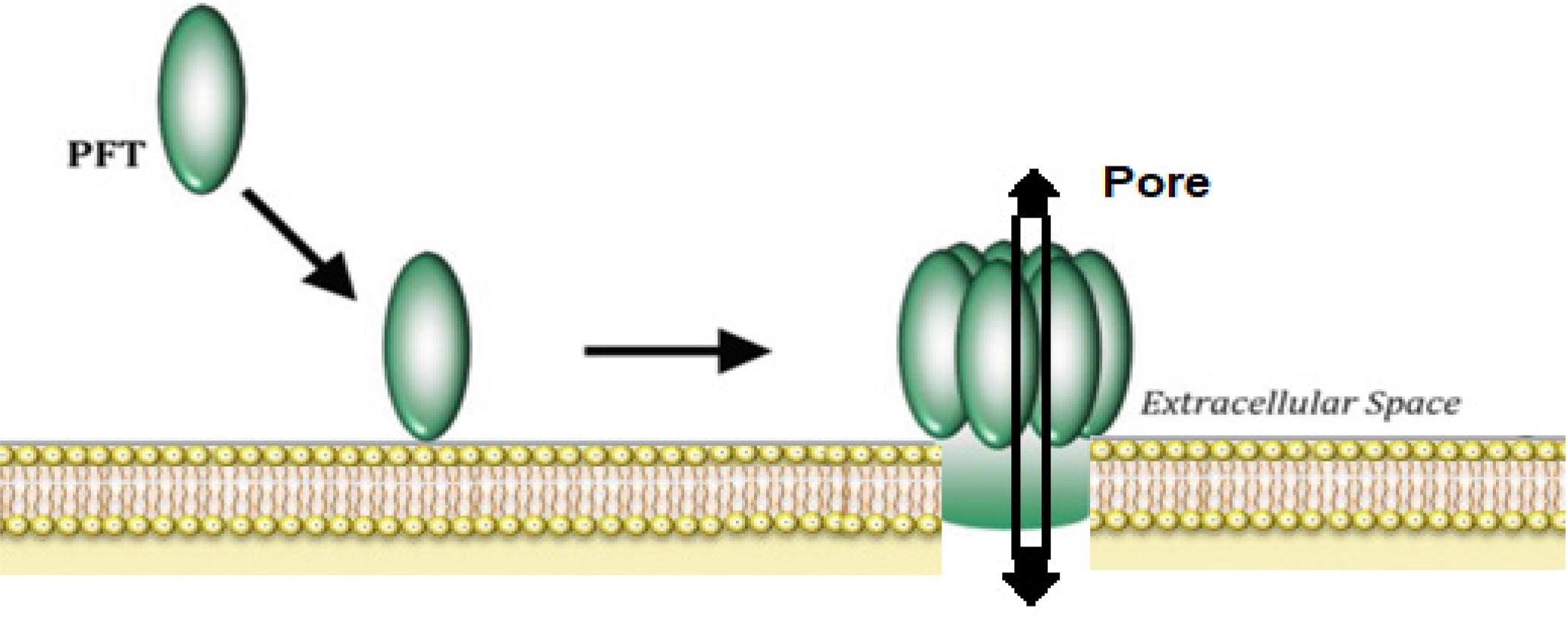
Diagram illustrating the generalized mechanism of pore-forming toxins (PFTs). Owing to oligomerization and membrane receptor binding, an aqueous hole is formed in the plasma membrane as PFPs attach to their target receptor through contact with an exposed receptor on the cell surface.
The second mechanism of cell death induced by PS involves triggering the breakdown of the plasma membrane and the formation of aggregates on the membrane surface. Both PFTs and the detergent model can affect the levels of toxin; the detergent effect is predominant at high PS toxins concentrations, whereas oligomerization and hole formation are more likely to occur at low PS concentrations (Lee et al. 2001). Consequently, at high PS concentrations, the cell membranes of target cells are unable to form oligomers (Singh et al. 2022).
In a previous study, activated PS2Aa1, which was obtained from a novel B. thuringiensis strain, 4R2, demonstrated selectively cytotoxic activity against various cells, including colon, HepG2, PC-3, and MCF-7 cells. Morphological alterations were observed upon treatment of PS2Aa1 with proteinase K. Western blot analysis further indicated that only the cancer cell lines cleaved caspase-3 (CASP3) and CASP9, which is indicative of apoptosis or programmed cell death. In the PC-3 and HepG2 cell lines, PS2Aa1 dramatically boosted both CASP3 and CASP7 activity following a 24-hour treatment. In addition, it exhibited minimal toxicity against normal cells (Brasseur et al. 2015).
