The prevalence of environmental contaminants, exacerbated by anthropogenic activities and industrialization, poses a substantial threat to both human and animal health (Shetty et al., 2023). Aflatoxin B1 (AFB1), a potent mycotoxin commonly found in food, presents a significant health hazard due to its mutagenic, carcinogenic, hepatotoxic, immunotoxic, and teratogenic properties (Hamed et al., 2023; Abdelnour et al., 2024; Jaćević et al., 2023). AFB1 contamination can result in growth retardation, metabolic disruptions, and immune suppression (Cui et al., 2023; Imbabi et al., 2024), leading to significant economic losses, particularly within the livestock industry. The liver, a major metabolic center, is a primary target organ for AFB1-induced toxicity (Zhang et al., 2019; Al-Azhary et al., 2023). Although significant attention has been directed towards understanding the liver's impact (Rashad et al., 2024), the consequences of AFB1 exposure on male reproductive function, particularly concerning testicular function and sperm quality, warrant further investigation due to the limited research in this area.
Laboratory animals, particularly rabbits, are essential in biomedical research, contributing significantly to our understanding of human diseases and the development of pharmaceuticals (Fan and Watanabe, 2003). Rabbits exhibit a heightened sensitivity to aflatoxin B1 (AFB1) toxicity, resulting in impaired growth, reduced feed conversion efficiency, and disruptions in blood hemostasis (Soga and Oluremi, 2022), ultimately compromising productivity. Studies have shown that the oral LD50 for AFB1 varies among different animal species (Popescu et al., 2022; Pożarska et al., 2024). In rabbits, it ranges from 0.3 to 0.5 mg/kg body weight, while in ducklings, it is 0.3 to 0.6 mg/kg. For pigs, the range is 0.3 to 0.6 mg/kg, for calves it is 0.5 to 1.0 mg/kg, and for sheep, it is 2 to 5 mg/kg (Popescu et al., 2022; Pożarska et al., 2024). AFB1 suppressed testosterone secretion by inhibiting the expression of key proteins crucial for steroid synthesis (Adedara et al., 2014). Moreover, AFB1 induces hepatocyte pyroptosis via pro-inflammatory signaling, leading to liver inflammatory injury through its regulation of the NLRP3/COX2 pathway (Zhang et al., 2019). Exposure to AFB1 can impair male gametogenesis by disrupting the structure and functionality of reproductive organs, ultimately reducing fertility (Ye et al., 2024).
The rigorous evaluation of feed additives is crucial for ensuring animal health and food safety, with regulatory bodies requiring comprehensive scientific data before market approval (Hamed et al., 2023). These additives, including antitoxins, coccidiostats, and natural growth promoters, are commonly added to animal feed to enhance quality, nutrition, health, and performance (Saleem et al., 2017; Sun et al., 2018; Tsiouris et al., 2021; Rashad et al., 2024). AFB1-induced oxidative stress is a key mechanism through which this mycotoxin negatively impacts male reproductive function (Fallahi et al., 2021), including testicular integrity and sperm viability, leading to reduced reproductive efficiency (Ye et al., 2024). Consequently, there is growing interest in exploring natural compounds as potential countermeasures against AFB1 toxicity. Several classes of natural substances, including microalgae (Abdelnour et al., 2024), plant extracts (Saleem et al., 2017; Rashad et al., 2024), and essential oils (Miri et al., 2023) have demonstrated promise in mitigating the adverse effects of AFB1 in animal feed.
Phoenix dactylifera L. (date palm) is a prominent member of the palm family (Arecaceae), particularly in Arab nations and the Middle East (Otify et al., 2021). This tree is revered as “the tree of life” due to its fruits. Despite extensive research on the uses and potential benefits of date fruit (Otify et al., 2021), limited attention has been given to the importance of other parts of the tree, such as pollen and seeds. Date palm pollen (DPP) from Phoenix dactylifera has a long history of traditional use in folk medicine, particularly for addressing male infertility and as an aphrodisiac (Farag et al., 2023). This traditional application may be attributed to DPP's rich content of estrogenic compounds, including estrone, which can act as gonad-stimulating agents (Shehzad et al., 2021). Furthermore, DPP is a source of various bioactive phytochemicals, such as saponins, polyphenols, steroids, and terpenoids (Farag et al., 2023; Otify et al., 2021). These diverse constituents have been associated with a range of beneficial properties, including antimicrobial, cytotoxic, aphrodisiac, antidiabetic, antioxidant, and anti-inflammatory activities (Maqsood et al., 2020). While the potential of DPP to support male reproductive health has been recognized, its efficacy in mitigating the detrimental effects of AFB1 remains largely unexplored. There is a limited understanding of the specific mechanisms through which AFB1-contaminated diets impact male rabbit reproductive physiology. Recognizing DPP's established antioxidant and anti-inflammatory properties, we hypothesized it could mitigate AFB1-induced toxicity in rabbits through dietary supplementation. This study thus aimed to investigate the impact of AFB1 exposure on key immune-physiological parameters, metabolic responses, oxidative stress markers, adipokine profiles, DNA damage, pyroptosis-related gene expression, semen quality, and testicular histopathology in male rabbits, while also assessing DPP's protective role against AFB1-induced reproductive dysfunction.
Aflatoxin B1 (A6636, purity >98.0%, Figure 1) was purchased from Sigma-Aldrich (Darmstadt, Germany) and dissolved in methanol. Phoenix dactylifera pollen grains (DPP) were collected from date palm trees during August and September, washed, dried, and stored at 4°C. The collected pollen was washed, dried, and preserved in a refrigerator at 4°C until added to the diet according to the study protocol. This study was reviewed and approved by the ZU-IACUC committee (ZU-IACUC/2/F/25/2023) in accordance with the U.K. Animals (Scientific Procedures) Act, 1986, and associated guidelines (Hollands, 1986), EU Directive 2010/63/EU for animal experiments, the National Research Council's Guide for the Care and Use of Laboratory Animals (NIH Publications No. 8023, revised 1978), and in compliance with the ARRIVE guidelines.
