The treatment of infectious diseases relies on antibiotics to kill or inhibit the growth of pathogenic bacteria (Abushaheen et al. 2020). The inappropriate use of antibiotics affects the patient’s clinical outcomes, such as the prognosis for healing, the potential for side effects, and the length of treatment, and becomes the dominant factor causing resistance; thus, the handling and treatment of infectious diseases are increasingly complicated, hospitalization is more extended, and has an impact on the treatment costs (Painuli et al. 2023). The widespread use of antibiotics, whether appropriate or not, exerts selective pressure by reducing the reproductive success of certain microorganisms, thereby accelerating the development of antimicrobial resistance (WHO 2018).
Antibiotic consumption is increasing globally, especially in low- and middle-income countries (Yin et al. 2018). The irrational use of antibiotics includes prescribing antibiotics for viral infections; prescribing the wrong type of antibiotic; prescribing the wrong dose, duration, or route of administration; increased antibiotic use in agriculture; and the frequent use of broad-spectrum and last-resort antibiotics. There is a clear association between antibiotic consumption and the emergence of antibiotic-resistant microorganisms (Wojkowska-Mach et al. 2018). Antibiotic consumption is an important parameter in the study of antibiotic use, and several metrics can be used to measure it. The most used method is the one recommended and updated by the WHO Collaborating Centre for Drug Statistics Methodology, World Health Organization (WHO 2015). Expressing antibiotic consumption in DDD per 100 patient-days or DDD per 100 bed-days (DDD/100-BD) allows hospitals to compare their consumption with that of other hospitals regardless of differences in the quality and quantity of antibiotics (Amaha et al. 2020).
Surgical antibiotic prophylaxis (SAP) is an important intervention for antimicrobial resistance. Guideline compliance has been poor across countries, leading to the inappropriate and overuse of antibiotics (Van Tuong et al. 2021). Although the principles of prophylactic antibiotic use have been established and a few guidelines have been published, adherence to these guidelines remains limited. A few common reasons include inadequate knowledge among health workers, improper use of preventive antibiotics, and a lack of policies (Musmar et al. 2014).
A study evaluating the pattern of antibiotic usage and its appropriateness in hospitals in Indonesia showed that the most prescribed antibiotic for sepsis in the intermediate care ward of the internal medicine department of Dr. Soetomo General Hospital by Adiwinoto et al. (2018) was monotherapy ceftriaxone IV, followed by the combination of cephalosporin IV combined with metronidazole and the combination of cephalosprin IV with quinolone IV. The most common antibiotics prescribed in orthopedic hospitals were monotherapy ceftriaxone IV, with 12.2% of antibiotics administered inappropriately. The proportion of inappropriate antibiotic prescriptions was 14.3% of the total antibiotic prescriptions (Adiwinoto et al. 2018). In a review to explore the antibiotic use pattern in the internal medicine department based on the DDD/ATC system, a primary criterion was to ensure that all subjects in publications were adults aged above 18 years; penicillin was the most consumed (127.9 DDD/100 bed-days), followed by cephalosporin (41.42D DD/100 bed-days), and fluoroquinolone (25.87 DDD/100 bed-days) (Andarsari et al. 2023).
Qualitative assessments of antibiotic use play an essential role in antibiotic stewardship programs (ASPs), as they identify patterns of inappropriate antibiotic prescription, establish intervention strategies, and evaluate the effectiveness of these interventions (Barlam et al. 2016). Several components associated with antibiotic prescriptions should be considered during their qualitative assessment. One suggested method is to use a set of quality indicators with sufficient evidence and measurable elements (Park et al. 2022; 2023).
The Regulation on Antimicrobial Resistance Control Program in Indonesia was authorized in 2015 and primarily focuses on antimicrobial resistance and surveillance of antimicrobial use, and it recommends extracting antimicrobial quantity data from medical or pharmacy records and quality data from antimicrobial usage forms. Data were analyzed using the Gyssens flowchart to assess the use of antibiotics received by patients (MoH Indonesia, 2015). Based on this background, this study aimed to analyze trends in the quantity and quality of antibiotics used in the surgical and medical wards of general academic hospitals.