Various methods have been used to alter the PS binding specificity and affinity. The result is a genetically modified toxin that combats resistance that has emerged in the field and targets new pest species. Alterations to the binding affinity and specificity of PS can be achieved through truncation, site-directed mutation, domain exchanges, and the synthesis of PS proteins and their subsequent detection in mutant toxin-carrying phage libraries, as well as from any of these processes (Suárez-Barrera et al. 2021).
The cytocidal activity of PS proteins produced by B. thuringiensis renders them potential medical anti-cancer candidates. Increasing the potency of B. thuringiensis strains through genetic modifications to produce innovative biopesticides should be considered in future endeavors. Over the past four decades, genetic engineering has been employed to alter several Cyt proteins to create chimeric toxins characterized by high toxic potential and a reduced degree of complexity in their quaternary structures (Torres-Quintero et al. 2018). GBP 3.1 attaches to the stomach epithelia of the midgut and hindgut of pea aphids and, to a lesser extent, to the aphids of green peaches and soybeans. The Cyt chimeric toxin was the first effective outcome of the insertion of GBP 3.1 (amino acid sequence: TCSKKYPRSPCM), a pea aphid gut-binding peptide, into the amino acid sequence of the Cyt2Aa toxin. This modification prevented plant viruses transmitted by pea aphids, which act as vectors for the virus, from infecting the cell (Chougule et al. 2013). To enhance binding to Manduca sexta receptors and increase toxicity against two lepidopteran pests, M. sexta and Plutella xylostella, the loop 3 amino acid sequence from the Cry1Ab toxin (FRSGFSNSSVSI) was inserted into the cyt1Aa gene via a PCR technique (Torres-Quintero et al. 2018).
In receptor and channel protein-binding investigations, modified proteins with different amino acid substitutions are utilized to enhance the features of the proteins, such as thermal activity, selectivity, specificity, or folding (He et al. 2012; Kim and Izumi 2014). Protein engineering, which involves the synthesis of proteins with improved functionality and the design and production of artificial polypeptides, plays a crucial role in maximizing the generation of mutant libraries to replicate the experimental conditions of directed mutagenesis procedures (Pinzon et al. 2017; Stimple et al. 2020). Moreover, a previous study demonstrated that three of the five Cry11 variants generated via DNA shuffling exhibited harmful effects against Aedes aegypti and Culex quinquefasciatus (Florez et al. 2018).
Limited in vivo research indicates inconsistent findings for the anticancer effectiveness of parasporin proteins in animal models, and no clear data supports their direct usage as systemic cancer treatment. Although these proteins show encouraging selective cytotoxicity against several cancer cell lines in vitro, their potential for whole-animal therapeutic application has not been extensively or reliably investigated. (Okassov et al. 2015). This is an important issue since many compounds that exhibit action in cancer cells in culture can be inert in animal models. To ensure human safety, the compounds’ toxicity profiles must be thoroughly examined before being used in clinical oncology. Several studies have revealed the anticancer efficacy of these isolated crystal proteins from B. thurigiensis in animal models. In 1973, an Indian group of researchers evaluated the toxicity and effectiveness of the pure crystal from the bacteria against Yoshida ascites sarcoma (YAS) in vivo. Wistar A/IISc inbred rats weighing around 100–120 g and Swiss mice (20–25 g), got 20 million of the cultivated YAS cells. The rats were given a single intraperitoneal dosage of the bacterial protein twenty-four hours following the tumor implantation. The highest tolerable dose of 120 mg/kg for the pure crystal is 100 times higher than the minimum effective dose of 1 mg/kg stated by the authors. Furthermore, a single dosage of crystal, delivered in the first month after tumor implant might protect the animals for the observation period of six months, despite receiving a dose of cancer cells every month (Prasad S et al. 1973). In 1975, the same group determined how the toxin affect the rise in the humoral immune response in rats. The authors stated that the hemagglutination and hemolysin titers of the animals treated with the PS crystals exhibited a substantial rise in proportion to the untreated immunized controls, due to the increase in the IgM and IgG antibodies (Prasad SSSV and Shethana 1975). However, these crystal proteins’ sequencing results are not available, making it impossible to confirm their function. The results of an in vivo toxicity investigation have only been published for one of the sequenced and registered PS4. A Japanese research group described an LD50 of PS4 as 160 μg/kg in male ICR mice (via subcutaneous route), which despite being lesser in terms of lethality compared to most of the bacterial toxins, it can be nevertheless called a harmful chemical. The scientists also reported an impairment in the kidney function in ICR mice after having been intraperitoneally injected with PS4, associated with destruction of the proximal renal tubule of the organ. However, when administered orally, no toxic effects were observed (Okumura et al. 2014). Thus, further in vivo investigations of PSs are certainly required. Limitation of testing PSs in vivo, such as those linked to the degradation of PS proteins in the gut or their breakdown during cellular metabolism can be overcome by nanoencapsulation and/or use of nasal and parenteral route that avoid degradation in the gastrointestinal tract (GIT). The nanoencapsulation strategy provides protection for therapeutic proteins, such as Bt-derived PS proteins, from enzymatic degradation in the gut, thereby improving tumor targeting. They include innovative techniques that generate nanocarriers such as microemulsion, nanoemulsion, liposomes, and nanogels. These techniques collectively improve the penetrability of biological obstacles such as blood brain barrier, GIT as well as skin barriers (Han and Santos 2024). However, more research should be directed to the nano- and micro-encapsulation strategies and evaluating its utility in overcoming limitations of peptide- and protein-based cancer therapeutics such as instability, short circulation time, and poor trans-membrane transportation, and immunogenicity (Han and Santos 2024). The use of delivery systems is also a potential alternative to boost their therapeutic value, while lowering toxicity, by controlled release, and selectively targeting cancer-affected tissues. In this context, employing selective receptor targeting strategies have been reported to improve tumor targeting potential, selectivity index and reduce off-target toxicity of anthrax toxins-based therapeutics (Misra et al. 2025).