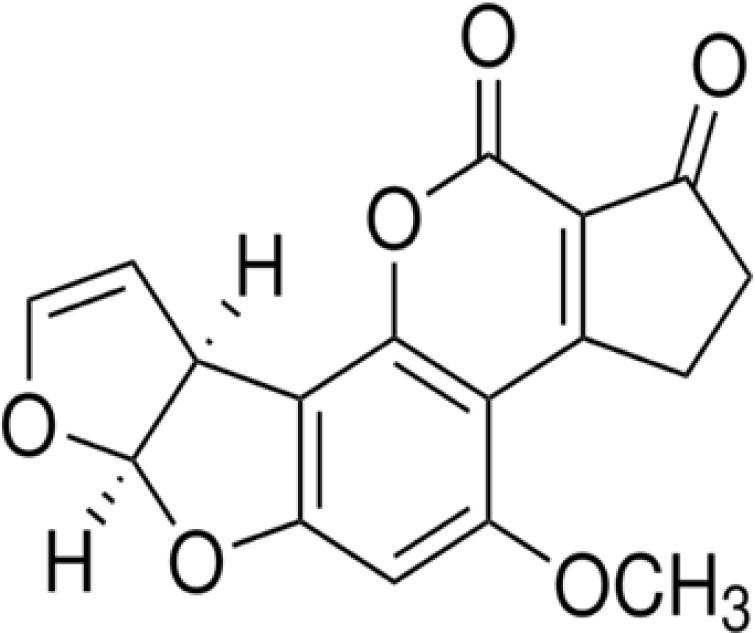
Aflatoxin B1 chemical structure
Thirty-two healthy 4-month-old male rabbits (3.02±0.11 kg) were randomly divided into four groups: control, AFB1, DPP, and AFB1+DPP. The rabbits were individually housed in galvanized wire cages (40 × 50 × 35 cm) with natural ventilation. The control group received a basal diet, while the other groups were fed as follows: AFB1 group (0.3 mg AFB1/kg diet), DPP group (basal diet + 300 mg DPP/kg), and AFB1+DPP group (basal diet + 0.3 mg AFB1/kg + 300 mg DPP/kg). The diet composition and nutritional values are presented in Table S1. During the diet preparation the AFB1 was incorporated and added into the diets by mixing with wheat bran. The AFB1 dosage was determined according to previous studies (Zhang et al., 2024 b), while the AFB1 therapy of DPP was based on previous studies (Attia et al., 2011; Abdelreheim et al., 2024). The rabbits had ad libitum access to feed and water throughout the two-month experimental period. All animals were housed under identical hygiene and management conditions.
Semen samples were collected from the bucks using an artificial vagina and a teaser doe. Samples were collected from each group once a week (8 samples per group) over the last 4 weeks (weeks 5 to 8) to evaluate sperm quality parameters. The criteria outlined in the International Rabbit Reproduction Group guidelines (Boiti et al., 2005) were followed to assess sperm concentration, viability, abnormality, and progressive motility. After diluting the semen (1:100) in formaldehyde phosphate-buffered saline solution, the concentrations of sperm cells were evaluated using a modified Neubauer chamber slide (Brandstwiete 4, Germany) and a light microscope at 40× magnification (Olympus Optical Co. Ltd., Tokyo, Japan). Forward motility was estimated using a light microscope with a hot stage at 100× magnification by visually examining numerous microscopic fields for each semen ejaculate sample. The expected results ranged from 0% to 100%. Sperm viability and abnormality were assessed by calculating 300 sperm cells stained with an eosin-nigrosine blue staining mixture. Unstained cells were considered viable, while cells with partial or complete staining were classified as dead spermatozoa.
The hypo-osmotic swelling test (HOST) was used to assess the membrane function ratio. A mixture of 50 μL of sample with 500 μL of HOST solution was incubated for 30 minutes at 37°C, then placed on a glass slide, covered with a coverslip, and observed at 400× magnification under a phase-contrast microscope (Leica DM 500, Switzerland). The swelling capability of 200 sperm cells was evaluated in HOST. To obtain seminal plasma, the whole semen samples were centrifuged for 20 minutes at 700 ×g and stored at –20°C for further studies. The levels of glutathione (GSH) (Beutler et al., 1963), total antioxidant capacity (TAC) (Young, 2001), malondialdehyde (MDA) (Draper and Hadley, 1990), and nitric oxide (Ghasemi et al., 2010) in seminal plasma were determined using commercial kits provided by Bio-Diagnostic Co. The assays were conducted using an ultraviolet–visible spectrophotometer (Bio-Rad, USA).
At the end of the experimental period, sterilized syringes were used to collect the blood samples (n=6 in each group) from the ear vein. After collecting, samples were held at room temperature for two hours before centrifugation to isolate the serum, which was then stored in Eppendorf tubes at –20°C for subsequent biochemical analysis. We determined various blood biochemistry parameters using commercial kits (BioSystem S.A., Barcelona, Spain) and colorimetric methods. These parameters included: total protein (TP; g/dL), albumin (ALB; g/dL), triglycerides (TG; mg/dL), creatinine, urea, total cholesterol (TC; mg/dL), glucose concentration (mg/dL), high-density lipoprotein (HDL), total bilirubin, and low-density lipoprotein (LDL). Globulin levels (GLB g/dL) were derived by subtracting albumin from total protein. The liver enzymes activities, including lactate dehydrogenase (LDH; U/L), aspartate transferase (AST; U/L) and gamma-glutamyl transferase (GGT; U/L) were determined by commercial kits (BioSystems, Barcelona, Spain) based on the manufacturer's instructions.
The redox status indicators in blood plasma, such as the activity of superoxide dismutase (SOD; IU), total antioxidant capacity (TAC; mM/L), glutathione (GSH; mmol/ml), malondialdehyde (MDA; nmol/mL), were analyzed using commercial kits purchased from Biodiagnostic (Giza, Egypt) through spectrophotometric procedures. Immunoglobulins (IgG and IgM) were estimated following the protocol directions using an ELISA kit acquired from Elabscience company (Houston, Texas, 77079, USA) as reported by Saghir et al. (2023). A competitive sandwich ELISA kit (Trevigen, Gaithersburg, MD, USA) following the manufacturer's instructions as reported by Koçak et al. (2023) and Saghir et al. (2023), was used to measure the concentration of 8-hydroxy-2′-deoxyguanosine (8-OHdG) in ng/mL, which is considered an indicator of DNA damage. The serum levels of interferon-gamma (IFN-γ) (El-Ratel et al., 2025), interleukin-10 (IL-10) (Xia et al., 2023), and interleukin-6 (IL-6) (Hassan et al., 2024), were measured using commercial ELISA kits with catalog numbers MBS2021850, MBS2019804, and MBS220076 provided by MyBioSource Company (San Diego, CA, USA), respectively. Lysozyme activity was assessed using the turbidimetric technique (Mörsky, 1983).
For the assessment of testosterone and adipokines, three samples were utilized from each group. Serum testosterone, leptin, and visfatin levels were measured using commercially available ELISA kits (MyBioSource, USA). Testosterone levels were quantified using a competitive immunoassay with a detection range of 0.625–10 ng/ml and a sensitivity of<0.625 ng/ml. Leptin and visfatin levels were measured using a quantitative sandwich ELISA (Mlyczyńska et al., 2023).
