Orthodontics is a branch of dentistry that treats malocclusion, a condition in which teeth are not correctly aligned when the mouth is closed, leading to improper bites. Orthodontics plays a significant role in the treatment of children with cleft lip or palate (Robinson 2017; Redlich and Tenne 2019). Orthodontic treatment may have adverse effects during treatment. The adverse effects include pain, root resorption, and periodontal disease. If the length of treatment increased, root resorption risk also increased. Ketchman first discussed the problem of root resorption and discussed nutritional deficiencies, along with various orthodontic treatments, which contribute to root resorption. It is a risk that may cause further root shortening (Ketcham 1927; Talic 2011). The second risk factor is pain, which is associated with orthodontic treatment, where 70–95% of the patients experience this problem. Sometimes the pain is unbearable, so it may be a reason to discontinue the treatment due to pain. A third risk factor is pulpal changes during orthodontic treatment; there is a possibility of loss of pulp vitality. The fourth risk factor is enamel decalcification, commonly known as white spots. It can be considered as the first step towards the cavity. It occurs in 50% of patients. Zinc oxide nanoparticles show antibacterial activity. It can damage the bacterial cell wall and interrupt DNA replication. The nanoparticles release ions inside cells and produce reactive oxygen species (ROS). Various studies have reported that zinc oxide nanoparticles have strong potential to generate ROS. This is why it damages the cell wall and increases permeability. These nanoparticles have shown antimicrobial activity against various foodborne pathogens, including Escherichia coli, Salmonella species, and Staphylococcus aureus. It also prevents the growth of Gram-positive and Gram-negative bacteria. Zinc oxide nanoparticles are identified as “Generally Recognised As Safe” (GRAS) by the US FDA, which shows the minimum toxicity to humans (Mokammel et al. 2019). Zinc oxide nanoparticles are a very advanced new dental material that exhibits antibacterial activity when incorporated into composite resins (Adams et al. 2006; Tavassoli Hojati et al. 2013). Dental adhesives with composite resins exhibit mechanical properties when zinc oxide nanoparticles are incorporated into the fillers (De Souza Rastelli et al. 2016). Pal et al. (2018) studied the synthesis of zinc nanoparticles and nanoparticles modified with cotton fabric. Li et al. (2019) studied the formulation and characterization of zinc oxide nanoparticles and antimicrobial bioceramics. Nanoparticles increase the acid resistance of cement, thereby increasing the longevity of the prosthesis (Nguyen et al. 2019).
Recent research highlights several antibacterial agents used in orthodontics to reduce biofilm formation and enamel decalcification, including silver nanoparticles, chitosan composites, and bioactive glasses. While these materials show antibacterial activity, limitations such as cytotoxicity, discoloration, or inconsistent mechanical properties have been reported. In comparison, zinc oxide nanoparticles are considered safe, exhibit broad-spectrum antibacterial action through ROS generation, and are compatible with dental composites and cements. However, their incorporation into Orthodontic Portland Cement (Grade 43) and the combined evaluation of antimicrobial and mechanical properties remain inadequately explored, which is the focus of this study.
The primary purpose of the study was to evaluate the antibacterial activity of the zinc oxide nanoparticles. It was determined by the crystal violet staining method, and the samples were compared with the control samples. This method is very beneficial for treating tooth caries or for orthodontic tooth treatment. Zinc oxide nanoparticles have been explored in dental materials, but their integration into Orthodontic Portland Cement (Grade 43) and the simultaneous evaluation of antibacterial and mechanical properties are less studied. This work addresses that gap by showing that ZnO nanoparticle incorporation effectively inhibits Streptococcus mutans and Lactobacillus casei biofilm formation while maintaining essential properties such as flexural and tensile strength and setting time. These findings suggest a promising strategy to enhance the durability and antibacterial performance of orthodontic cements.
Sodium hydroxide (NaOH), zinc chloride (ZnCl2), 2-propanol (99%), 1,2-ethanediol (99%) were purchased from Merck KGaA, Germany. Doubled-distilled water was used throughout the synthesis. Silver nitrate (AgNO3) waspurchased from Merck KGaA, Germany. The synthesis was carried out at a high degree to achieve a nucleation rate greater than the growth rate.
