Cancer therapy-related cardiac dysfunction (CTRCD) is an important form of cardiovascular toxicity observed in patients treated by cardiotoxic drugs. CTRCD can include a wide range of potential presentations, such as cardiomyopathies and heart failure [1]. Various chemotherapy agents can cause CTRCD, with anthracyclines and human epidermal growth factor receptor 2 (HER2)-targeted therapies being among the most frequently associated, particularly due to the high incidence of breast cancer requiring treatment [2]. Cisplatin is a platinum-based chemotherapeutic agent, used in many types of cancer, particularly favored for its efficacy against several solid tumors and malignancies, including lymphomas, leukemia, breast cancer, cervical cancer, ovarian cancer, testicular cancer, head and neck cancers, and sarcomas [3]. In fact, it is primarily known as a vasculotoxic agent, associated with a high risk of developing acute coronary syndromes, arterial thromboses (such as peripheral artery disease or stroke), and hypertension [1]. Despite the limited frequency, several case reports have described mild to moderate myocardial dysfunction attributed to cisplatin-based chemotherapy [4], particularly when it is administered in combination with other cardiotoxic agents [5].
Evaluation of baseline cardiovascular (CV) toxicity risk prior to the initiation of anticancer treatment is essential to determine the pretreatment risk of developing cancer therapy-related cardiovascular toxicity. According to current risk assessment scores, the presence of cardiac dysfunction, cardiomyopathy, or heart failure categorizes patients as high-risk and therefore requires close monitoring and secondary prevention strategies [1].
Dilated cardiomyopathy (DCM) is characterized by left ventricular (LV) enlargement and a reduction in systolic function, which cannot be attributed solely to coronary artery disease, hypertension, valvular disease, or congenital heart conditions[6]. The etiology of DCM is diverse and includes both inherited causes and acquired contributors. It is estimated that up to 40% of dilated cardiomyopathy cases may have a genetic origin[7]. In these patients, it is known that some external factors, such as pregnancy or exposure to toxins like excessive alcohol consumption or chemotherapeutic agents, can trigger or accelerate DCM, and this is called “the second hit theory” [8]. Identification of cardiomyopathy in young cancer patients who have undergone oncological treatment may have a genetic basis. It is known that these patients harbor rare, unrecognized variants, with titin-truncating variants (TTNtv) being the most commonly implicated, occurring in approximately 10% of these patients [9].
We present the case of a 54-year-old female patient, without any CV risk factors, who was brought to the emergency room with shortness of breath at mild exertion, which had started one month before and had progressively worsened in the last 2 weeks. Her medical history includes six series of cisplatin-based chemotherapy, local radiotherapy, and total hysterectomy with bilateral salpingooophorectomy for locally advanced invasive cervical carcinoma at 47 years old. After one year, she developed a post-radiotherapy vesicovaginal fistula, with failed surgical correction, and now has bilateral ureterostomy. She had never undergone a cardiologic evaluation before starting chemotherapy.
The cardiac physical examination highlighted a grade 2/6 systolic murmur, best heard at the apex, mild leg swelling, a low blood pressure of 90/60 mmHg, and a heart rate of 64 bpm, with rhythmic heart sounds. Biologically, she presented elevated NTproBNP of 5453pg/ml, normal level of troponin, and elevated serum creatinine level of 1,6 mg/dl, corresponding to a glomerular filtration rate of 35 ml/min/1.73 m2. The electrocardiography noted sinus rhythm, with normal QRS axis, narrow QRS (95ms), T-wave inversion in leads V4-V6, DII, DIII, and aVF (Figure 1A), with a corrected QT interval of 470ms; without any significant arrhythmia during 24-hour Holter monitoring.