From each group, three bucks were humanely sacrificed, and their testicular tissues were then carefully extracted following a standardized procedure (Bilinska et al., 2018). For histological examination, three samples were collected and fixed in Bouin's solution. Additional testicular samples were promptly preserved in liquid nitrogen to facilitate subsequent gene expression analysis. The Bouin-fixed samples were embedded in paraffin and cut into 5 μm-thick sections using a sledge microtome (Jung AG, Heidelberg, Germany). The sections were then stained with hematoxylin-eosin (HE) and evaluated with a light microscope (Axioskop, Carl Zeiss AG, Oberkochen, Germany). After staining, the alterations in testicular tissue sections were detected in specific fields under blindfolded conditions using standard light microscopy (Olympus BX 51, Japan) to explore the pathological changes. The histopathological lesion grading was determined by interpreting histomorphological changes in five fields per section according to the protocol of Gibson-Corley et al. (2013).
The testicular samples were frozen in liquid nitrogen, and RNA was isolated from the tissues using Trizol reagent (Invitrogen, USA) following the manufacturer's instructions. The RNA quality and quantity were assessed using NanoDrop 1000 at a 260/280 wavelength. The cDNA was synthesized from RNA using the RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific, USA). Specific primer pairs for the mRNA of pyroptosis-related genes (Caspase-1, NLRP3, GSDMD, and IL-18) were designed using Primer-BLAST software and are listed in Table S2. The reverse transcription was performed using the PrimeScript RT reagent kit (Takara, China). qRT-PCR analyses were conducted using the QuantStudio 6 Flex system (Applied Biosystems, USA) with SYBR Premix Ex Taq™ II reagents (No. RR820A, Takara, China). The expression of the selected genes was calculated using the 2ΔΔCt method as described by Livak and Schmittgen (2001).
The data were first checked for normality and homogeneity of variances using the Shapiro-Wilk and Levene tests. Data was analyzed using one-way ANOVA in SPSS Statistics Software (version 25, IBM Corp., Armonk, NY, USA). Differences between group means were assessed using Duncan's post-hoc test. Statistical significance was set at P≤0.05. The following statistical model was used with the data:
Yij is the indicated value of the concerned treatment, μ is the indicated mean for the concerned treatment, Ti is the treatment effect, and eij is the error related to individual observation.
Figure 2 highlights the detrimental impact of AFB1 on growth performance in rabbit bucks. AFB1-contaminated diets significantly impaired weight gain compared to the control and DPP groups (P<0.001). However, DPP supplementation effectively counteracted the negative effects of AFB1, restoring body weight to levels comparable to the control and DPP groups.
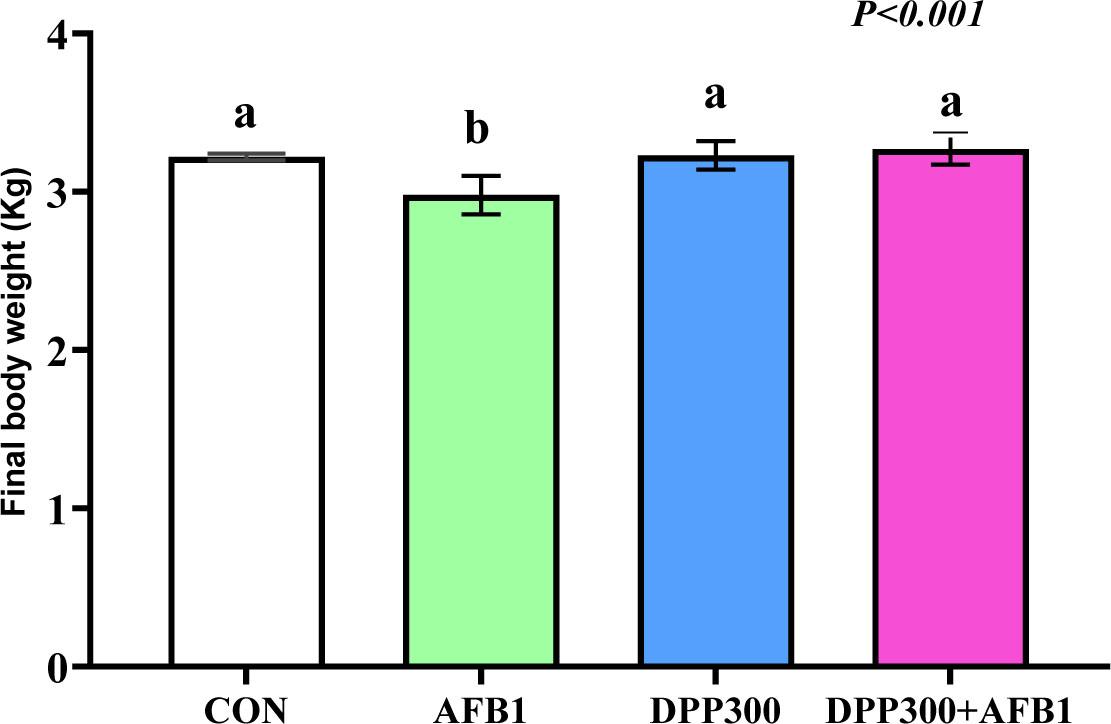
The final body weights of rabbit bucks were measured in response to dietary inclusion of AFB and treatment with date palm pollen (DPP). The rabbit bucks were fed a basal diet (CON), contaminated diets with AFB1 (AFB1), date palm pollen (DPP), or AFB1+DPP for two months. a, b – values with different letters indicate a significant difference (P<0.05)
Table 1 shows that AFB1 consumption significantly increased levels of TC, TG, and LDL compared to the control and DPP groups (P<0.05). However, DPP supplementation significantly reduced these levels by 61.2%, 26.9%, and 24.16%, respectively (P<0.05). Bucks fed AFB1-contaminated diets exhibited higher levels of creatinine, urea, and total bilirubin compared to other groups, while DPP therapy significantly decreased these levels by 48.7%, 25.4%, and 5.7%, respectively, compared to the AFB1 group. AFB1 significantly reduced blood protein fractions (total protein, albumin, and globulin) compared to other groups, while the highest levels were observed in the DPP300+AFB group (P<0.01). Additionally, AFB1 significantly increased liver enzymes (AST, GGT, and LDH) compared to other groups (P<0.01). The inclusion of DPP in AFB1-contaminated diets significantly mitigated the elevation of these enzymes induced by AFB1 (P<0.01). Interestingly, both DPP and DPP300+AFB1 groups exhibited significantly improved HDL levels (P<0.05).
