Numerous pathogenic diseases are currently emerging and re-emerging, posing a threat to animal health, human health, and the quality of dairy and beef products. Brucellosis is an important zoonotic illness that is emerging among most nations where people have a high dependency on livestock (Wareth et al. 2020). Marston identified brucellosis, also known as Mediterranean Gastric Remittent Fever, in Malta in 1861 (Herron and Alexander Thomas Dunbar 2018).
Brucellosis is caused by a Gram-negative, coccobacilli, facultative, intracellular, and non-motile bacterium. Brucellosis is synonymously called Bang’s disease, Mediterranean fever, Mediterranean remittent fever, Malta fever, or Undulant fever. Twelve species to date belong to the genus Brucella and are categorized based on the preference of the organism for the host. Only four of them are discussed in the literature: Brucella abortus, Brucella melitensis, Brucella suis, and Brucella canis, and have been linked to human infection (aside from biovar 2) (Yousaf et al. 2021). Cattle are the primary hosts of B. abortus, while goats and sheep are the primary hosts of B. melitensis and Brucella ovis. However, both B. abortus and B. melitensis infect humans. Human cases susceptible to the latter are more frequently documented (Bonfoh et al. 2012). Brucellosis results in significant economic losses in terms of abortions, stillbirths, placenta retention, a decrease in milk output, and low fertility rates in both males and females. Animals that are asymptomatic and suffering from the disease typically release infectious materials into the pasture or watering troughs, which are significant sources of infection for healthy animals (Ntivuguruzwa et al. 2020).
As a zoonotic illness, brucellosis primarily spreads from livestock to people (especially those in close contact with animals, such as livestock producers, dairy workers, veterinarians, slaughterhouse employees, and lab workers). The mode of transmission is either through encountering the placenta, fetus, fluid of fetus, and vaginal secretions or through contaminated animal discharges such as blood, milk, semen, and urine. Furthermore, equipment and clothes also carry the infection (John et al. 2010).
The detection of brucellosis in animals can be made with the help of disease signs and symptoms, blood tests, or cultures of contaminated materials. Different biochemical tests that are used in the diagnosis of brucellosis include the Rose Bengal Plate Test (RBPT), Milk Ring Test (MRT), Serum Agglutination Test (SAT), and Standard Tube Agglutination Test (STAT) (Godfroid et al. 2010). Molecular techniques, such as polymerase chain reaction (PCR), are also used to detect Brucella sp. from blood and serum samples. The PCR can detect a very low level of bacteria in the sample and hence is widely used as a tool for the diagnosis of infectious diseases (Gumi et al. 2013). The enzyme-linked immunosorbent assay (ELISA) test is also used, particularly when IgM and IgG antibodies need to be detected. Among various serological and molecular tests, ELISA has several advantages; it can detect incomplete antibodies commonly reported in chronic Brucella-infected patients (Pathak et al. 2014).
Asia, Africa, and Latin America still have uncontrolled brucellosis (Shahzad et al. 2017). Pakistan is an agricultural country where most people, especially from rural areas, depend on agriculture and livestock; as a result, this illness has a substantial influence on the health of humans and animals (Gwida et al. 2011). In the subcontinent, particularly in Pakistan, India, and Bangladesh, brucellosis rates remained comparatively higher. Across the country, the seropositivity of brucellosis in livestock ranged from 0% to 76% (Gul and Khan 2007).
In various parts of Pakistan, the prevalence of B. abortus has been recorded up to 3.25% (Abubakar et al. 2012). In Pakistan, investigations on brucellosis primarily rely on prevalence studies. These findings indicate that brucellosis is becoming increasingly common across the country. Recent studies show a rise in prevalence rates between 21.05% and 26.1%, compared to earlier studies suggesting rates between 0.33% and 0.65% (Shahzad et al. 2017; Saeed et al. 2019). Numerous researchers have observed brucellosis among animal handlers, with prevalence rates ranging from 0.95% to 8.58% (Munir et al. 2011). The disease prevalence among occupational workers includes veterinary surgeons (5.3–11.1%), butchers (2.5%), milkers (18.6%), and farmers (2.6–21.6%). According to published reports, this disease affects both humans and animals. A coordinated effort is needed to eliminate and control brucellosis in domesticated animals (El-Hady et al. 2016). Studies in Khyber Pakhtunkhwa (Peshawar, Charsadda), Azad Jammu, and Kashmir have been conducted to assess the prevalence of brucellosis using various diagnostic techniques (Niaz et al. 2021).
The District of Bannu is located on the Kurram River in southern Khyber Pakhtunkhwa, Pakistan. The primary sources of income and food for the people in Bannu are agriculture and livestock. Recognizing the economic significance of animal brucellosis, this study was designed to determine the seroprevalence of bovine brucellosis in the Bannu district, Khyber Pakhtunkhwa.