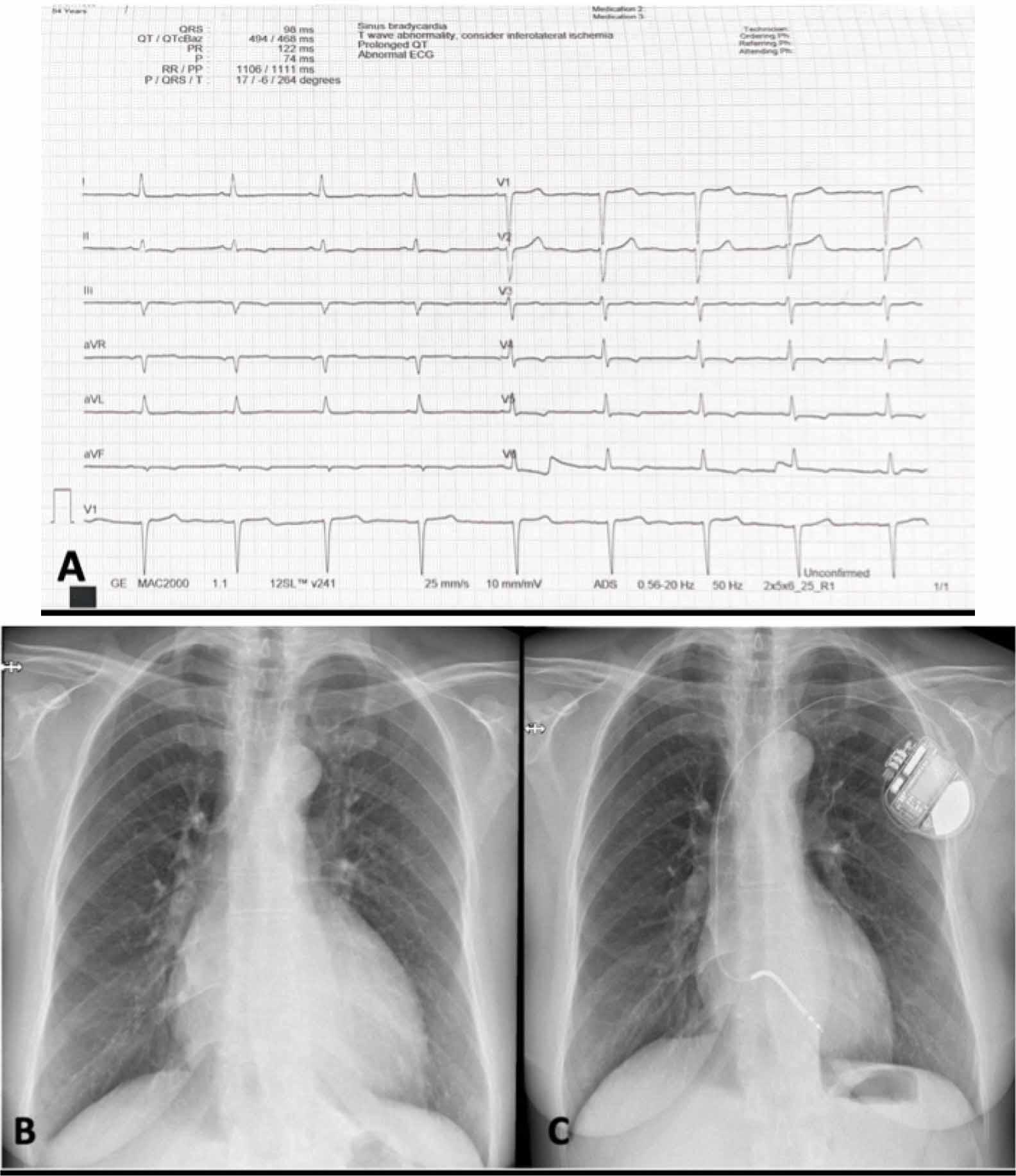
Electrocardiogram findings showing T wave inversion in inferior and lateral leads with a QTc of 470 ms (A), Admission chest X-ray reflecting prominent convexity of the lower right and left arches suggestive for left atria, respectively left ventricle enlargement, and marked interstitial pulmonary pattern (B), After 3 months, the post-implant radiography of the patient with unicameral implantable cardioverter-defibrillator (C).
At presentation, transthoracic echocardiography showed dilated LV with severe diffuse hypokinesia of the walls, an estimated ejection fraction of 15% (Supplementary material online, Video 1), with severe mitral functional regurgitation. Additionally, she presented global and longitudinal RV systolic dysfunction, defined by a fractional area change (FAC) of 29% and a tricuspid annular systolic velocity of 8.3 cm/s, and also a moderate tricuspid regurgitation, with systolic pulmonary pressure estimated at 35 mmHg, which indicates a low probability for pulmonary hypertension (Figure 2).