Impacts of dietary date palm pollen (DPP) in mitigating the negative effect of aflatoxin B1 (AFB1) on the blood metabolites of male rabbits
| Item1 | Treatments2 | P value | |||
|---|---|---|---|---|---|
| CON | AFB1 | DPP300 | DPP300+AFB1 | ||
| TP, mg/dL | 5.89±0.10 b | 4.33±0.11 c | 6.77±0.10 b | 8.15±0.15 a | <0.001 |
| ALB, mg/dL | 4.63±0.14 b | 3.13±0.05 c | 4.53±0.17 b | 5.82±0.33 a | <0.001 |
| GLO, mg/dL | 1.26±0.23 b | 1.20±0.16 b | 2.24±0.10 a | 2.34±0.36 a | 0.013 |
| CREA, mg/dL | 1.42±0.01 b | 2.34±0.01 a | 1.20±0.02 b | 1.20±0.01 b | <0.001 |
| Urea, mg/dL | 52.25±1.04 b | 70.99±0.80 a | 36.43±1.84 c | 52.92±1.16 b | 0.086 |
| TB, mg/dL | 0.61±0.01 c | 0.82±0.02 a | 0.76±0.01 b | 0.77±0.02 b | <0.001 |
| TC, mg/dL | 75.53±0.64 b | 122.75±1.01 a | 75.32±0.67 b | 74.54±0.93 b | <0.001 |
| TG, mg/dL | 81.71±0.28 b | 111.19±3.09 a | 81.40±0.70 b | 81.18±0.93 b | <0.001 |
| LDL, mg/dL | 31.94±0.47 b | 41.84±0.60 a | 32.49±0.31 b | 31.76±0.87 b | <0.001 |
| HDL, mg/dL | 33.57±2.03 b | 25.04±0.91 c | 42.82±1.19 a | 41.89±0.93 a | <0.001 |
| LDH, U/L | 31.89±0.86 b | 66.89±2.21 a | 33.53±0.36 b | 31.45±1.67 b | <0.001 |
| GGT, U/L | 22.00±0.58 b | 26.00±1.15 a | 19.00±0.51 c | 17.00±0.58 c | <0.001 |
| AST, U/L | 19.67±0.87 b | 34.67±0.84 a | 22.00±0.52 b | 20.00±0.58 b | <0.001 |
Total protein (TP), albumin (ALB), globulin (GLO), creatinine (CREA), total bilirubin (TB), total cholesterol (TC), triglycerides (TG), low density lipoprotein (LDL), high density lipoprotein (HDL), lactate dehydrogenase (LDH), gamma-glutamyl transferase (GGT) and aspartate transferase (AST).
– values with different letters within the same row mean significant difference (P<0.05). Data presented as mean ± SEM (standard error of means). The rabbit bucks were fed a basal diet (CON), contaminated diets with AFB1 (AFB1), date palm pollen (DPP), or AFB1+DPP for two months.
Rabbits fed AFB1-contaminated diets exhibited lower sperm concentration compared to the control and DPP groups (Table 2). However, the addition of DPP to the AFB1 diet increased sperm concentration. The DPP and control groups demonstrated higher sperm concentration than the AFB1 group (P<0.05). Rabbits fed diets supplemented with DPP alone exhibited superior sperm viability, motility, and membrane function compared to other groups (P<0.05). Similarly, adding DPP to AFB1-contaminated diets improved sperm motility, viability, and membrane function compared to the AFB1 group (P<0.05). The AFB1 group exhibited significantly higher sperm abnormality percentages compared to other groups, while DPP supplementation to the AFB1 diet mitigated this increase (P<0.05). DPP supplementation significantly increased GSH levels compared to other groups (P<0.05), while TAC and nitric oxide levels were lowest in the AFB1 group (P<0.05). Enriching AFB1 diets with DPP significantly improved TAC and nitric oxide levels in seminal plasma. MDA levels were lowest in the DPP group, and DPP-enriched AFB1 diets reduced MDA levels compared to the AFB1 group (P<0.05). Overall, DPP supplementation effectively alleviated the decline in semen quality caused by AFB1, significantly improving sperm count, motility, and reducing sperm abnormalities by supporting antioxidant defense and reducing lipid peroxidation in seminal plasma.
Impacts of dietary date palm pollen (DPP) in mitigating the negative effect of aflatoxin B1 (AFB1) on semen quality and seminal plasma antioxidants of male rabbits
| Item1 | Treatments2 | P value | |||
|---|---|---|---|---|---|
| CON | AFB1 | DPP300 | DPP300+AFB1 | ||
| Sperm concentration, 106/mL | 335.00±3.87 b | 285.00±9.75 c | 329.60±2.94 b | 363.00±6.24 a | <0.001 |
| Sperm motility, % | 50.20±1.28 b | 40.60±1.69 c | 62.40±1.03 a | 49.40±1.91 b | <0.001 |
| Viability, % | 52.60±1.17 b | 31.80±0.92 c | 69.60±1.03 a | 51.20±1.91 b | <0.001 |
| Membrane function, % | 49.40±0.87 b | 37.40±2.18 c | 63.60±1.36 a | 51.00±1.05 b | <0.001 |
| Sperm abnormality, % | 19.20±1.02 b | 22.40±0.81 a | 17.60±1.03 b | 18.60±0.88 b | 0.013 |
| Seminal plasma | |||||
| GSH, mmol/mL | 38.88±0.59 b | 20.29±8.23 c | 44.95±0.66 a | 36.08±0.63 b | 0.005 |
| TAC, nmol/L | 0.26±0.01 a | 0.19±0.01 b | 0.24±0.01 a | 0.25±0.01 a | <0.001 |
| MDA, nmol/mL | 47.07±1.17 c | 64.76±1.07 a | 34.78±0.90 d | 56.26±2.82 b | <0.001 |
| nitric oxide, nmol/mL | 43.17±1.10 a | 33.39±0.73 b | 43.37±0.59 a | 45.93±0.67 a | <0.001 |
Total antioxidant capacity (TAC), glutathione (GSH), and malondialdehyde (MDA).
– values with different letters within the same row mean significant difference (P<0.05). Data presented as mean ± SEM (standard error of means). The rabbit bucks were fed a basal diet (CON), contaminated diets with AFB1 (AFB1), date palm pollen (DPP), or AFB1+DPP for two months.
Figure 3 (A–D) illustrates the impact of dietary DPP on mitigating the negative effects of AFB1 on the serum redox homeostasis of male rabbits. Rabbits fed AFB1-contaminated diets exhibited significantly lower levels of TAC (Figure 3 A), SOD (Figure 3 B), and GSH (Figure 3 C) in their blood compared to other groups (P<0.05). All tested groups, except for the AFB1 group, showed similar levels of SOD, GSH, and MDA (P>0.05). MDA levels (Figure 3 D) were significantly higher in rabbits fed contaminated diets, but DPP treatment significantly reduced this elevation (P<0.05).