Due to the previously unknown status of the disease, a cross-sectional study was conducted to find out the seroprevalence of bovine brucellosis in the Bannu district, located in the Southern region of Khyber Pakhtunkhwa province of Pakistan. The district comprises an area of 1,972 km2 and has approximately one million people. In this study, a serological survey and PCR-based detection of brucellosis were conducted on cattle from different small-scale dairy farms and domestic cattle across different areas of the district of Bannu. The selection of farms and animals was based on their geographical distribution and population density.
Random blood samples were collected from 384 cattle in the targeted regions. Animals that had been vaccinated against brucellosis or were suffering from chronic or untreated diseases were excluded from the study.
A questionnaire was designed to record epidemiological data related to the risk factors of brucellosis, including age, sex, breed, management practices, health status, disease history, abortion history, and reproductive disorders (i.e., infertility, retention of fetal membrane, stillbirth, repeat breeding, etc.).
A total of 384 blood samples were randomly collected from cattle in District Bannu. Approximately 7 ml of blood was drawn from each cattle sample, with 4 ml transferred to an EDTA tube and 3 ml to the gel clot vacutainer, each labeled with a unique number. The gel clot vacutainers were positioned at a slanting position overnight and then centrifuged at 3,000 rpm for 3 minutes to separate the serum from the blood samples. After serum collection, it was transferred into Eppendorf tubes and stored in a freezer at –20°C until further serological examination. The whole blood samples collected in EDTA tubes were used for molecular characterization of Brucella pathogens using AMOS-PCR.
Two types of diagnostic tests were conducted to assess the serological status of the samples: the Rose Bengal Plate test and the Indirect ELISA. RBPT was used as an initial screening test due to its ease and rapid applicability in field conditions. Indirect ELISA was subsequently employed for its superior sensitivity and specificity, particularly useful in chronic brucellosis cases. AMOS-PCR was used for molecular confirmation and species differentiation.
The Rose Bengal test was performed to detect Brucella antibodies in serum samples (Gómez et al. 2008). The required reagents included control serum provided by the Brucella section of the Veterinary Research Institute, Lahore, and the antigen against which was sourced from the Veterinary Research Institute, Lahore. For the test, a drop of serum and a drop of Rose Bengal antigen were added to a clean glass slide and thoroughly mixed. After a few minutes, the reaction was observed under a bright light source to detect agglutination, indicating an antigen-antibody reaction. The results were classified as negative, positive, moderately positive, or strongly positive. Serum samples that tested positive or were suspected of brucellosis underwent further confirmation using an indirect ELISA.
All serum samples were further analyzed using an i-ELISA to eliminate the possibility of false-positive results. The test was conducted according to the instructions provided with the commercially available kit. The multi-species brucellosis indirect ELISA (ID Vet, 310, rue Louis Pasteur – Grabels, France) was used to detect antibodies directed against B. abortus (bovine), B. melitensis (ovine and caprine), and B. suis (pigs). For the assay, 190 μl of dilution buffer 2 was added to each well of a microtiter plate. A positive control of 10 μl was added to wells A1 and B1, and 10 μl of the negative control was added to wells C1 and D1. After that, 10 μl of each sample or a pool of 10 sera to be examined was added to the remaining wells. The microtiter plate was shaken, sealed, and incubated at 21°C (± 5°C) for 45 minutes. After incubation, the wells were emptied and washed 3 times with approximately 300 μl of the wash solution. The wells were filled to the top, rinsed, emptied, and tapped firmly to remove residual liquid. The concentrated conjugate was diluted in dilution buffer 3 at a 1:10 ratio for a short incubation. Then, 100 μl of the diluted conjugate (1×) was added to each well. The plate was sealed and incubated for 30 minutes ± 3 min at 21°C (± 5°C). Following the incubation, the wells were emptied and washed three times with approximately 300 μl of the wash solution to remove any unbound material. Subsequently, 100 μl of the substrate solution was added to each well, and the plate was incubated in the dark at 21°C (± 5°C) for 15 minutes. The reaction was then terminated by adding 100 μl of stop solution to each well. The optical density value was measured at 450 nm within 10–15 minutes after stopping the reaction. The optical density values of the positive and negative control samples and the test samples were recorded to assess the seropositivity status.
Genomic DNA was extracted from blood samples that tested positive for brucellosis in ELISA using a commercially available DNA extraction kit (QIAamp DNA Mini Kit). Electrophoresis was performed for 35 minutes at 120 V, beginning with an initial high voltage (120 V for 5 min) to facilitate DNA entry into the gel. The gel image was obtained by adjusting the brightness, contrast, sharpness, and focus to ensure clear visualization. The extracted DNA was subsequently stored at –20°C for further use.
The AMOS PCR assay was used to detect and differentiate four species of Brucella (B. abortus, B. melitensis, B. suis, and B. canis). This method was employed to evaluate the molecular characteristics of pathogens in seropositive samples (Akhtar et al. 2010). After DNA extraction and preparation of the PCR mixture, amplification was conducted using a multigenic thermocycler. The PCR process commenced with an initial denaturation at 95°C for 5 minutes (one cycle), followed by 35 cycles of annealing at 94°C for 30 seconds, annealing at 58°C for 40 seconds, and extension at 72°C for 50 seconds. A final extension step was performed at 72°C for 7 mins (one cycle). PCR results were on a 1.5% Agarose gel using a gel documentation system. Each DNA band was compared against reference strains (B. melitensis, B. abortus) and a molecular weight marker. The primers given in Table I were used in the PCR reaction.