Bacterial strains such as S. mutans and L. casei were used. Brain Heart Infusion Broth (BHI) was purchased from Merck KGaA, Germany. Orthodontic Portland Cement (OPC) was procured from Jiangxi, China.
Bacterial strains of S. mutans and L. casei were used. These bacterial strains were selected as commonly implicated in dental caries and biofilm formation in orthodontic patients. Orthodontic Portland Cement (Grade 43) was used due to its clinical relevance. Sample size for each test (control and ZnO-incorporated cement) was fixed in triplicates based on preliminary trials to ensure statistical reliability and reproducibility.
About 7 g of ZnCl2 was taken into a beaker and dissolved in 200 ml of water, keeping the beaker in an oil bath at about 900°C. A 5 M NaOH solution was prepared and was added dropwise to the above solution. Slow stirring was done for about 10 min at 900°C to disperse the particles. It was settled for about half an hour. Particles were sedimented to the bottom; the supernatant was separated and discarded. The sediment portion was washed 4–5 times with distilled water to reduce the NaCl concentration. The quantity of NaCl remaining in the suspension was analyzed with a solution of silver nitrate (AgNO3). The purified particles were separated out with an ultrasonic bath of 2-propanol solution for about 15 min at room temperature. This process, called peptization, is essential for separating particle agglomerates and releasing the nano units of zinc oxide. Using centrifugation, particles were collected at 6,000 rpm for 10 min. Particles were washed repeatedly about three times. Thermal treatment was given at 250°C for 4 hours to form zinc oxide nanoparticles. 1,2-Ethenediol can also be used for synthesis by using the same procedure, only at a temperature of 150°C.
For the preparation of orthodontic cement, Orthodontic Portland Cement (OPC) of grade of 43 was used. Orthodontic Portland Cement (OPC) Grade 43 is classified as a hydraulic cement under IS 8112 standards. It achieves 43 MPa compressive strength at 28 days, suitable for general structural and orthodontic applications. Orthodontic Portland Cement is also available in Grades 33 and 53.
Grade 33 offers lower strength for plastering/masonry, while Grade 53 provides high early strength for heavy structural works. It was adapted from Jiangxi, China. It was grey cement, free of lumps. Chemical and physical properties were provided in Table I. The specific gravity of the cement was 1.81 g/cm3. With the help of a concrete drum mixer, a series S mixture consisting of coarse aggregates (30% by weight), i.e., a combination of crushed basalt and fine sand, was used. Cement and ZnO nanoparticles were mixed. The cement was powder material. Mixing was done for about 2 minutes, then water was added, and mixing continued for about 3 minutes. The formulated fresh concrete was evaluated for different parameters. It was immediately evaluated for flexural strength (Huang et al. 2001; Arefi and Rezaei-Zarchi 2012).
Physicochemical properties of the OPC.
| Chemical properties | |||||
|---|---|---|---|---|---|
| Material | SiO2 | Al2O3 | Fe2O3 | CaO | MgO |
| Cement | 20.92 | 5.12 | 3.9 | 51.14 | 6.11 |
| Material | SO3 | Na2O | K2O | Loss on ignition | |
| Cement | 2.93 | 0.22 | 0.92 | 3.21 | |
Color, odor, appearance of the formulation was checked by visual observation. pH was measured.
It is the structural design criteria that serve as the general indicators of concrete strength. The flexural strength test was conducted using a mixing procedure in which cylinders with a 150 mm diameter and 300 mm height were cast and compacted in two layers mounted on a vibrating table, with each layer vibrating for about 15 sec (ASTM C348). The moulds, which are filled with formulation, are covered with polyethylene sheets, and it was moistened for about a night. It was then demolded and cured in water at 250°C before use in the experimental period, and strength was measured at 7 and 14 days. It was carried out on three samples, and the average of flexural strength was obtained (Huang et al. 2001; Jatania and Shivalinga 2014).