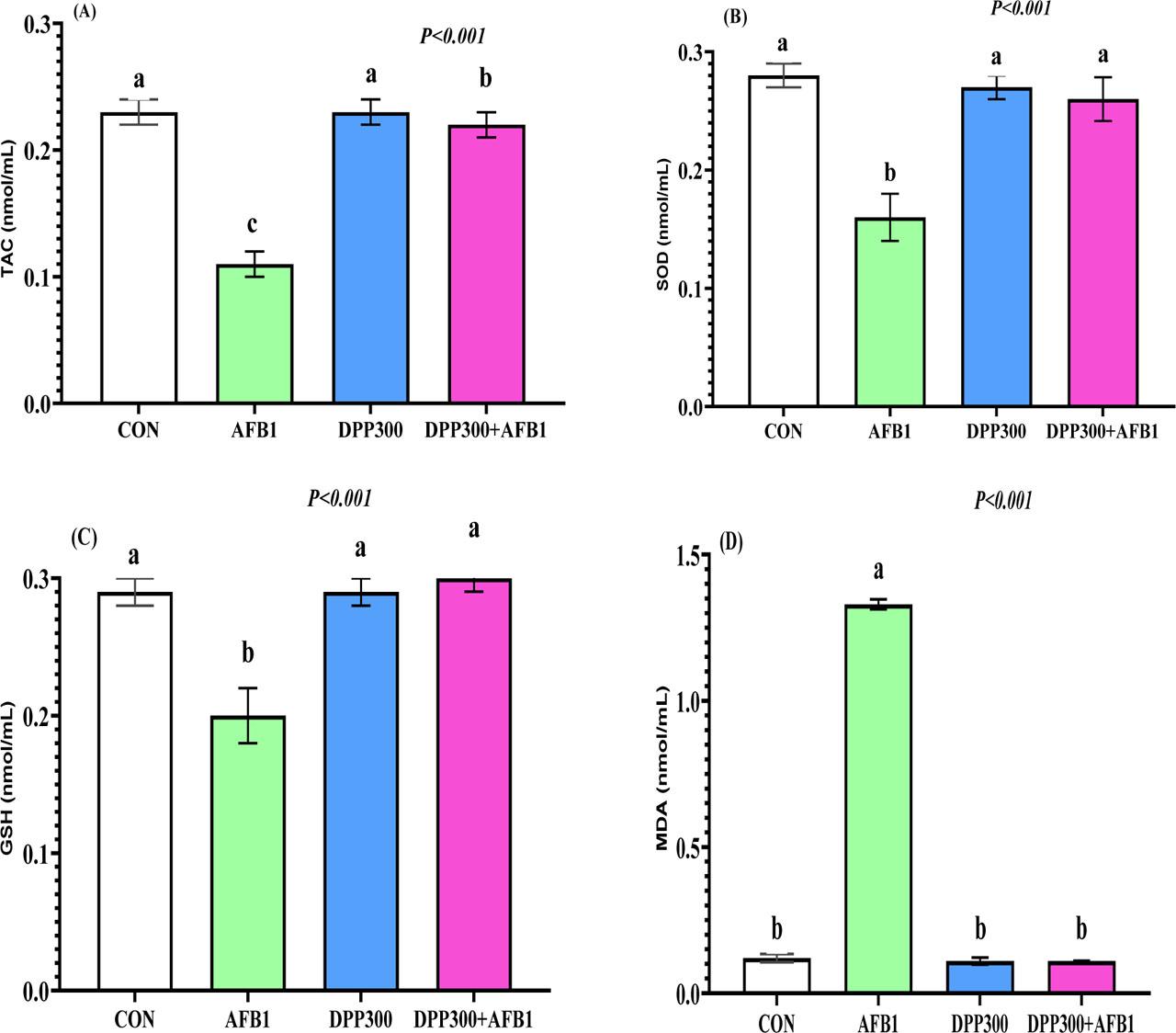
(A–D). Impacts of dietary date palm pollen (DPP) in mitigating the negative effect of aflatoxin B1 (AFB1) on serum redox homeostasis of male rabbits. a, b, c – values with different letters indicate a significant difference (P<0.05). Data presented as mean± SEM (standard error of means). The rabbit bucks were fed a basal diet (CON), contaminated diets with AFB1 (0.3 mg/kg), date palm pollen (DPP, 300 mg/kg diet), or AFB1+DPP for two months
The impacts of dietary DPP in mitigating the negative effects of AFB1 on DNA oxidative damage, immunoglobulins (IgM and IgG), and pro-inflammatory cytokines in male rabbits are illustrated in Figure 4. Dietary AFB1 significantly increased the DNA damage marker (8-OHdG) compared to the other groups (P<0.001; Figure 4 A). AFB1 enriched with DPP can reduce the DNA damage marker similar to the untreated (control) and DPP groups (P>0.05). IgM (Figure 4 C) and IgG (Figure 4 B) levels were significantly reduced in the AFB1 group compared to the other groups (P<0.001). The highest levels of IgG and IgM were observed in the DPP300 group, with no significant difference when compared with the AFB1-treated DPP therapy group (P>0.001). AFB1 significantly increased the circulating pro-inflammatory markers such as IL-6 (Figure 4 D) and IFN-γ (Figure 4 E), while enriching AFB1 diets with DPP significantly reduced this elevation (P<0.001). All treatments had lower IL-6 and IFN-γ levels compared to the AFB1 group (P<0.05). IL-10, as an anti-inflammatory agent, was improved in the AFB1+DPP therapy group compared to the AFB1 group (P<0.001). AFB1 significantly reduced nitric oxide (Figure 4 G) and lysozyme activity (Figure 4 H) compared to the other groups. DPP addition alone or AFB1+DPP improved lysozyme activity and nitric oxide levels (P<0.001).
