FIGURE 1.
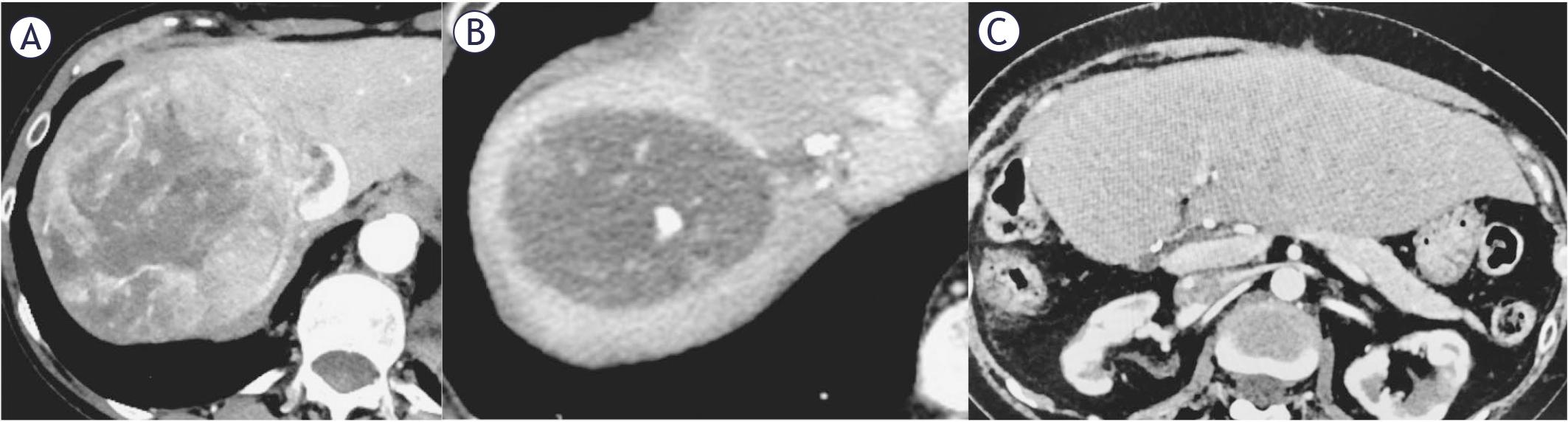
FIGURE 2.
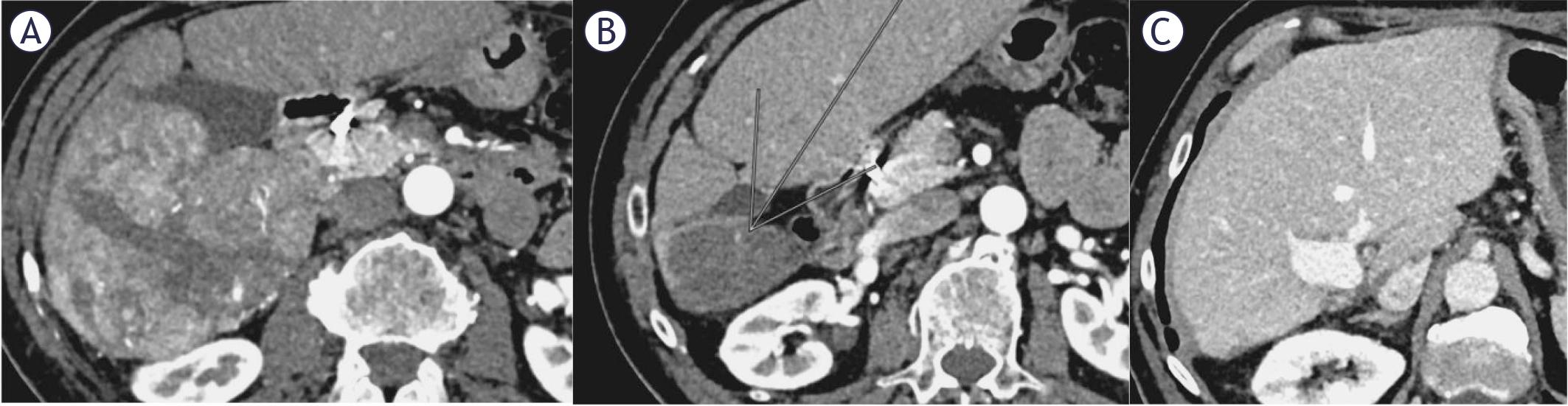
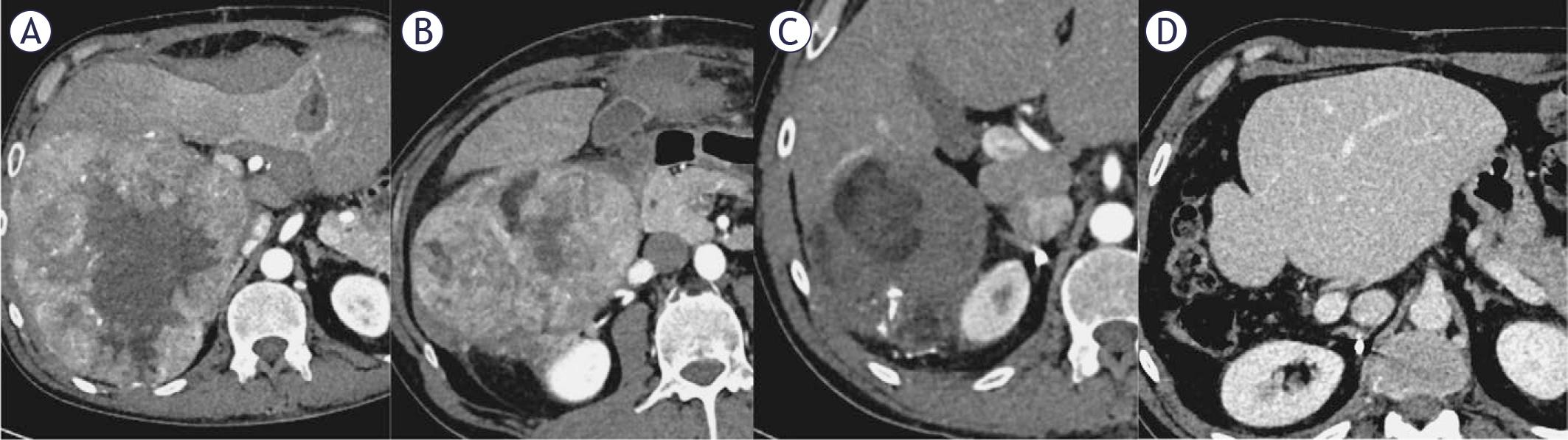
FIGURE 3.
Comparison of literature reported transarterial radioembolization (TARE) + immunotherapy treatment
| Study | Study design | No. of patients | HCC stage | Locoregional therapy | Immunotherapy regimen | CRR | Median OS |
|---|---|---|---|---|---|---|---|
| Villalobos et al.2 | Retrospective | 19 | Advanced unresectable HCC (84% BCLC C) | TARE | Atezo/Bev (n = 10); nivolumab (n = 9) | 16% | 12.9 months |
| Yu et al.21 | Retrospective | 10 | 60% BCLC C, 40% BCLC B | TARE | Atezo/Bev | 75% | 90% (6 months), 77.1% (12 months) |
| Yeo et al.23 | Retrospective | 142 | BCLC C | TARE | Mixed ICIs | Not reported | 19.8 months |
| Presen study | Case series | 3 | BCLC C | TARE (± DEBTACE) | Atezo/Bev | 100% | Ongoing; no recurrence at 13–53 months |
Summary of key results
| Variable | Patient 1 | Patient 2 | Patient 3 |
|---|---|---|---|
| Age / Sex | 58 / Female | 69 / Male | 50 / Male |
| Liver cirrhosis | Non-cirrhotic liver | Child–Pugh A (6 points) | Non-cirrhotic liver |
| Performance status | 0 | 1 | 0 |
| Baseline AFP (kU/L) | 7.9 | 524 | 2.5 |
| Tumor size (mm) | 116 + 15 mm satellite lesion | 100 | 170 |