This study was conducted at Dr. Soetomo General Academic Hospital in Surabaya, Indonesia, the main referral hospital in East Java Province.
This retrospective observational study collected data from pharmacy and medical records of patients treated in two low-care surgical wards and medical wards in internal medicine between January and May 2019. Demographic data for the patients were collected. The quantity of antibiotics used was analyzed using the DDD per 100 patient-days or DDD per 100 bed-days (DDD/100-BD) method, and the quality of antibiotic use was analyzed using the Gyssens category method, which was reviewed by the ASPs team. This study was approved by the ethics committee of Dr. Soetomo General Academic Hospital (ref. no: 1078/LOE/301.4.2/X/2022).
The inclusion criteria were that patients were male or female adults aged 18 years and above at the time of admission, had received systemic antibiotics (parenteral and oral), and had been hospitalized for more than 3 × 24 hours. Moreover, the exclusion criteria were incomplete medical records and patients discharged from the hospital or who died less than 3 × 24 hours after discharge.
The summary of demographic data of the patients was shown in Table I. At the same time, the detailed characteristics of the patients in the surgery and the medical room were shown in Table II and III.
Summary of demographic data in surgical and medical wards.
| Treatment group | Number of patients (n) | Number of patients who used antibiotics n (%) | Total sample (according to inclusion criteria) randomly (n) |
|---|---|---|---|
| Surgical wards | 1,167 | 675 (57.84%) | 225 |
| Medical wards | 1,426 | 1,115 (78.19%) | 222 |
Characteristics of patients in the surgical wards.
| Number of samples | n = 225 | % |
|---|---|---|
| Gender | ||
| Male | 126 | 56 |
| Female | 99 | 44 |
| Age | ||
| 18–40 years old | 88 | 39.11 |
| 41–60 years old | 95 | 42.22 |
| > 60 years old | 42 | 18.67 |
| Case distribution | ||
| Digestive Surgery | 50 | 22.22 |
| Orthopaedics and Traumatology | 47 | 20.89 |
| Surgical Oncology | 34 | 15.11 |
| Urology | 25 | 11.11 |
| Neurosurgery | 20 | 8.89 |
| TKV Surgery | 18 | 8 |
| Head and Neck Surgery | 11 | 4.89 |
| Plastic Surgery | 11 | 4.89 |
| Oral Surgery | 9 | 4 |
Characteristics of patients in the medical wards.
| Number of samples | n = 222 | % |
|---|---|---|
| Gender | ||
| Male | 110 | 49.55 |
| Female | 112 | 50.45 |
| Age | ||
| 18–40 years old | 40 | 18.02 |
| 41–60 years old | 98 | 44.14 |
| > 60 years old | 84 | 37.84 |
| Case distribution | ||
| Nephrology | 67 | 30.18 |
| Gastro-hepatology | 45 | 20.27 |
| Hemato-oncology | 44 | 19.82 |
| Endocrinology | 29 | 13.06 |
| Respirology | 11 | 4.95 |
| Tropical Infection | 22 | 9.91 |
| Rheumatology | 4 | 1.81 |
During the study, 1167 patients were in the surgical wards, of whom 675 (57.84%) had received antibiotics. The most frequently used antibiotics were ceftriaxone (41.67 DDD/100-BD), levofloxacin (22.82 DDD/100-BD), and cefazoline (17.75 DDD/100-BD). In contrast, there were 1426 patients in the medical wards of internal medicine, of whom 1115 (78.19%) had received antibiotics. The most consumed antibiotic in the medical wards was ceftriaxon (106.22 DDD/100-BD), followed by levofloxacin (34.95 DDD/100-BD) and metronidazole (26.25 DDD/100-BD). The trends in antibiotic consumption in the surgical and medical wards are shown in Fig. 1.