Large-scale recombinant expression of functional parasporins remains technically demanding. Many PSs form inclusion bodies in heterologous systems such as E. coli, requiring optimized refolding protocols that may affect yield and bioactivity (Suárez-Barrera et al., 2021; Santos et al., 2022).
Like many protein therapeutics, parasporins may be susceptible to proteolytic degradation in vivo and could elicit immune responses upon systemic administration, potentially limiting their therapeutic window and necessitating immunogenicity assessments (Los et al., 2013; Mendoza-Almanza et al., 2020).
While PSs show selectivity toward cancer cells in vitro, achieving tumor-specific targeting in vivo without off-toxicity remains a hurdle. Advanced delivery systems—such as nanoparticle encapsulation, antibody-drug conjugates, or peptide-directed carriers—could enhance tumor accumulation and reduce systemic exposure (Ulhuq & Mariano, 2022; Santos et al., 2022).
Protein engineering approaches, including domain swapping, site-directed mutagenesis, and directed evolution, offer avenues to enhance parasporin specificity, stability, and potency (Suárez-Barrera et al., 2021). Combining parasporins with conventional chemotherapeutics or immunomodulators may also yield synergistic effects. Moreover, rigorous in vivo studies in immunocompetent animal models are urgently needed to evaluate pharmacokinetics, toxicity, and antitumor efficacy, paving the way for potential clinical application (Mendoza-Almanza et al., 2020; Santos et al., 2022). Future research must prioritize well-designed in vivo studies in immunocompetent and xenograft models that not only assess tumor regression but also evaluate systemic toxicity, immune response, and optimal dosing regimens. Addressing these challenges is not merely the next step but a prerequisite for translating parasporins from promising laboratory toxins to viable clinical candidates.
B. thuringiensis, known for its insecticidal properties, produces PSs that hold promise as potential anti-cancer candidates. However, their direct clinical application may cause unwanted immunological reactions in patients. One approach that could help address this drawback involves identifying PS-specific cell receptors in cancer cells. Currently, there are 19 identified PSs, categorized into six families (PS1–PS6), produced by at least 11 B. thuringiensis strains. Each PS family exhibits a specific cancer cell-killing mechanism. The PS proteins are not associated with insect toxicity or hemolytic action. Although in vivo studies remain limited, preliminary investigations in mouse xenograft models have shown that PS2Aa1 and PS4Aa1 can inhibit tumor growth without significant systemic toxicity (Brasseur et al., 2015; Okumura et al. 2011). These findings underscore the importance of further in vivo evaluations to assess pharmacokinetics, immunogenicity, and long-term safety. However, their in vivo antitumor activity in animal models, such as mice and rats, is yet to be fully elucidated. Reports on acute and chronic toxicity of PSs in animal models are also limited. This is a significant limitation, as many molecules demonstrating efficacy in cultured cancer cells may be inactive in animal models. Therefore, additional in vivo studies, particularly in rats, on PSs are imperative to ascertain their potential application in clinical cancer treatment. Furthermore, the shift from in vitro potency to in vivo applicability is obstructed by challenges including scalable production, protein stability, potential immunogenicity, and the necessity for tumor-targeted delivery systems. To make parasporins more useful in cancer treatment, we need to get past these problems by using protein engineering, advanced formulation, and thorough preclinical testing (Suárez-Barrera et al., 2021; Santos et al., 2022).