| Vascular invasion | Hepatic vein + IVC (Vv3) | Main portal vein (Vp4) | Portal vein (Vp3) + hepatic vein + IVC (Vv3) |
| Extrahepatic disease | Lung metastasis (7 mm) | None | None |
| TARE type | 90Y resin microspheres | 90Y resin microspheres | 90Y glass microspheres |
| Additional locoregional therapy | None | None | DEB-TACE |
| Absorbed tumor dose (Gy) | 55 | 45 | 100 |
| Time to immunotherapy | 4 months post-TARE | 4 months post-TARE | 1 month post-TARE |
| Immunotherapy cycles | 13 | 13 | 7 preoperatively + 12 adjuvant |
| Best radiological response (mRECIST) | CR after 8 months | CR after 14 months | CR after 4 months |
| Serious adverse events following TARE + atezo/beva | No | No | No |
| Surgical procedure | Right hemihepatectomy + partial IVC resection | Right hemihepatectomy + portal vein resection | Right hemihepatectomy + marginal IVC excision |
| Pathological response | Complete necrosis, no viable tumor | Complete necrosis, no viable tumor | Complete necrosis, no viable tumor |
| Postoperative complications | Pneumonia | None | None |
| Length of stay (POD) | 12 | 6 | 7 |
| Follow-up outcome | No recurrence; died of cardiac arrest | No recurrence at 53 months | No recurrence at 13 months |
| Overall survival | 20 months | Ongoing (> 53 months) | Ongoing (> 13 months) |