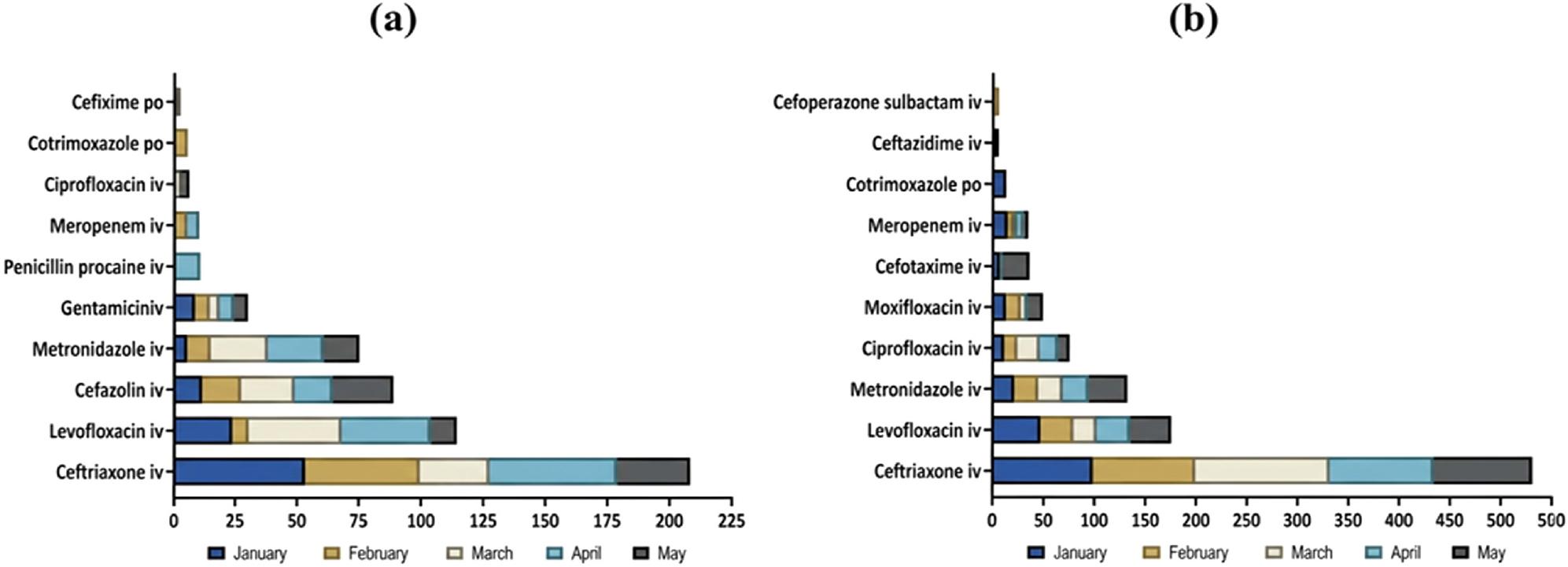
Trend of top ten antibiotics consumed in January–May 2019 (DDD/100-BD) in (a) surgical wards and (b) medical wards.
We collected data according to the inclusion criteria using random sampling from January to May 2019. The quality analysis of 225 antibiotic use cases in surgical wards included orthopedics, neurosurgery, digestive, oncology, cardiovascular, thoracic, and head-and-neck surgeries. The average length of stay (AvLOS) was 13 days. Indications for antibiotic use were prophylaxis (52.44%), empiric therapy (40.89%), and definitive therapy according to microbiological culture results (6.67%). The quality of antibiotic use in the surgical wards was assessed using the Gyssens category and is shown in Table IV.
Gyssens category of antibiotic use in the surgical wards.
| Gyssens category | Explanation | n | % |
|---|---|---|---|
| V | No indication | 72 | 17,2 |
| IV | Inappropriate selection of antibiotic type | 57 | 13,6 |
| III A | Duration of administration is too long | 32 | 7,6 |
| III B | Duration of administration is too short | 15 | 3,6 |
| II | Inappropriate dosing regimen | 20 | 4,8 |
| 0 | Appropriate antibiotic use | 223 | 53,2 |
| Total | 419 | 100 | |
The 222 cases in the medical wards included: sepsis with comorbidities, such as diabetes mellitus; acute renal failure; chronic kidney disease and cirrhosis; hepatic disease; pneumonia; and UTI. The average length of the hospital stay (AvLOS) was 10 days. Indications for antibiotic use as empiric therapy accounted for 86.94% of cases, and definitive therapy, based on microbiological culture results, accounted for 13.06%. The quality of antibiotic use in the medical wards was assessed using Gyssens’ category and is shown in Table V.