Primer used for detection of brucellosis.
| Primer Name | Sequence (5′–3′) | Amplicon (bp) |
|---|---|---|
| Brucella abortus IS711 | F: GACGAACGCAATTTTTCCAATCCC | 498 |
| Brucella melitensis IS711 | F: AAATCGCGTCCTTGCTGGTCTGA | 731 |
| Brucella ovis IS711 | F: CGGGTTCTGGCACCATCGTCG | 976 |
| Brucella suis IS711 | F: GCGCGGTTTTCTGAAGGTGGTTCGGG | 285 |
Prevalence (%) along with the corresponding 95% confidence interval was calculated by using SPSS v25. The chi-square test was performed to determine significant differences among different risk factors of the disease. Additionally, univariate analysis was performed to calculate the odds ratio (OR).
A total of 384 blood samples were randomly collected from cattle in the Bannu district. These samples were initially screened for brucellosis using RBPT and i-ELISA. i-ELISA positive samples were further analyzed using AMOS-PCR to identify and characterize the Brucella species in the study area. The seroprevalence of brucellosis in cattle was 18.75% through RBPT and 8% i-ELISA, respectively. Molecular analysis using AMOS-PCR confirmed the presence of B. abortus in the cattle samples (Table II).
Overall seroprevalence of brucellosis in cattle through RBPT, i-ELISA in district Bannu, Khyber Pakhtunkhwa, Pakistan.
| Animals type | Total number of samples | Serological examination | Molecular identification | |
|---|---|---|---|---|
| Cattle | 384 | RBPT | i-ELISA | (AMOS-PCR) |
| 72 (18.75%) | 31 (8%) | 4 (1.04%) | ||
The breed, age, sex, and purpose of animal keeping showed a non-significant association with the seroprevalence of brucellosis (p > 0.05) as determined by the RBPT. Among the breeds, 29 out of 179 local animals (16.2%), 15 out of 72 cross-bred (20.8%), and two out of 33 exotic animals (6.1%) tested positive. In terms of age, 8.5% of animals younger than 5 years, 16.7% of those older than 5 years, and 33.3% of those older than 10 years were positive. The results indicated that males had a higher positivity rate than females, 11 out of 54 males, 20.4% testing positive, compared to 35 out of 230 females (15.2%). The purpose of animal keeping was categorized into two groups: dairy and beef. Beef animals exhibited a higher positivity rate than dairy animals, according to RBPT results. Additionally, grazing practices were significantly associated with the disease (p < 0.05). The RBPT revealed that 36 out of 256 animals with open grazing (14.1%) and 10 out of 28 with confined grazing (35.7%) tested positive.
In the breeding method, a highly significant association was found between natural mating and brucellosis. Out of 35 animals, 40% (20/50) tested positive for brucellosis with natural mating, while only 8.3% (15/180) tested positive with artificial insemination, as determined by RBPT (p < 0.05). Factors such as immunization against brucellosis, other vaccinations, health status of animals, pregnancy status, and previous calving history were non-significantly associated with the disease (p > 0.05).
The retained placenta demonstrates a strong correlation with the seroprevalence of brucellosis. In the RBPT, 25.9% of animals with a history of retained placenta tested positive, compared to 11.6% of those without retained fetal membranes. This difference is statistically significant (p = 0.0001). Among the 35 positive animals, 51.6% had a history of abortion. Additionally, repeat breeding was significantly associated with the seroprevalence of the disease (p < 0.05), with 28.6% of animals that tested positive in the RBPT having a history of repeat breeding (Table III).
Seroprevalence of brucellosis in cattle through RBPT and relationship with different disease-related risk factors.