It was carried out in accordance with ASTM C496 standards. After the material’s curing period, the concrete cylinders were allowed to split tensile strength, which was determined using a specially designed universal testing machine. The average of the three samples’ strengths would be the result of split tensile strength (Huang et al. 2001)
The setting time of the sample with ZnO particles incorporated into the cement was determined according to ASTM standards. With the help of the Vicat needle, the setting time of hydraulic cement was determined (Huang et al. 2001)
The lower incisor human tooth was removed with no history of previous endodontic treatment or dental caries, resorption, or cracks. This extracted tooth was placed in double-distilled water. It was appropriately cleaned, all debris removed, and its teeth were sterilized in an autoclave. Below the junction of cement and enamel, the tooth was resected horizontally. The resected crowns reacted with phosphoric acid, and the adhesive and brackets were applied. A test group was prepared and treated with the formulation i.e., cement incorporated with zinc oxide nanoparticles (Sharon et al. 2018)
The test organisms used for this analysis are S. mutans and L. casei. These test organisms were correctly cultured overnight at 37°C in Brain Heart Infusion broth supplemented with 5% sucrose. Optical density of the suspensions was adjusted to 1 at 570 nm (Sharon et al. 2018).
About 2 ml of phosphate-buffered saline (PBS) was taken in an Eppendorf tube, and each crown was placed in it. This setup is incubated for 48 hours at 37°C. The PBS was always replaced with a fresh PBS solution every 48 hours for one week. These samples were then allowed to grow bacteria. Coated crown groups were subjected to bacterial outgrowth, and bacterial strains, i.e., S. mutans and L. casei (n = 10), were added to the evaluation sample. A control group was also prepared, with n = 10 strains (i.e., S. mutans and L. casei) added to the crown without the application of the formulation. Separate suspension of S. mutans with 2 ml and another 2.5 ml suspension of strain L. casei was prepared and taken into the test tubes in which a crown was inserted, and then incubated for 48 hrs. at 37°C. The crystal violet staining method was used to determine the antibacterial effect of zinc oxide. After 48 hours of incubation, the crowns were removed and transferred into the new test tube containing 5 ml of PBS. This PBS solution was replaced three times every 30 seconds. The last collected PBS was inoculated onto a blood agar plate for the evaluation of bacterial growth (Sharon et al. 2018).
For this staining, well plates are used, in which each crown was placed in a well, and 1 ml of 100% methanol solution was added over the crown without touching the crown or the well edges. About 1 ml of crystal violet solution was added to each well for about 15 minutes, and the crown was switched to the next well. With double-distilled water, the crown was washed off to dissolve the stained dye. 100% ethanol was added to the crown, and the mixture was vortexed for 30 seconds with a pipettor. From all the wells, collect all the ethanol, and the optical density was determined by UV visible spectrophotometry at a wavelength of 540 nm (Sharon et al. 2018).
The formulation appeared like the tooth material, and the color was white. It was odorless and neutral in pH.
It was determined for the S1–S5 samples, both with and without ZnO nanoparticles. Table II shows the results for flexural strength. Flexural strength increases with an increase in the concentration of the ZnO particles. In S1, the strength was 2.5 MPa and decreased to 2.31 MPa. As the concentration of ZnO increases, the tensile strength value increases. S4 showed a 3.1 MPa strength, which decreased to 2.45 MPa in 28 days.
Flexural tensile strength with and without ZnO particles.
| Sample No. | conc.ZnO particles | Flexural tensile strength | |||
|---|---|---|---|---|---|
| 7 days | 14 days | 21 days | 28 days | ||
| Control | 0 | 2.3 | 2.4 | 2.13 | 2.1 |
| S1 | 0.05 | 2.5 | 2.4 | 2.34 | 2.31 |
| S2 | 0.15 | 2.7 | 2.54 | 2.22 | 2.1 |
| S3 | 0.25 | 2.88 | 2.44 | 2.1 | 2.1 |
| S4 | 0.5 | 3.1 | 3.2 | 2.99 | 2.45 |
| S5 | 1 | 3 | 3.1 | 2.23 | 2.11 |
Split tensile strength was determined for samples S1–S5 with varying concentrations of Zinc oxide nanoparticles. It was also determined that the control sample means were without Zinc oxide nanoparticles. The determination was done on days 7, 14, 21, and 28, which is represented in Table III.