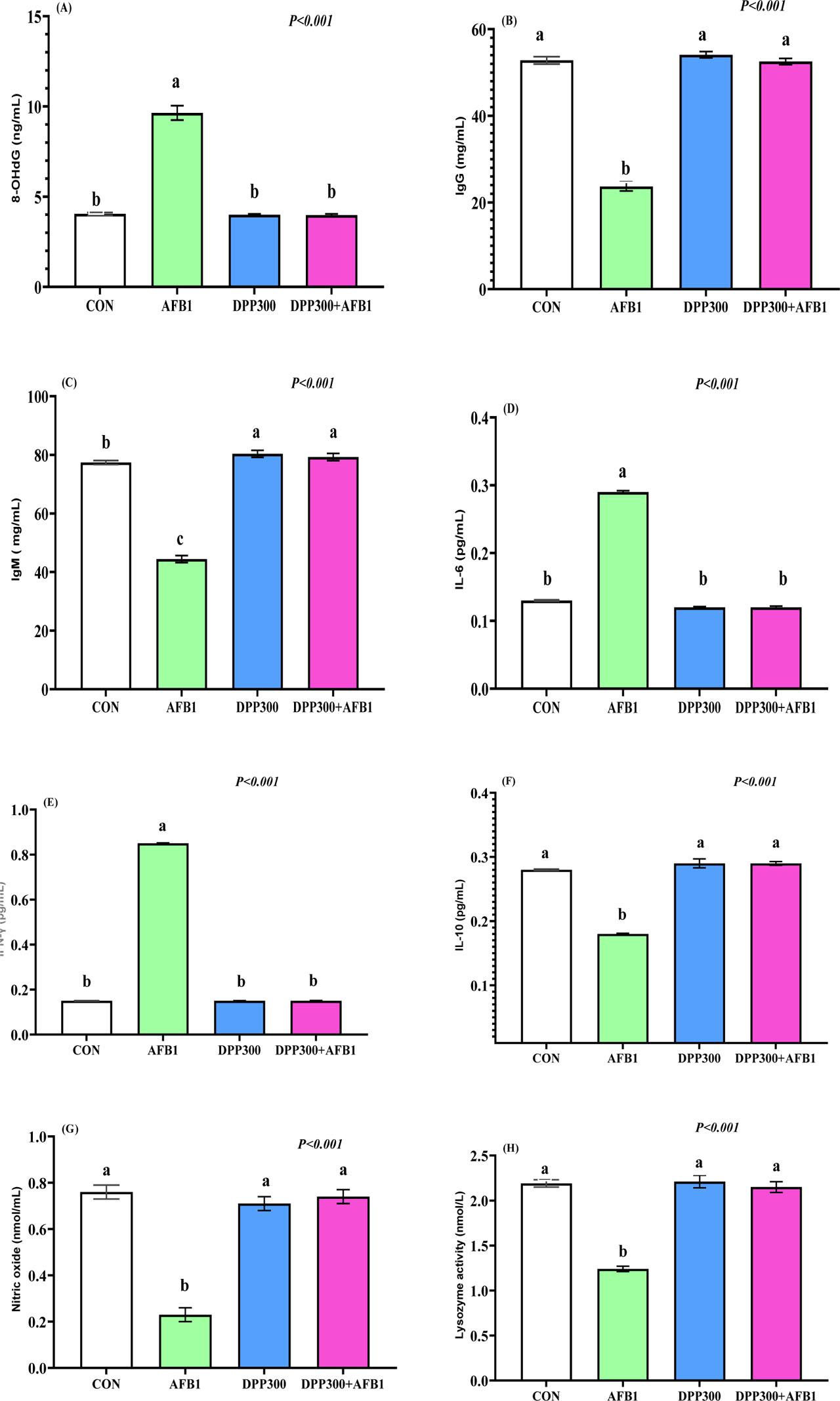
(A–H). Impacts of dietary date palm pollen (DPP) in mitigating the negative effect of aflatoxin B1 (AFB1) on DNA of oxidative DNA damage (Figure 4 A), immunoglobins G (IgG, Figure 4 B) and immunoglobins M (IgM, Figure 4 C), and pro-inflammatory cytokines such as IL-6 (Figure 4 D) and interferon-γ (IFN-γ; Figure 4 E), IL-10 (Figure 4 F), nitric oxide (Figure 4 G) and lysosome activity (Figure 4 H) of male rabbits. a, b, c – values with different letters indicate a significant difference (P<0.05). Data presented as mean ± SEM (standard error of means). The rabbit bucks were fed a basal diet (CON), contaminated diets with AFB1 (0.3 mg/kg), date palm pollen (DPP, 300 mg/kg diet), or AFB1+DPP for two months
AFB1 significantly reduced testosterone levels in rabbit bucks, while enriching AFB diets with DPP can ameliorate this reduction (Figure 5 A). In contrast, AFB increased the serum values of adipokines such as leptin (Figure 5 B) and visfatin (Figure 5 C) in rabbit bucks (P<0.05). DPP therapy can significantly decrease the levels of visfatin and leptin induced by AFB1 (P<0.001).
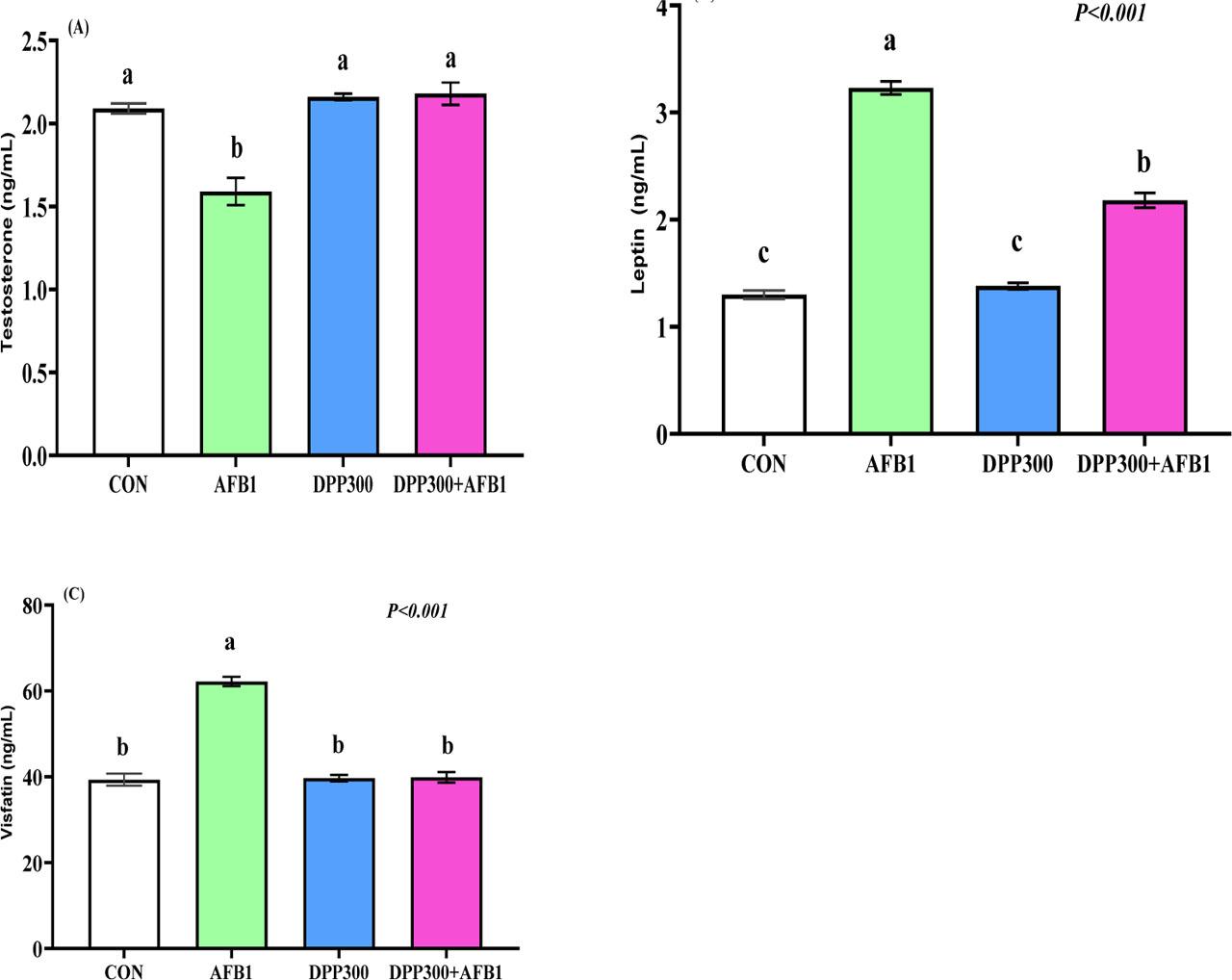
(A–C). Impacts of dietary date palm pollen (DPP) in mitigating the negative effect of aflatoxin B1 (AFB1) on serum testosterone (Figure 5 A) and adipokines such as leptin (Figure 5 B) and vistatin (Figure 5 C) of male rabbits. a, b, c – values with different letters indicate a significant difference (P<0.05). Data presented as mean± SEM (standard error of means). The rabbit bucks were fed a basal diet (CON), contaminated diets with AFB1 (0.3 mg/kg), date palm pollen (DPP, 300 mg/kg diet), or AFB1+DPP for two months