| Variables | Category | N | RBPT (positive) | RBPT (negative) | p-value | OR | Confidence interval | |
|---|---|---|---|---|---|---|---|---|
| L.L | U.L | |||||||
| Breed | Local | 179 | 29 (16.2%) | 150 (83.8%) | 0.162 | |||
| Cross bred | 72 | 15 (20.8%) | 57 (79.2%) | |||||
| Exotic | 33 | 02 (6.1%) | 31 (93.9%) | |||||
| Age | < 5years | 47 | 4 (8.5%) | 43 (91.5%) | 0.070 | |||
| > 5 years | 222 | 37 (16.7%) | 185 (83.3%) | |||||
| > 10 years | 15 | 05 (33.3%) | 10 (66.7%) | |||||
| Sex | Male | 54 | 11 (20.4%) | 43 (79.6%) | 0.355 | 1.425 | 0.671 | 3.028 |
| Female | 230 | 35 (15.2%) | 195 (84.8%) | |||||
| Purpose | Dairy | 229 | 35 (15.3%) | 194 (84.7%) | 0.394 | 0.722 | 0.340 | 1.531 |
| Beef | 55 | 11 (20%) | 44 (80%) | |||||
| Grazing | Open grazing | 256 | 36 (14.1%) | 220 (85.9%) | 0.003* | 0.295 | 0.126 | 0.689 |
| Confined grazing | 28 | 10 (35.7%) | 18 (64.3%) | |||||
| Breeding protocol | A.I | 180 | 15 (8.3%) | 165(91.7%) | 0.0001* | 0.136 | 0.063 | 0.296 |
| Natural mating | 50 | 20 (40%) | 30 (60%) | |||||
| Immunized against brucellosis | Yes | 0 | 0 | 0 | ||||
| No | 284 | 46(16.2%) | 238(83.8%) | |||||
| Other vaccination | Yes | 257 | 42 (16.3%) | 215 (83.7%) | 0.838 | 1.123 | 0.369 | 3.415 |
| No | 27 | 4 (14.8%) | 23 (85.2%) | |||||
| Health status | Healthy | 244 | 42 (17.2%) | 202 (82.8%) | 0.251 | 1.871 | 0.632 | 5.539 |
| Emaciated | 40 | 04 (10%) | 36 (90%) | |||||
| Retained placenta | Yes | 58 | 15 (25.9%) | 43 (74.1%) | 0.009* | 2.651 | 1.252 | 5.614 |
| No | 172 | 20 (11.6%) | 152 (88.4%) | |||||
| Abortion history | Yes | 31 | 16(51.6%) | 15 (48.4%) | 0.0 001* | 10.105 | 4.326 | 23.604 |
| No | 199 | 19 (9.5%) | 180(90.5%) | |||||
| Pregnancy status | Pregnant | 78 | 7 (9%) | 71 (91%) | 0.059 | 0.437 | 0.181 | 1.051 |
| Non-pregnant | 152 | 28 (18.4%) | 124 (81.6%) | |||||
| Repeat breeding | Yes | 70 | 20 (28.6%) | 50 (71.4%) | 0.0001* | 3.867 | 1.840 | 8.126 |
| No | 160 | 15 (9.4%) | 145 (90.6%) | |||||
| Previous calving | Normal | 204 | 33 (16.2%) | 171 (83.8%) | 0.257 | 2.316 | 0.522 | 10.274 |
| Dystocia | 26 | 2 (7.7%) | 24 (92.3%) | |||||
The breed, sex, age, and purpose of animal keeping showed a non-significant association with the seroprevalence of brucellosis (p > 0.05) when assessed using indirect ELISA. In the context of grazing practices, confined grazing exhibited a highly significant association with the disease. Specifically, 25% (7 out of 28) of animals with confined grazing tested positive for the disease through i-ELISA, compared to only 3.9% (10 out of 256) of those with open grazing. Regarding breeding methods, a significant association was identified between natural mating and brucellosis. Out of 230 animals, 16% (8 out of 50) were found to be positive for natural mating, while only 2.2% (4 out of 180) tested positive for artificial insemination, as determined by indirect ELISA (p < 0.05). Factors such as immunization against brucellosis, other vaccinations, the health status of animals, pregnancy status, retained placenta, and previous calving history showed no significant association with the disease (p > 0.05).
Abortion was significantly associated with the seroprevalence of brucellosis. Among the animals with a history of abortion, 29% (9 out of 31) tested positive using the i-ELISA, compared to only 1.5% (3 out of 199) of those without such a history. Additionally, repeat breeding was also significantly associated with the seroprevalence of the disease (p < 0.05). Of the animals that tested positive via i-ELISA, 12.9% (9 out of 70) had a history of repeat breeding, while only 1.9% (3 out of 160) of those without a repeat breeding history tested positive (Table IV).
The seroprevalence of brucellosis in cattle using i-ELISA and relationship with different disease-related risk factors in district Bannu, KPK, Pakistan.