Split tensile strength of cement containing different concentrations of ZnO nanoparticles.
| Sample No. | conc.ZnO T | Split tensile strength MPa | |||
|---|---|---|---|---|---|
| 7 days | 14 days | 21 days | 28 days | ||
| Control | 0 | 3.9 | 4.2 | 4.2 | 4.3 |
| S1 | 0.05 | 5.1 | 5.3 | 5.2 | 5.11 |
| S2 | 0.15 | 5.41 | 5.25 | 5.19 | 5.04 |
| S3 | 0.25 | 6.47 | 6.15 | 5.96 | 5.29 |
| S4 | 0.5 | 6.87 | 6.28 | 6.17 | 6.04 |
| S5 | 1 | 6.3 | 6.11 | 5.38 | 5.11 |
After analysis, it was observed that the tensile strength of the material increased when Zinc oxide nanoparticles were added. The addition of ZnO nanoparticles was initiated at 0.05% in the S1 sample after 1 week, and the maximum split tensile strength was 5.2 MPa. It was also observed that with an increase in ZnO nanoparticle concentration, the tensile strength decreases; at a concentration of 1% it was 6.3 MPa.
Fig. 1 and 2 represent the initial and final setting times, respectively, showing the effect of increasing Zinc oxide nanoparticle concentration on the setting time. The initial and final setting times of the samples S1 to S5 decreased as the concentration increased.
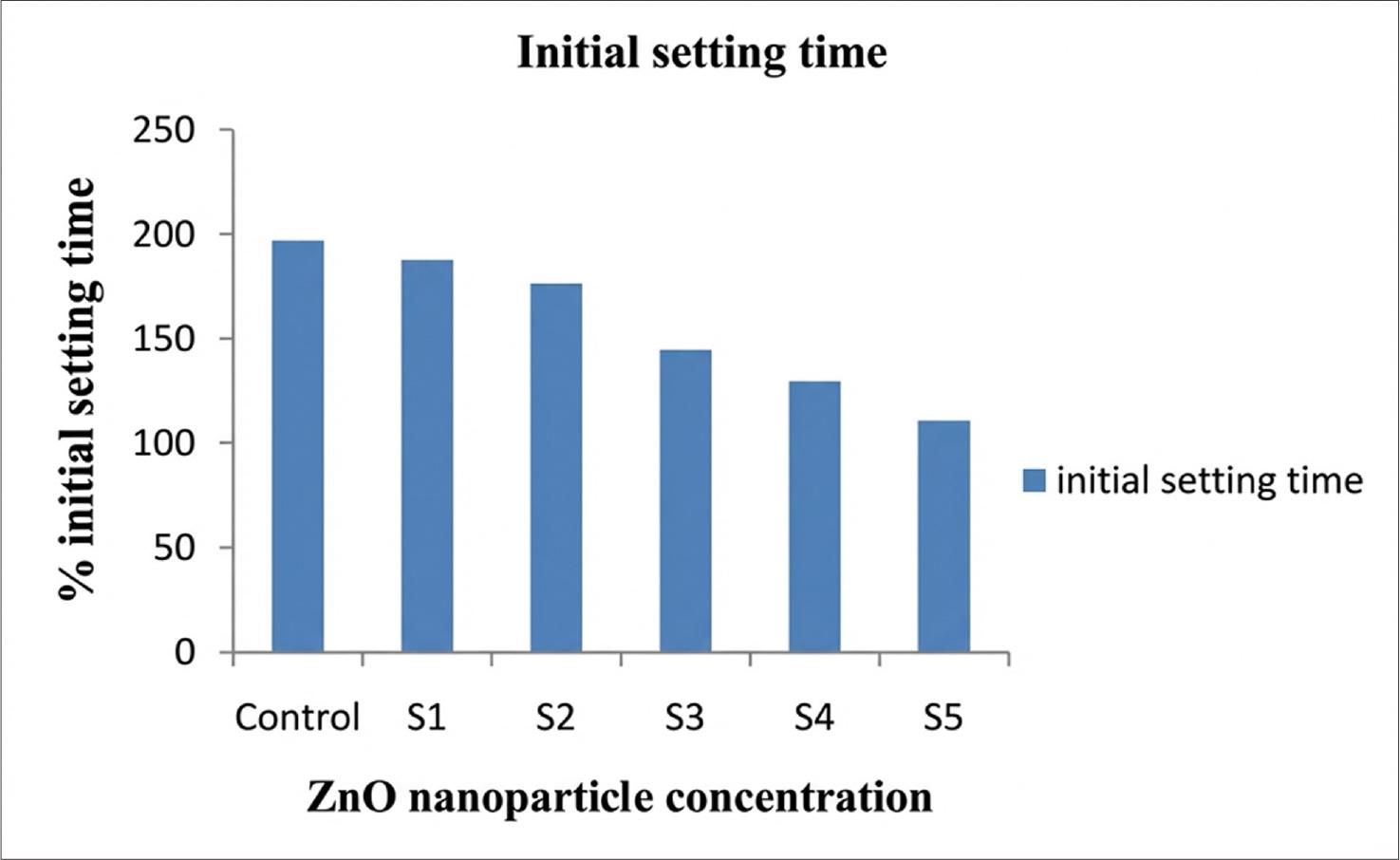
Initial setting time for the sample with and without ZnO particles.
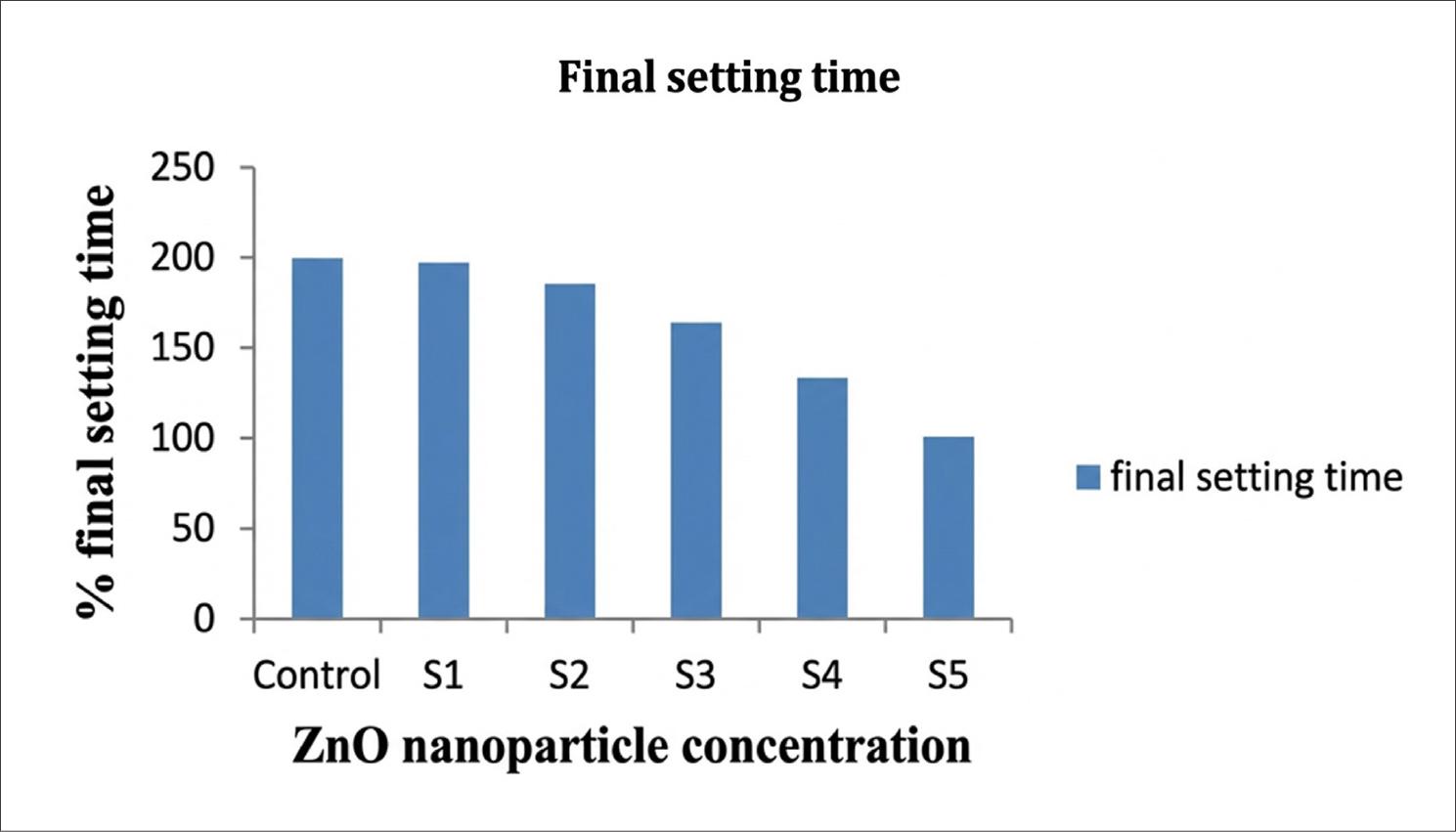
Final setting time for the sample with and without ZnO particles.
The crystal violet staining method was used to determine the antibacterial effect. Biomass was measured for S. mutans and L. casei strains. The biomass of S. mutans in the presence of zinc oxide was lower, i.e., 39.2%, compared to the control group. Also, the antibacterial effect of zinc oxide was lower than the biomass growth of S. mutans and L. casei, which was 64.22% (Fig. 3). There was inhibition of biofilm formation in the presence of zinc