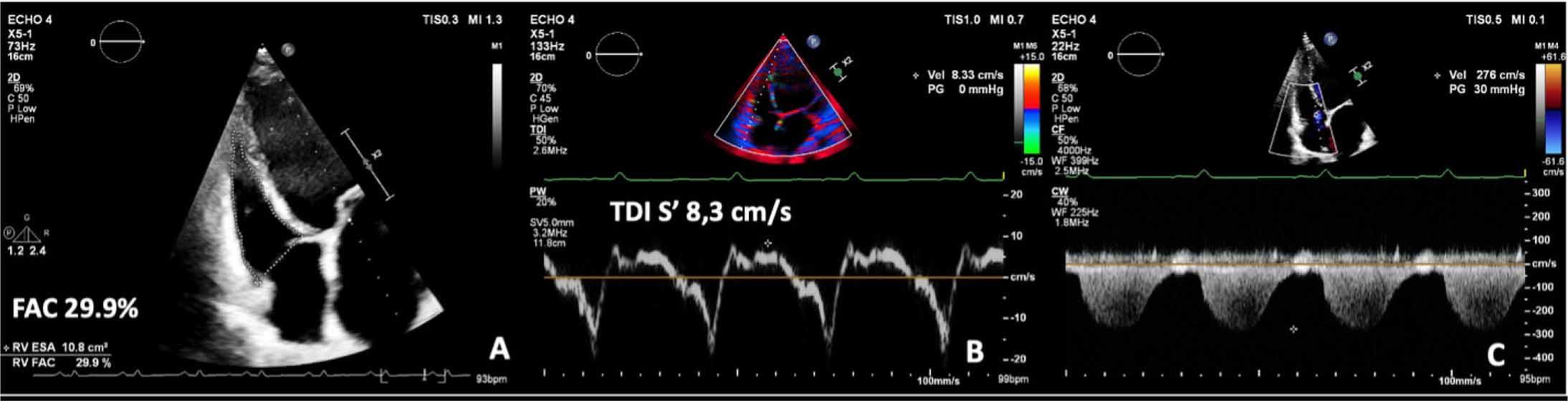
Assessment of right ventricle function showing a fractional area change (FAC) estimation of 29.9% (A) and a tricuspid annular systolic velocity (S′) by tissue Doppler imaging (TDI) of 8,33 cm/s; a peak velocity of the tricuspid regurgitation envelope of 2,76 cm/s indicates a low probability for pulmonary hypertension (C).
An ischemic etiology was considered but deemed unlikely, given the age of the patient, without cardiovascular risk factors or symptoms suggestive of angina, and invasive coronary evaluation was therefore not considered necessary. There was also no history of recent viral infection or alcohol use, and inflammatory markers were within normal limits, making viral or alcohol-related cardiomyopathy less probable.
We initiated treatment with intravenous loop diuretics administered intermittently for three days until euvolemia was achieved. Subsequently, she was transitioned to an oral diuretic regimen (furosemide 40 mg once daily). Additionally, she was started on optimal heart failure therapy for reduced ejection fraction at the maximum tolerated doses, including a beta-blocker (bisoprolol 5 mg once daily) and a mineralocorticoid antagonist (MRA - spironolactone 25 mg once daily). Considering the presence of a bilateral ureterostomy, a bacteriologic examination was performed, revealing a positive result for Enterococcus faecalis. Following antibiotic treatment and confirmation of a sterile urine culture, a sodium-glucose cotransporter-2 (SGLT2) inhibitor (dapagliflozin 10 mg once daily) was introduced. Despite ESC guideline recommendations for sacubitril/valsartan as first-line therapy, symptomatic hypotension (80 mmHg) occurred even at the lowest dose of ramipril (2.5 mg once daily), which precluded ARNI initiation. Additionally, because she remained with a reduced LVEF of less than 35% after 3 months of maximally tolerated optimal therapy, a unicameral implantable cardioverter-defibrillator (ICD) was considered for primary prevention of SCD (Figure 1C).
When looking closer at her familial history, it was significant for SCD of her mother at 54 years old, who also was diagnosed with cardiomyopathy, and her brother with SCD at 34 years old (Figure 3). Considering these findings and the disproportionately severe systolic dysfunction, which could not be solely attributed to the cardiotoxic effects of alkylating agents, we performed genetic testing for a DCM panel, which detected a heterozygous variant of pathogenic significance in the TTN gene (c.64915C>T, p.(Arg21639Ter)).