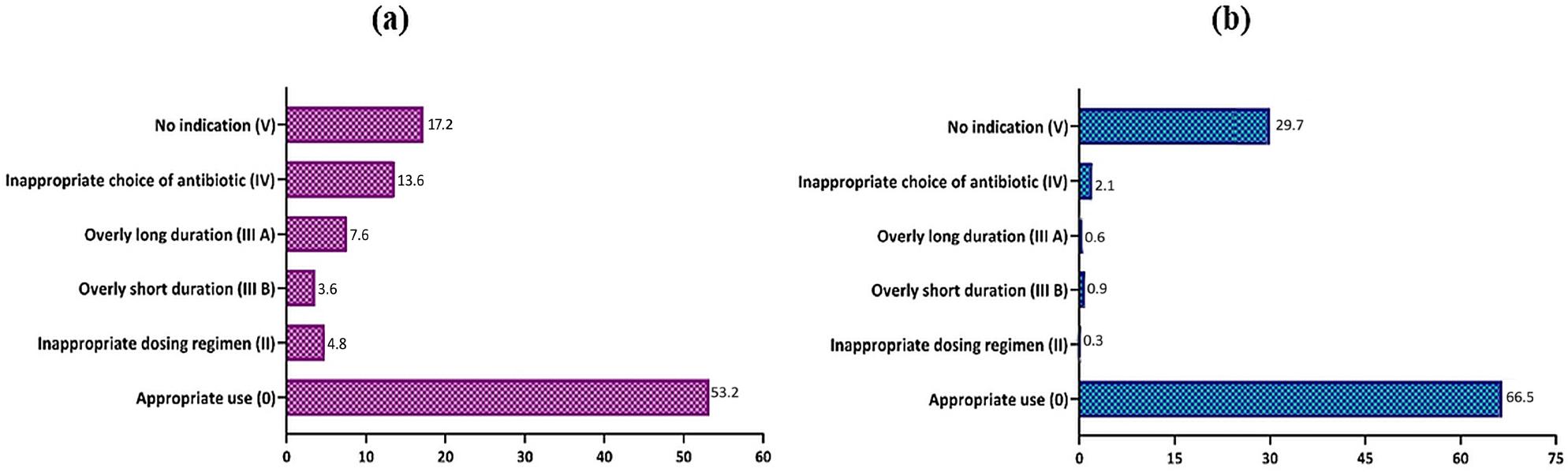
Quality of antibiotic use (% Gyssens category) in (a) surgical wards and (b) medical wards.
Gyssens category of antibiotic use in the medical wards.
| Gyssens category | Explanation | n | % |
|---|---|---|---|
| V | No indication | 101 | 29.7 |
| IV | Inappropriate selection of antibiotic type | 7 | 2.1 |
| III A | Duration of administration is too long | 2 | 0.6 |
| III B | Duration of administration is too short | 3 | 0.9 |
| II | Inappropriate dosing regimen | 1 | 0.3 |
| 0 | Appropriate antibiotic use | 226 | 66.7 |
| Total | 340 | 100 | |
The parameter for accurately reflecting antimicrobial usage is the defined daily dose (DDD), promoted by the WHO, which defines DDD as the assumed average daily maintenance dose for a drug used for its main indication in adults. To estimate the total number of days of antimicrobial therapy, healthcare personnel divided the total grams of each antimicrobial used during a given period by the World Health Organization-defined DDD for that antimicrobial. As DDD is a standardized unit of measure, it allows comparisons of antimicrobial use with other hospitals and countries (Septimus 2014). Qualitative assessment of antibiotic prescriptions can be conducted by dividing it into sub-items; for example, the appropriate dosage and administration interval can be evaluated by considering the patient’s kidney function, weight, liver function, age, and underlying disease. In addition, depending on whether culture test results are available, the appropriateness of antibiotic prescriptions can be evaluated by dividing them into empirical and definitive antibiotic prescriptions. Moreover, it is possible to evaluate whether the duration of antibiotic use is appropriate (van Den Bosch et al. 2015). A Gyssens flowchart was developed to assess the quality of the individual antimicrobial prescriptions. The outcomes were explained in terms of insufficient data; not indicated; inappropriate in efficacy, toxicity, cost, or the breadth of spectrum; inappropriate duration of treatment; and inappropriate in dosage (dose, dose interval, administration, and timing. To reach those conclusions, expert evaluation was required (Gyssens 2005).