oxide nanoparticles incorporated into the adhesive system (i.e., the cement material), thereby enhancing the antimicrobial effect. These differences in the biomass growth formation were significant (Table IV).
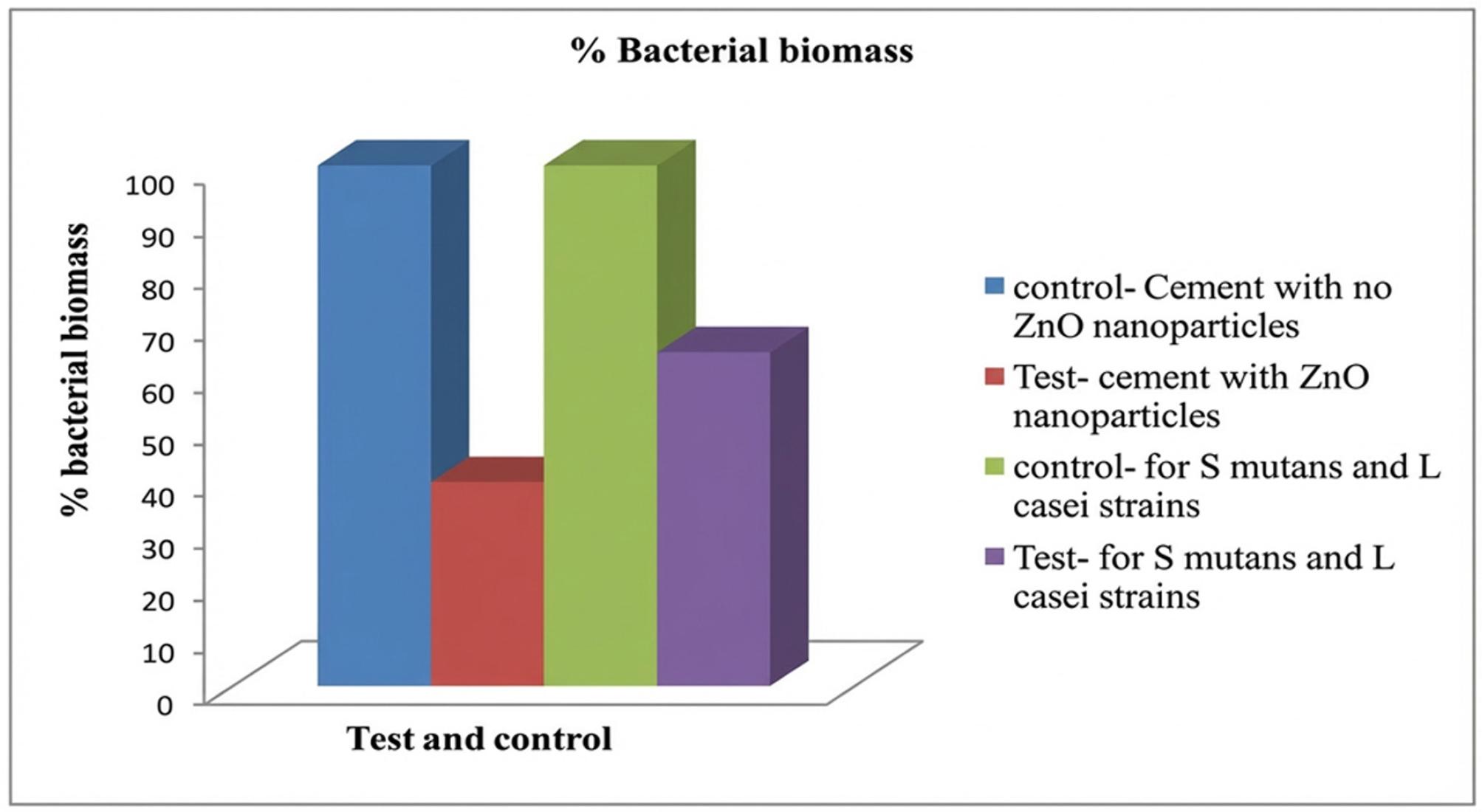
Percentage of bacterial biomass for the test, i.e., cement with ZnO nanoparticles and control samples with no application of ZnO nanoparticles, with the use of Streptococcus mutans and Lactobacillus casei strains.
Percentage of biomass for the test groups and controls against Streptococcus mutans and also the combination of S. mutans and Lactobacillus casei.
| Strain | Streptococcus mutants | Streptococcus mutants + Lactobacillus casei | ||
|---|---|---|---|---|
| Cements incorporated with ZnO nanoparticles | Control – No. ZnO nanoparticles | Cements incorporated with ZnO nanoparticles | Control – No. ZnO nanoparticles | |
| 1 | 57.34 | 100% | 40.5 | 100% |
| 2 | 43.22 | 100% | 41.54 | 100% |
| 3 | 34.21 | 100% | 37.45 | 100% |
| 4 | 42.11 | 100% | 32.76 | 100% |
| 5 | 43.55 | 100% | 35.376 | 100% |
| 6 | 43.11 | 100% | 30.89 | 100% |
| 7 | 34.24 | 100% | 36.84 | 100% |
| 8 | 32.77 | 100% | 32.54 | 100% |
| 9 | 30.4 | 100% | 29.32 | 100% |
| 10 | 32.33 | 100% | 25.38 | 100% |
| Average | 39.2 | 100% | 64.22 | 100% |
| SD | 6.89 | 0 | 5.05 | 0 |
Incorporating zinc oxide nanoparticles into Orthodontic Portland Cement significantly reduced biofilm formation by S. mutans and L. casei without affecting the cement’s pH, color, or mechanical properties. The antibacterial effect is attributed to the release of zinc ions and the generation of reactive oxygen species. Compared to other nanoparticles, ZnO offers broad-spectrum activity, lower cytotoxicity, and recognized safety, making it a promising option for longterm orthodontic applications.