| Variables | Category | N | i-ELISA (positive) | i-ELISA (negative) | p-value | OR | Confidence interval | |
|---|---|---|---|---|---|---|---|---|
| L.L | U.L | |||||||
| Breed | Local | 179 | 11 (6.1%) | 168 (93.9%) | 0.245 | |||
| Cross bred | 72 | 06 (8.3%) | 66 (91.7%) | |||||
| Exotic | 33 | 0 (0%) | 33 (100%) | |||||
| Age | < 5 years | 47 | 3 (6.4%) | 44 (93.6%) | 0.453 | |||
| > 5 years | 222 | 12 (5.4%) | 210 (94.6%) | |||||
| > 10 years | 15 | 02 (13.3%) | 13 (86.7%) | |||||
| Sex | Male | 54 | 05 (09.3%) | 49 (90.7%) | 0.26 | 1.854 | 0.624 | 5.504 |
| Female | 230 | 12 (5.2%) | 218 (94.8%) | |||||
| Purpose | Dairy | 229 | 12 (5.2%) | 217 (94.8%) | 0.28 | 0.553 | 0.186 | 1.641 |
| Beef | 55 | 05 (9.1%) | 50 (90.9%) | |||||
| Grazing | Open grazing | 256 | 10 (3.9%) | 246(96.1%) | 0.001* | 0.122 | 0.042 | 0.353 |
| Confined grazing | 28 | 07 (25%) | 21 (75%) | |||||
| Breeding protocol | AI | 180 | 04 (2.2%) | 176(97.8%) | 0.0001* | 0.119 | 0.034 | 0.415 |
| Natural mating | 50 | 08 (16%) | 42 (84%) | |||||
| Immunized against brucellosis | Yes | 0 | 0 | 0 | ||||
| No | 284 | 17 (6%) | 267 (94%) | |||||
| Other vaccination | Yes | 257 | 14 (05.4%) | 24 3(94.6%) | 0.238 | 0.461 | 0.124 | 1.718 |
| No | 27 | 03 (11.1%) | 24 (88.9%) | |||||
| Health status | Healthy | 244 | 17 (07%) | 227 (93%) | 0.085 | 1.176 | 1.118 | 1.237 |
| Emaciated | 40 | 0 | 40 (100%) | |||||
| Retained placenta | Yes | 58 | 03 (5.2%) | 55 (94.8%) | 0.986 | 0.988 | 0.258 | 3.78 |
| No | 172 | 09 (5.2%) | 163 (94.8%) | |||||