The assessment of surgical antibiotic prophylaxis at Ayder Referral Hospital concluded that 62.2% of the selected participants received SAP, with ceftriaxone being the most commonly used SAP 85.2%. Out of those patients using ceftriaxone, 58.7% had indications for use, while 3.6% had no indication (Mohamoud and Aklilu Yesuf 2016). In a study at Nekemte Referral Hospital, approximately 59% of patients took a single prophylactic drug, and approximately 39% took a combination of two drugs. Approximately 84% of the participants received ceftriaxone, and metronidazole (35.3%) was the second most prescribed prophylactic antimicrobial, followed by ampicillin (19.6%) (Alemkere 2018). Another study, conducted at the Duc District Hospital, showed a correlation between the duration of hospital stay and the use of prophylactic antibiotics/treatment with antibiotics. The rate of patients with a treatment duration of 5 days using prophylactic antibiotics (21.0%) was lower than in patients with a treatment duration of 5 days or less (50.9%), with a statistical difference p < 0.05 (Van Tuong et al. 2021).
In this study, the highest antibiotic consumption in the surgical ward was ceftriaxone (41.6 DDD/100-BD) as empirical therapy, used in 40.89% of cases of infection in neurosurgery, orthopedics, and general surgery. Cefazoline (17.75 DDD/100-BD) was used for surgical antibiotic prophylaxis in 52.44% of cases. Furthermore, the use of definitive antibiotic therapy based on microbiological results remains low at 6.6%. The quality of antibiotic use was assessed as follows: 16.9% had no indication, 13.9% had an inappropriate antibiotic choice, and 7.6% had an overly long duration.
According to national antibiotic guidelines, prophylactic antibiotics are administered before, during, and after surgical procedures to prevent infectious complications, including surgical site infections (SSIs). SAP is used in clean and clean-contaminated surgical procedures, administered within 24 h of surgery, and the choice of SAP for surgery is generally the first-generation systemic cephalosporin group, cefazoline (MoH Indonesia, 2021).
In the medical wards, the most consumed antibiotic was ceftriaxone (106.22 DDD/100-BD) as empirical therapy in 86.94% of cases of sepsis and sepsis with comorbidities, such as diabetes mellitus, acute renal failure, chronic kidney disease, and hepatic cirrhosis. Levofloxacin (34.95 DDD/100-BD) was used as empirical therapy for pneumonia. The definitive therapy, according to microbiological culture, consists of 13.06% of cases. The quality of antibiotic use was assessed as follows: 66.5% appropriate use; 29.7% no indication for patients with comorbidities, but clinical and laboratory findings do not show any signs of infection; and 2.1% inappropriate antibiotic choice.
The selection of empiric antibiotic therapy for sepsis patients is based on broad-spectrum antibiotics because they can inhibit growth and kill all types of bacteria, both gram-positive and gram-negative. However, broad-spectrum antibiotics do not exclusively benefit patients with sepsis but can also cause side effects and life-threatening complications due to antimicrobial resistance (Martínez et al. 2020). Culture results play an important role in optimizing antibiotic therapy in patients with sepsis. According to the 2016 Surviving Sepsis Campaign (SSC) recommendations, appropriate microbiological culture results should be obtained before initiating antimicrobial therapy (Martínez et al. 2020). Rational antibiotic use can improve patient outcomes while reducing the risk of antibiotic resistance. Otherwise, the irrational use of antibiotics can increase antibiotic resistance. High rates of antibiotic resistance increase the morbidity, mortality, and health costs (Andrajati et al. 2017; Masyrifah et al. 2022).