Flexural strength of the ZnO particles increased with increasing concentration. It was observed that hydration occurred, and rapid consumption of the Ca(OH)2 occurred. It happened during the hydration of cement. Split tensile strength was determined on the control samples, without Zinc oxide nanoparticles. The tensile strength of the material increased when Zinc oxide nanoparticles were added. It was also observed that with an increase in ZnO nanoparticle concentration, the tensile strength decreases; at a concentration of 1% it was 6.3 MPa. After 14, 21, and 28 days, there were no significant changes in the tensile strength. At a concentration of 1% tensile strength was found to be decreased, that is, 6.3% this is due to the quantity of ZnO having been increased in the mixture, which is higher than the amount required for combination with liberated lime at the time of the hydration process, and hence, excess silica leaches out, causing the strength deficiency. Nanoparticles cause the weak zones (Li et al. 2007; Givi et al. 2010; Arefi and Rezaei-Zarchi 2012). The initial and final setting times of the samples S1–S5 decreased with increasing concentration. It was due to the ZnO particles having a higher hydration capacity than the cement itself; hence, these particles are characterized by higher surface energy and unique surface effects. A smaller particle size provides a larger surface area, leading to a faster rise in the number of surface atoms. These atoms are very reactive and unstable, and their reactions are more rapid. Hence, precautions should be taken for setting the time of cement paste at the time of application of ZnO nanoparticles (Kuo et al. 2006; Li et al. 2007; Wang et al. 2019).
Phillips et al. (1968) studied zinc oxide and eugenol cements for permanent cementation, and they showed results comparing zinc phosphate and zinc oxide combined with eugenol. However, in the presented study, zinc oxide showed better results. The application of nanoparticles is gaining interest in the field of dentistry. Some studies have reported the use of antimicrobial nanoparticles as topical agents in dental materials. In addition to zinc oxide, molecules used as antibacterial agents include titania and copper. TiO2 nanotubes can also be synthesized via anodization (David et al. 2018). Depan and Misra (2014) studied the role of the titania network in the silicon structure in preventing bacterial colonization. This network structure has antimicrobial activity. Girase et al. (2011) and Misra et al. (2012) studied the antimicrobial activity of the Silver-clay nanohybrid structure, which is effective and exhibits diffusion-controlled antimicrobial activity. Much research has been done on antimicrobial drugs, but antimicrobial use in dentistry applications is minimal. Hence, the presented study aims to formulate a cement containing zinc oxide nanoparticles, which is innovative and used in the treatment of orthodontic teeth.
Zinc oxide nanoparticles added to Orthodontic Portland Cement reduced bacterial biofilm formation while maintaining acceptable pH, color, and strength. Their antibacterial action is linked to zinc ion release and ROS generation. The dual benefits of antimicrobial protection and mechanical stability make ZnO-modified cement suitable for prolonged orthodontic treatments.
There was inhibition of biofilm formation in the presence of zinc oxide nanoparticles, which are incorporated into the adhesive system, i.e., in cement material, which enhances the antimicrobial effect. These differences in the biomass growth formation were significant. Along with the antibacterial activity, tensile strength and flexural strength were also determined, where it was found that ZnO nanoparticle incorporated cements had higher tensile strengths as compared to the control samples. So, it can be concluded that the addition of zinc oxide nanoparticles to the cement system will enhance antibacterial activity and increase cement density, acting as a filler and thereby decreasing cement porosity. Therefore, this method is very beneficial for the treatment of tooth caries or for orthodontic tooth treatment. This study demonstrates that incorporating zinc oxide nanoparticles into Orthodontic Portland Cement effectively reduces biofilm formation by S. mutans and L. casei while maintaining acceptable physicochemical and mechanical properties. However, the findings are limited to in vitro conditions and do not account for factors such as salivary flow, dietary variations, and long-term ion release in the oral cavity. Future research should focus on in vivo validation, long-term antimicrobial efficacy, and comparative studies with other nanoparticles or antibacterial agents to optimize clinical application in orthodontics.