| Abortion history | Yes | 31 | 09 (29%) | 22 (71%) | 0.0001* | 26.727 | 6.731 | 106.128 |
| No | 199 | 03 (1.5%) | 196 (98.5%) | |||||
| Pregnancy status | Pregnant | 78 | 03 (3.8%) | 75 (96.2%) | 0.503 | 0.636 | 0.167 | 2.418 |
| Non-pregnant | 152 | 09 (5.9%) | 143(94.1%) | |||||
| Repeat breeding | Yes | 70 | 09 (12.9%) 03 (1.9%) | 61 (87.1%) | 0.001* | 7.721 | 2.022 | 29.479 |
| No | 160 | 0 | 157 (98.1%) | |||||
| Previous calving | Normal | 204 | 12 (5.9%) | 192 (94.1%) | 0.204 | 1.135 | 1.081 | 1.192 |
| Dystocia | 26 | 0 | 26 (100%) | |||||
DNA extracted from positive samples of ELISA and molecular detection of Brucella species was done through AMOS PCR. In molecular analysis through AMOS PCR, four samples of cattle were found to be positive for B. abortus (Fig. 1 and 2).
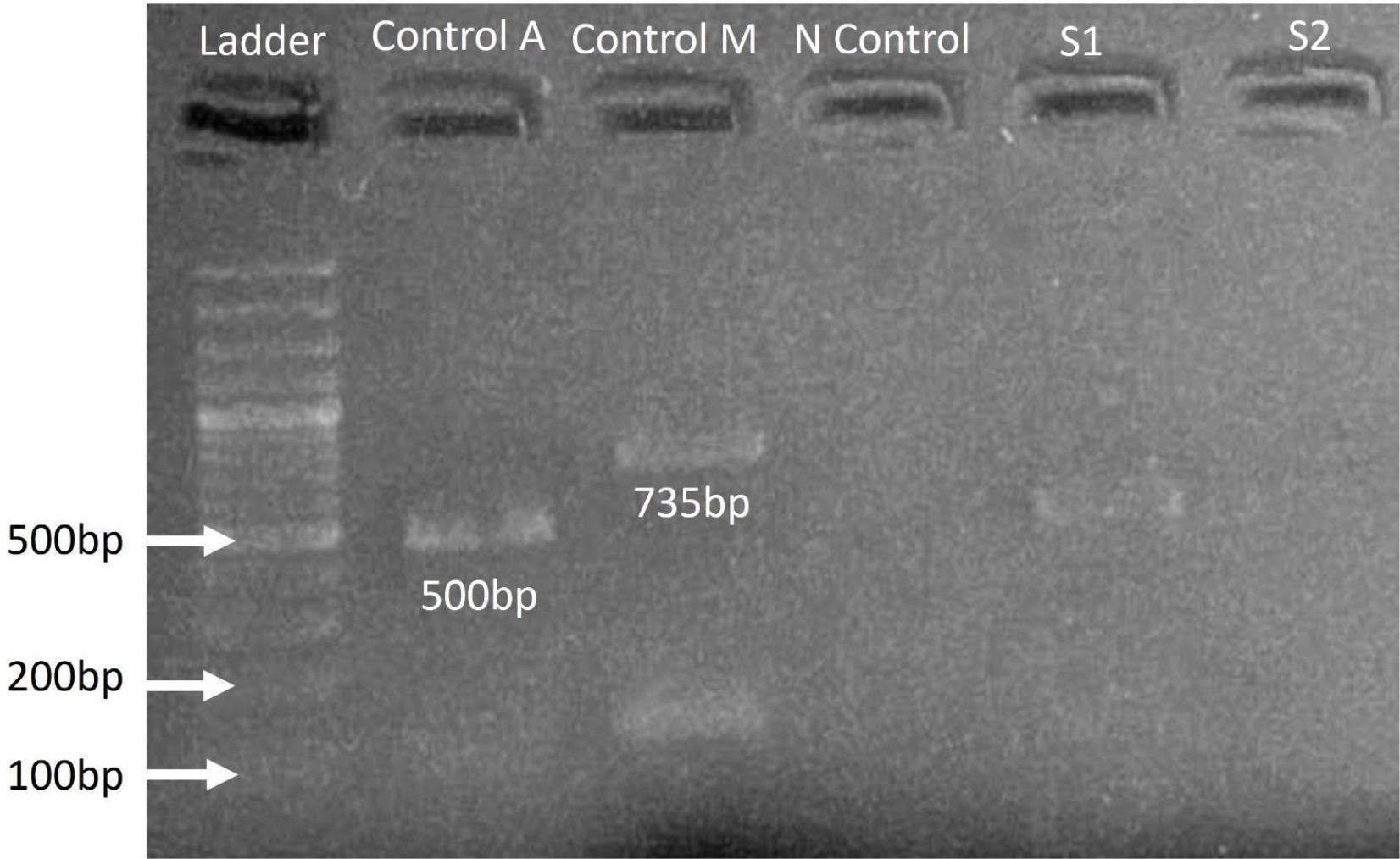
Demonstration of Brucella abortus species in the cattle samples. The results of the B. abortus AMOS polymerase chain reaction. In a sample taken from cattle in the province of Bannu, Khyber Pakhtunkhwa, Pakistan, the amplification product of the IS711 gene showed a band size of 498 bp, with a M = 100 bp DNA ladder indicating positive samples S1 and S2.
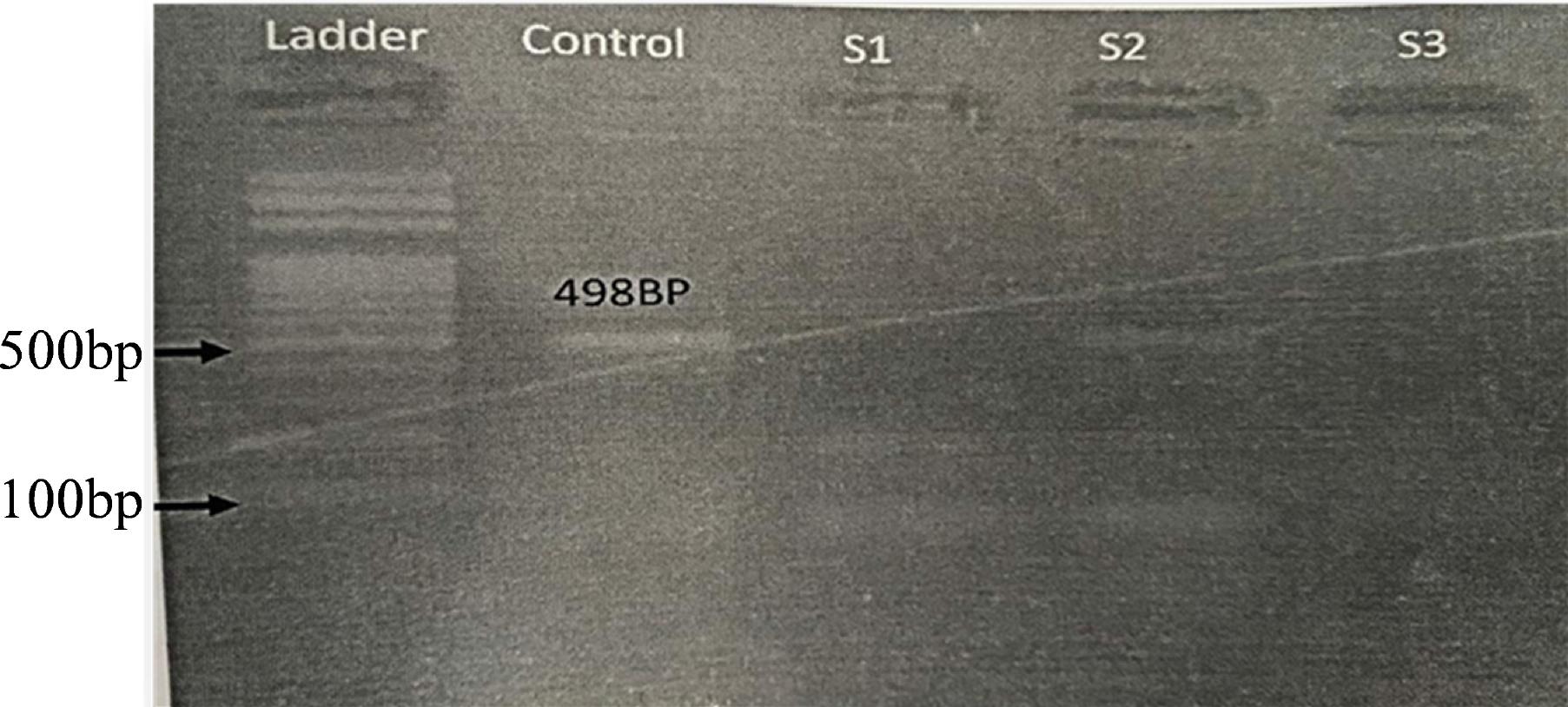
The AMOS polymerase chain reaction results for Brucella abortus. The amplification product of the IS711 gene depicted a band size of 498bp in a sample collected from cattle in district Bannu, Khyber Pakhtunkhwa, Pakistan, M = 100 bp DNA ladder, S2 showing positive samples.