Bucks in the AFB1-fed group showed tissue atrophy and necrotic germinal epithelial lining. Additionally, interstitial edema was observed in animals from this group (Figure 6 B). Bucks in the control group (Figure 6 A) and DPP (Figure 6 C) had normal histology of seminiferous tubules and Leydig cells. Bucks in the therapy group (Figure 6 D) had moderate architectural changes in the majority of seminiferous tubules, less edema, and a reduced number of germ cells in some tubules. Moreover, the seminiferous tubules exhibited normal spermatocytes, spermatogonia, spermatids, and centrally impacted sperms.
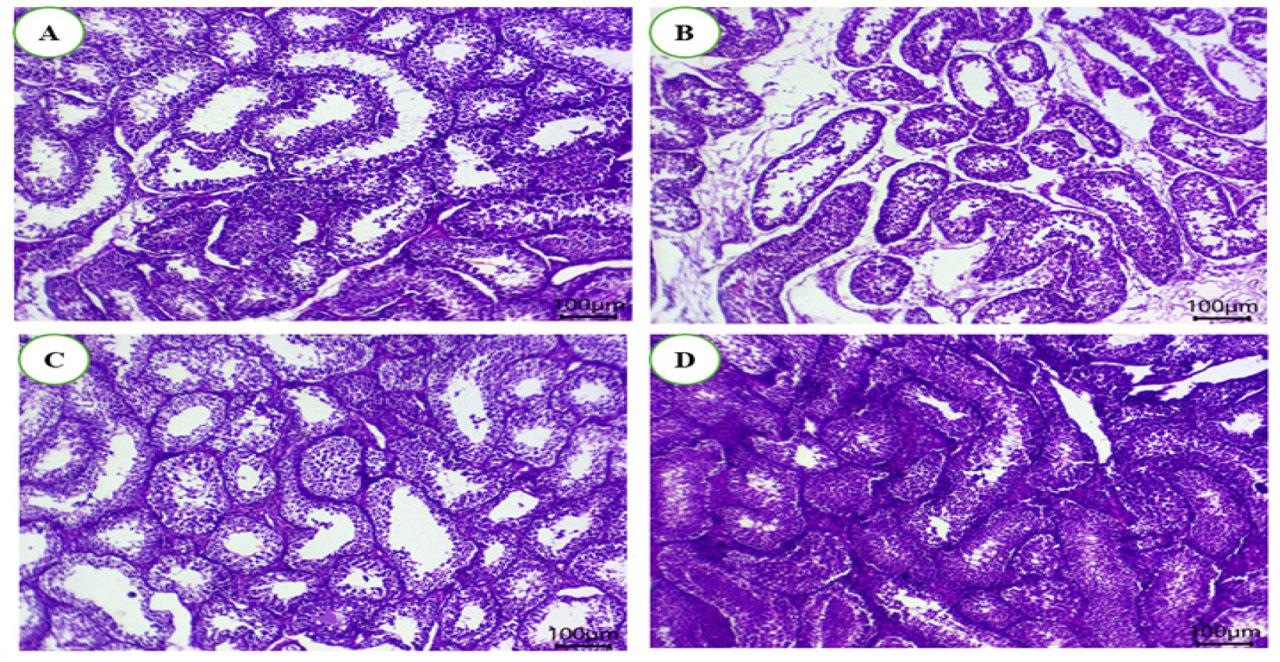
(A–D). The impact of dietary date palm pollen (DPP) on mitigating the negative effects of AFB1 on histopathological changes in testicular tissues of male rabbits. The rabbit bucks were fed a basal diet (CON), contaminated diets with AFB1 (0.3 mg/kg), date palm pollen (DPP, 300 mg/kg diet), or AFB1+DPP for two months. Bucks in the AFB1-fed group showed tissue atrophy and necrotic germinal epithelial lining. Additionally, interstitial edema was observed in animals from this group (Figure 6 B). Bucks in the control group (Figure 6 A) and DPP (Figure 6 B) had normal histology of seminiferous tubules and Leydig cells. Bucks in the therapy group (Figure 6 D) had moderate architectural changes in the majority of seminiferous tubules, less edema, and a reduced number of germ cells in some tubules
Pyroptosis-related genes (NLRP3, GSDMD, Caspase-1, and IL-18) expression in testicular tissues was significantly increased in bucks that received AFB in their diets (Figure 7 A–D). Bucks that received DPP along with AFB diets showed a significant downregulation of NLRP3 (Figure 7 A), GSDMD (Figure 7 B), Caspase-1 (Figure 7 C), and IL-18 (Figure 7 D) compared to the AFB1 groups (P<0.05). In general, the expression of all genes was downregulated in the control and DPP groups compared to the DPP300+AFB1 group (except for GSDMD) (P<0.05). Overall, DPP can mitigate AFB1 by decreasing pyroptosis-related factors in rabbit bucks.