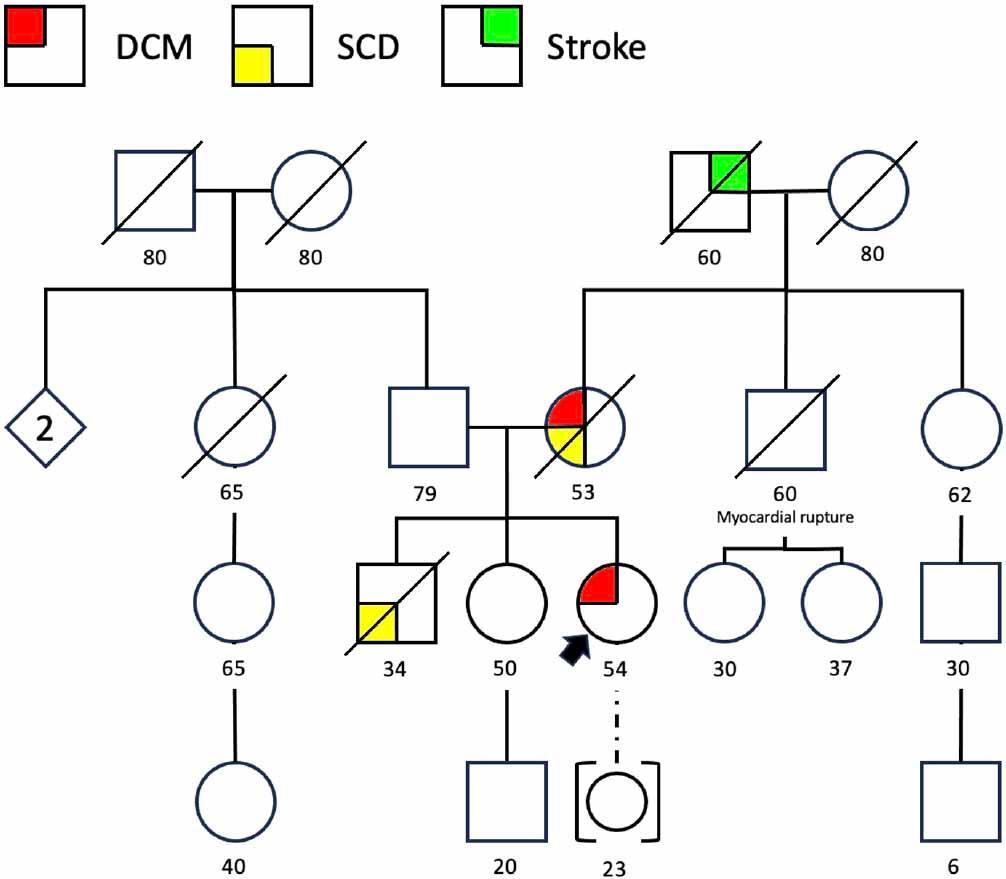
The pedigree of the family shows, with the black arrow, the proband with dilated cardiomyopathy (DCM) and her familial history of cardiomyopathy and sudden cardiac death (SCD) of her mother at 53 years old and SCD of her brother at 34 years old.
After one year of follow-up, being on maximal tolerated optimal therapy (SGLT2 inhibitor, MRA, and betablocker), she presented clinical and biological improvement. The patient had a significant improvement of symptoms, along with an important decrease in NTproBNP levels from 5453pg/ml to 672 pg/ml (Figure 3E). Also, the TTE examination revealed a slightly improved ejection fraction to 22% (Supplementary material online, Video 2), along with a significant improvement in stroke volume estimated by left ventricular outflow tract velocity-time integral, recovered RV systolic function, and the severity of mitral regurgitation was mitigated to a mild-moderate degree (Figure 4).