Brucellosis, an eminent zoonotic ailment, stands out as a prevalent affliction inflicting significant suffering upon both animals and humans worldwide. It holds the position among zoonotic bacterial infections, occupying the pinnacle of recognition, with a staggering annual incidence of approximately half a million cases reported in endemic regions (Khan and Zahoor 2018). It is a highly contagious zoonotic disease that manifests as a widespread concern, with the potential for transmission between animals and humans. The disease commonly spreads through the ingestion and inhalation of pathogens present in infected animals and their products. Reports of Brucella infections have emerged from diverse regions of the country. To effectively eliminate brucellosis from cattle populations, accurate diagnosis and thorough baseline surveillance assume paramount importance (Fero et al. 2020).
The present investigation was devised for brucellosis detection in the district of Bannu, Khyber Pakhtunkhwa, Pakistan, through a combination of serological techniques, namely RBPT and iELISA, along with molecular testing using the AMOS PCR for differentiation of species prevailing in cattle and livestock-related persons. A total of 384 blood samples were collected from cattle and screened for brucellosis. The overall prevalence of brucellosis in cattle was 18.75% by RBPT and 8% by i-ELISA. Through AMOS PCR, four ELISA-positive cattle samples showed 1.04% B. abortus species.
In the current investigation, the overall incidence of brucellosis in cattle by RBPT aligns with the results of (Khan et al. 2017), in which Brucella seroprevalence was 17% in animals. In the current research, a higher prevalence was spotted in cross-bred animals older than five years. A cross-sectional epidemiological survey conducted by Gumi et al. (2013) found a higher seroprevalence of brucellosis among older animals compared to younger ones. This aligns with Swai and Schoonman et al. (2010), who proposed that animals aged 6 years and above exhibited higher seropositivity compared to those below the age of 6 years. In pregnant animals, the production of erythritol in the placenta creates a favorable environment for the rapid multiplication of bacteria, resulting in conditions like endometritis, infections of cotyledons, and placentitis. This phenomenon is the result of several factors, including an impaired immune system brought on by disease, aging, and pregnancy in animals (Gul and Khan 2007). 40% of animals that are positive through RBPT have been conceived through natural mating. 51.6% of animals that have an abortion history are positive. Our findings align with (Miller et al. 2016), wherein brucellosis was found to be responsible for 13.8% of bovine abortions. However, Otlu et al. (2008) demonstrated a significantly greater percentage of cattle, this alteration in the seroprevalence is thought to be due to the variations in the immune status of animals under investigation, Shahzad et al. (2017) nearly support the findings of the present study, which reported in their study that the percentage of seroprevalence was 9.3% by applying the RBPT test, whereas Magona et al. (2009) disagree with our findings. They reported the animal-related prevalence as 5% using RBPT and SPAT.
The overall appearance of disease in cows through indirect ELISA is 8%. In managemental practices, confined grazing and natural mating in breeding protocols are associated with a highly significant increase in the seroprevalence of the disease in animals with abortion and repeat breeding history in our study. Animals above the age of 10 years show more positivity at 13.3%. Similarly, male animals have a higher prevalence (9.3%) than female animals. Our study links with Pathak et al. (2014), who reported the prevalence in occupationally exposed individuals. He also determined a 6% prevalence in humans by indirect ELISA. Indirect ELISA exhibited the lowest rate of brucellosis positivity, whereas the RBPT demonstrated a higher positivity rate. This could be due to i-ELISA being a quantitative test that specifically detects IgG, while RBPT and SPAT measure both IgG and IgM qualitatively (Valarmathy et al. 2007).