In this study, third-generation cephalosporins and fluoroquinolones were the most commonly used antibiotics in surgical and medical wards and are often used as empirical treatments in hospitals. This condition may be implicated in the development of extended-spectrum β-lactamase (ESBL)-producing bacteria. The Global Antimicrobial Resistance and Use Surveillance System (GLASS) report 2021 showed that the prevalence of Multidrug-Resistant Organisms (MDRO) indicators in Escherichia coli and Klebsiella pneumoniae bacteria producing ESBL, as part of a multicenter hospital surveillance in Indonesia, increased from 62.2% in 2019 to 66.7% in 2020 (WHO 2022). The dominant factor in the increasing prevalence of MDRO is the excessive use of antibiotics or the use of antibiotics with no indication (Kelly and Davies 2017).
In this study, appropriate antibiotic use was observed in 53.2% of cases in the surgical ward and 66.5% in the medical ward. These figures indicate that adherence to antibiotic guidelines remains below the recommended threshold of 75%. To improve compliance with appropriate antibiotic use, hospitals are encouraged to implement an Antimicrobial Stewardship Program (AMS), which requires a multidisciplinary team. The Centers for Disease Control and Prevention (CDC) outlines seven core elements essential for the successful implementation of AMS in healthcare settings: hospital leadership commitment, accountability, pharmacy expertise, action (interventions), tracking, reporting, and education (CDC 2019). These elements are interdependent and mutually reinforcing.
To address gaps in antibiotic prescribing, AMS initiatives must prioritize effective interventions such as pre-authorization, prospective audit and feedback, and adherence to treatment guidelines. Evidence indicates that pre-authorization and prospective audit with feedback are the most effective antimicrobial stewardship interventions in hospitals. Pre-authorization strategies optimize initial antibiotic therapy, whereas prospective audits enhance ongoing treatment (CDC 2019).
Pre-authorization requires prescribers to obtain approval before initiating certain antibiotics, based on the AWaRe classification. This facilitates expert input on the indications, selection, and dosing regimens of antibiotics, which are critical in life-threatening infections such as sepsis. Moreover, it helps prevent unnecessary or excessive antibiotic use (Tamma et al. 2019). Decisions regarding which antibiotic classes to include under pre-authorization should be guided by institutional policies (Anderson et al. 2019). This intervention requires adequate staffing and expertise within the AMS team to ensure timely approvals. Hospitals can adapt available resources – human, infrastructural, and procedural – to implement pre-authorization efficiently, including the use of dedicated forms or electronic systems to prevent delays in therapy for critically ill patients (Anderson et al. 2019).
Prospective audit and feedback involve an expert team (AMS team) reviewing antibiotic therapy after it has been initiated and providing recommendations to optimize usage (MacBrayne et al. 2020). Implementation can vary based on the level of infectious disease expertise available. Institutions with limited expertise may focus on specific types of infections or antibiotic classes, while those with more advanced teams may review complex infections or regimens. The effectiveness of this intervention can be enhanced through face-to-face feedback, known as handshake stewardship (MacBrayne et al. 2020).
The Antimicrobial Stewardship Program (AMS) is a strategic, systematic, integrated, and organized activity aimed at optimizing the prudent use of antimicrobials in terms of quantity and quality. The Infectious Diseases Society of America (IDSA) and the Society for Healthcare Epidemiology of America (SHEA) recommend that restrictive interventions, limiting antibiotic use through pre-authorization of prescribing based on AWaRe categories, be used to implement effective antimicrobial stewardship, with strong recommendations and moderate-quality evidence (Mijović et al. 2018).
In conclusion, third-generation cephalosporins and fluoroquinolones are the most commonly used antibiotics in surgical and medical wards. The quality of antibiotic use showed that appropriate use accounted for 53.2–66.5% of total antibiotic use in medical and surgical wards. Establishing an antimicrobial stewardship program would help control antibiotic consumption and optimize antibiotic use in hospitals.