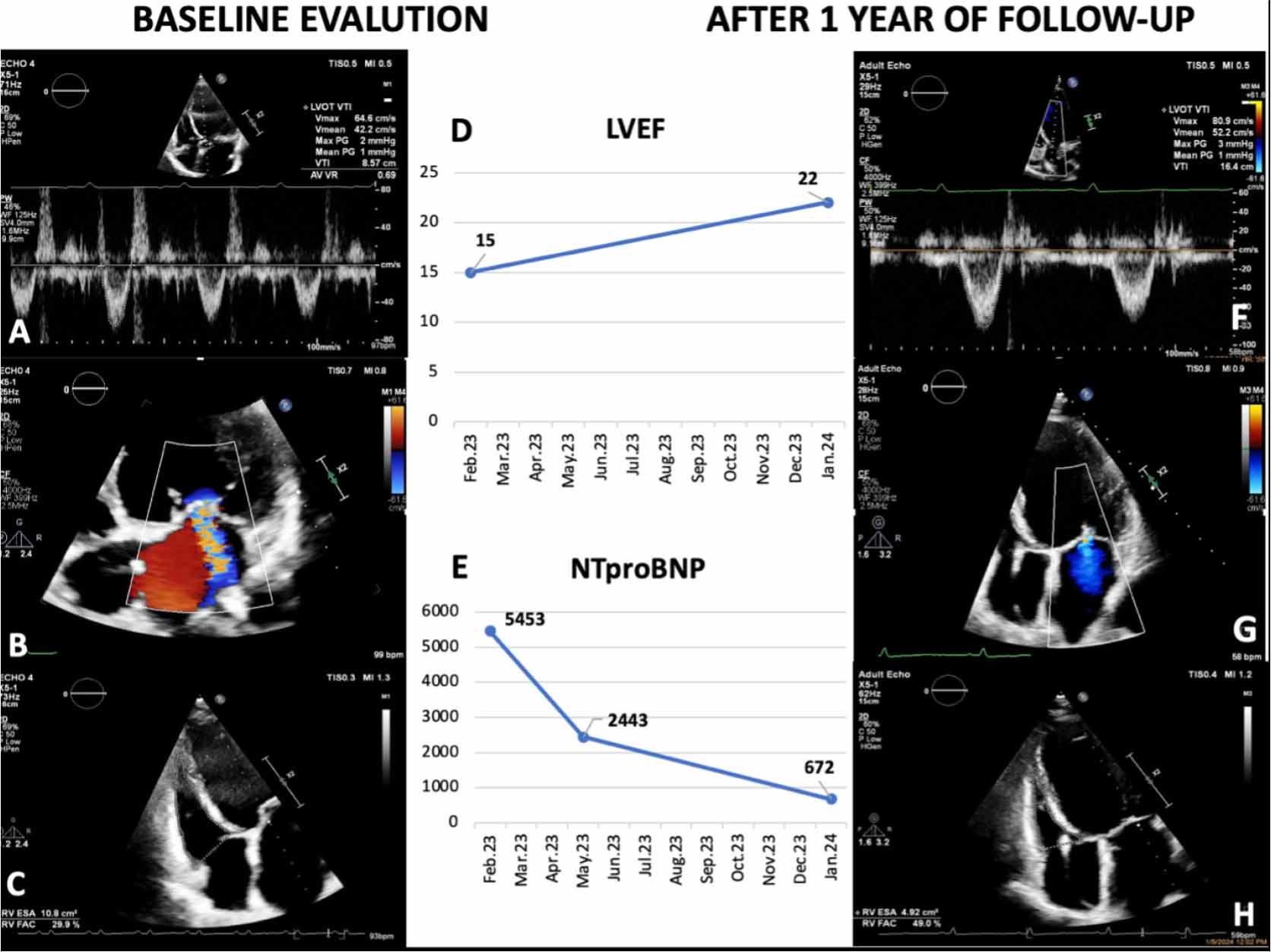
Dynamic evolution of echocardiographic and biological parameters at baseline and after one year of optimal treatment. Left ventricular outflow tract velocity-time integral (LVOT VTI) measured 8.5 cm at baseline (A) and 16.4 cm after one year (F). A two-dimensional view of the severe mitral regurgitation jet in color Doppler imaging in the apical 4-chamber view showed a turbulent jet with a large proximal flow convergence region extending deep into the left atrium (B), which was alleviated to a mild-moderate degree after 1 year (G). Assessment of right ventricular function showed a fractional area change (FAC) estimation of 29.9% at baseline (C) and 49% after one year. The dynamic changes of left ventricle ejection fraction (D) and NT-proBNP (E) at admission and after one year of treatment.
This case highlights the importance of baseline CV toxicity risk assessment prior to initiating anticancer therapy, together with a structured follow-up during cardiotoxic medication, and the challenging ways of diagnosing inherited DCM, which can be revealed by external factors, such as chemotherapeutic agents that accelerate the disease.
For an accurate diagnosis of dilated cardiomyopathy, it is essential to exclude ischemic cardiomyopathy and structural heart disease [6]. In the absence of any CV risk factors, specific symptoms, or any personal history of coronary heart disease, ischemic cardiomyopathy is unlikely. Given the patient’s age, family history, and the severity of the DCM phenotype, we performed a genetic test that identified a TTN gene mutation.