The introduction of infected animals to farms without going through the proper brucellosis screening tests, the failure to separate and eliminate animals with a high abortion rate, frequent mixing with infected flocks, insufficient disposal of aborted fetuses and placental membranes, and the consumption of contaminated water and feed by healthy animals all contribute to the observed seroprevalence (Sadhu et al. 2015). The increased sensitivity of ELISA in detecting anti-Brucella antibodies in different animals has been investigated for many years. Several studies have found that indirect ELISA is more sensitive than traditional assays. Those samples that were ELISA positive had their DNA extracted and subjected to AMOS PCR. The Brucella AMOS PCR assay is used to identify and differentiate four species of Brucella bacteria (B. abortus, B. melitensis, B. suis, and B. canis). AMOS-PCR is utilized to evaluate the molecular characterization of pathogens in seropositive samples (Kolo et al. 2019). AMOS PCR detected only one Brucella, and that was B. abortus in four samples of cattle. Pathak et al. (2014) supported our study by determining the brucellosis prevalence in occupationally exposed individuals and identifying only one species of Brucella, B. abortus. Saeed et al. (2019) performed qRT-PCR to determine the seroprevalence and predisposing factors for caprine, ovine, and bovine brucellosis. They identified B. melitensis in positive DNA samples. Ali et al. (2013) strongly linked us as they experimented to establish the seroprevalence of human brucellosis and identify risk factors. According to them, the overall seropositivity rate is 6.9%, and PCR techniques have determined that all patients were infected with B. abortus; those who consumed high rates of unpasteurized raw milk had a higher prevalence. Our study also revealed the same factors and identified those who have contact with animal aborted material are at greater risk and were positive through serological tests. PCR detects DNA in minimal quantities within blood samples, regardless of a high antibody titer. In contrast, the serum titer cannot be detected. However, the sample’s DNA content may be sufficient for PCR detection. Due to its capacity for antigenic detection rather than antibody detection, PCR is more dependable and sensitive (Akhtar et al. 2010). The combined use of RBPT, ELISA, and PCR allowed for robust detection across the acute and chronic phases of brucellosis. This tiered diagnostic approach increases reliability, especially in low-resource and endemic settings.
In the current investigation, the seroprevalence rate exhibited no significant variation between male and female animals. Interestingly, the absence of erythritol in a male’s reproductive system renders them less susceptible to infection (Jain et al. 2013). The higher occurrence of brucellosis in males can be attributed to the fact that most infected animals have reached sexual maturity, and male animals were frequently employed for breeding purposes. Additionally, a smaller number of samples were collected from male animals in the research area, which could influence the frequency of recorded Brucella cases in males. As indicated by Saeed et al. (2019) livestock owners often refrain from culling or removing brucellosis-infected animals from their herds. Instead, these are utilized for breeding purposes or sold to other ranchers. This practice provides a plausible explanation for the high prevalence of brucellosis in males.
In the present study, managemental practice grazing shows a highly significant association with the prevalence of the disease. Detecting Brucella species in the research area signifies the potential occurrence of interspecies transmission. Notably, factors such as shared watering spots, pasteur lands, and the mixing of sheep and goats with cattle can serve as sources for the transmission of brucellosis among different animal species. Moreover, the presence of brucellosis positivity in cattle is believed to be a result of natural infection since vaccination practices are not implemented in the research area of Pakistan.
In our research, seroprevalence exhibited a significant difference between animals with a history of abortion and those without. The prevalence of brucellosis among animals with a history of abortion was recorded at 29%, whereas in non-aborted animals, the prevalence stood at 1.5%, as determined through the indirect ELISA. Abortion at the late stages of pregnancy emerged as the most common clinical mark of brucellosis in breeding animals. In our study, we observed that animals that had previously experienced an abortion displayed a higher likelihood of infection compared to those that had never encountered such a complication. These findings align closely with the research conducted by Ali et al. (2013) who reported a significant correlation between brucellosis and abortions occurring during the final trimester of pregnancy in cattle.
Brucellosis remains a persistent problem among cattle in the country and specifically in the district of Bannu in Khyber Pakhtunkhwa, Pakistan. This study showed an overall prevalence of brucellosis of 18.75% using the Rose Bengal plate test and 8% by indirect ELISA, with B. abortus identified as the causative agent in Bannu’s cattle population. This presents a direct zoonotic risk to animal handlers and milk consumers in the region. Therefore, raising public awareness about the risks of brucellosis among the general public and high-risk groups is essential. Additionally, implementing improved management practices is highly recommended to reduce associated risk factors. Conducting a thorough screening of newly introduced animals before integrating them into herds is crucial to prevent disease introduction and spread. There is an urgent need to initiate a proper vaccination program, and a door-to-door vaccination campaign could significantly help control the disease’s occurrence. Addressing brucellosis effectively requires an integrated approach within the “One Health” framework, which unites multiple disciplines to ensure efficient control and eradication. As part of this strategy, isolating the prevalent Brucella species for further detailed studies is advised. Whole-genome sequencing and molecular epidemiology will aid in detecting new species and developing vaccines, which are vital steps for controlling the disease.
One limitation of this study is its focus on a single district, which might not fully reflect brucellosis prevalence trends in other parts of Pakistan. Future research involving multiple districts or provinces would improve the representativeness of the findings and help confirm the risk factors on a larger scale. Although several risk factors, such as abortion history, breeding methods, and grazing practices, were analyzed, factors like the disposal of aborted fetuses, water source contamination, and the introduction of unscreened animals were not directly examined and remain limitations. These will be addressed in future studies.