Titin is one of the most frequently known genes associated with inherited DCM, occurring in 25-30% of patients [10]. Moreover, the TTNtv mutations are a significant contributor to these cases, being present in approximately 25% of familial cases and 18% of non-familial cases [10]. Even though, in our patient, we cannot predict the moment of disease onset without a prior cardiological examination, we know that these mutations exhibit age-related penetrance, with a higher incidence observed after the age of 40-50 years[11].
When DCM is expressed, the presence of TTNtv indicates a worse prognosis, characterized by significantly impaired LVEF, reduced stroke volumes, and thinner LV walls compared to patients without TTNtv. Furthermore, in patients with TTNtv cardiac transplantation listing, the need for LV assist device, and all-cause mortality have occurred at younger ages and shortly after enrollment [12].
After 40 years old, the penetrance of TTNtv can be as high as 82-95% in familial contexts; these patients often exhibit subclinical changes that may escalate to heart failure when exposed to physiological stress or specific toxins like alcohol or cardiotoxic drugs [11]. In the general population, TTNtv variants are present in approximately 1% of individuals, but most do not develop DCM, indicating that most TTNtv are nonpathogenic or have very low penetrance outside a familial context [13]. Oncological treatment may significantly accelerate and precipitate the onset of inherited cardiomyopathy. Therefore, a study found that TTNtvs were most prevalent among oncological patients with induced cardiomyopathy, occurring in 7.5% of these patients, compared with 1.1% of individuals with breast and lung malignancies and 0.7% of healthy participants [9]. The challenge in our case, given the absence of a pre-treatment TTE evaluation, lies in determining whether DCM preceded the administration of chemotherapy or it was triggered by chemotherapeutic agents.
Cisplatin chemotherapy with concurrent radiotherapy is the preferred option for patients with local (for stage IB3, IIA2) and regional cervical cancer [14]. The underlying mechanisms of cisplatin-induced myocardial contractile dysfunction are only partially understood. It is known that an important target of cisplatin in malignant cells is the mitochondria, resulting in a decrease in its function[15,16]. Thus, in cisplatin-treated mice, cardiomyocyte dysfunction appears to be associated with alterations in mitochondrial membrane depolarization, leading to impaired function. This results in oxidative stress activation, increased production of reactive oxygen species, and decreased synthesis of adenosine triphosphate, which is needed for the contractile function of the cardiomyocyte. Also, another explanation is that cisplatin interferes with the activation of the endoplasmic reticulum signaling pathway, leading to an increase in caspase 3 activity and the induction of cardiomyocyte apoptosis [17,18].
In a clinical study of 110 cisplatin-treated testicular cancer patients, the survivors were followed up long-term (with a median time of 30 years), and they predominantly presented diastolic dysfunction, and more of them experienced heart failure with preserved EF. Also, the LVEF and global longitudinal strain were measured, and no evident or subtle decline in systolic function was detected. This indicates that the risk of long-term cisplatin-induced heart failure remains low [19,20]. Despite these findings, several case reports have described mild to moderate myocardial dysfunction, typically as an acute side effect, that tends to be reversible after discontinuation of cisplatinbased chemotherapy [4,20]. The administration of other cardiotoxic drugs and prior mediastinal radiotherapy appear to be more closely associated with the development of reduced LVEF [5].
The most crucial action in the management of cardiotoxicity is prevention. It implies important stages such as baseline and dynamic cardiac monitoring during and after treatment, detection of subclinical disease, initiation of prophylactic cardioprotective therapy, alternative chemotherapy regimens, or discontinuation of chemotherapeutic agent [18]. Additionally, there are some ongoing research aims to mitigate the side effects of chemotherapy, including the development of nanocarriers created for target delivery of the drug specifically to malignant cells, while shielding healthy cells from exposure, including cardiac cells [21]. Furthermore, due to the systemic toxicities associated with intravenous cisplatin chemotherapy, research has been conducted proposing the development of a device that releases cisplatin directly intra-cervically, thereby reducing systemic side effects [22]. Further research is needed to achieve a balance between the most efficient cancer treatment and the minimal cardiotoxicity.
Cisplatin-induced myocardial dysfunction is a rarely reported form of cardiotoxicity with underlying mechanisms that are not fully understood. Left ventricular myocardial dysfunction seems to be an early and reversible side effect after drug cessation, mostly associated with concomitant administration of other chemotherapy agents. A comprehensive baseline cardiovascular evaluation, including family history and, when indicated, genetic testing, may allow early detection of high-risk patients and optimize their management through timely intervention, individualized therapy, and structured follow-up.