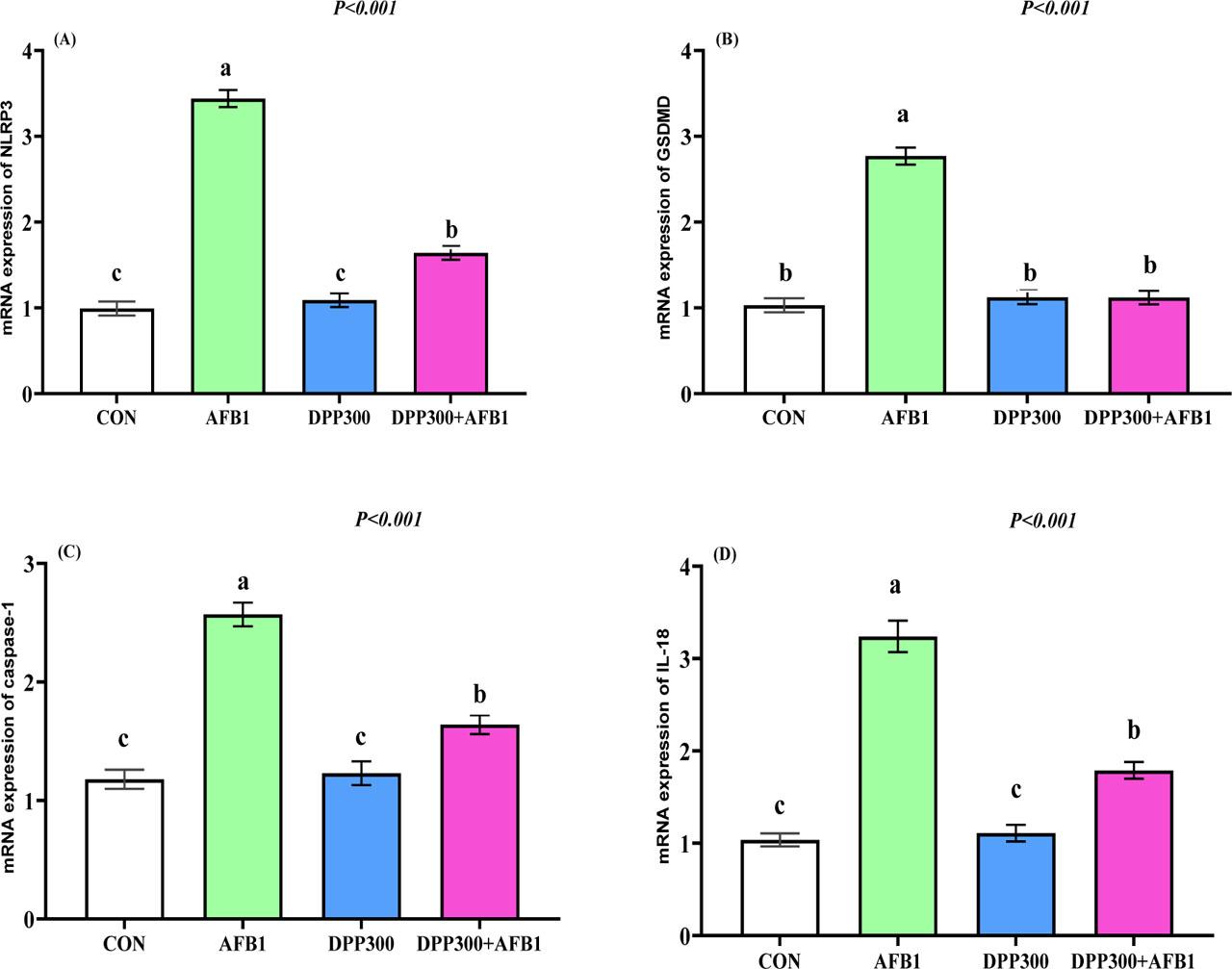
(A–D). Impacts of dietary date palm pollen (DPP) in mitigating the negative effect of aflatoxin B1 (AFB1) on pyroptosis-related genes such as NLRP3 (Figure 7 A), GSDMD (Figure 7 B), Caspase-1 (Figure 7 C), and IL-18 (Figure 7 D) in testicular tissues of male rabbits. a, b, c – values with different letters indicate a significant difference (P<0.05). Data presented as mean ± SEM (standard error of means). The rabbit bucks were fed a basal diet (CON), contaminated diets with AFB1 (0.3 mg/kg), date palm pollen (DPP, 300 mg/kg diet), or AFB1+DPP for two months
Aflatoxins, particularly AFB1, pose significant health risks, including reproductive dysfunction. Date palm pollen represents a promising source of natural compounds (Table 3) for the treatment of infertility disorders in animals and humans. This study investigates the detrimental effects of AFB1 on post-metabolic responses, immune function, semen quality, and reproductive health in male rabbits. Previous research has highlighted the negative impact of AFB1 on fertility (Adedara et al., 2014) and the potential benefits of DPP therapy (Fallahi et al., 2021), and this study provides further evidence (Iftikhar et al., 2011; Laghouati et al., 2021). The results indicate that AFB1 significantly disrupted blood biochemistry, leading to increased liver enzymes, impaired kidney function, and decreased blood proteins. Additionally, AFB1 exposure resulted in reduced semen quality, impaired immune and antioxidant responses, altered testicular histology, and decreased testosterone levels. Furthermore, AFB1 increased levels of adipokines, DNA damage markers, and pyroptosis-related genes. However, DPP administration effectively mitigated these adverse effects. DPP treatment reversed the elevation of liver and kidney function markers, enhanced immune and antioxidant responses, reduced DNA damage, inflammation, and pyroptosis, ultimately leading to improved semen quality and testosterone levels. This study emphasizes the crucial role of DPP in mitigating the harmful effects of AFB1 exposure in male rabbits. DPP emerges as a promising therapeutic agent to manage aflatoxin-induced infertility in male rabbits. Our data showed that AFB1-contaminated diets significantly decreased the final body weight of rabbit bucks. This finding is supported by studies conducted by various many authors (Prabu et al., 2013; Tsiouris et al., 2021; Soga and Oluremi, 2022; Rashad et al., 2024) who demonstrated that AFB1 negatively affected body weight, feed consumption and conversion in rabbits orally exposed to concentrations of 50–500 μg/kg of feed daily for 2 months. The harmful impacts of AFB1 on animal health may include an inability to consume food, anorexia, lipid formation, inhibition of DNA and RNA synthesis, and consequently, the inhibition of protein formation (Hassan et al., 2019). Increased exposure to aflatoxin in the diet can lead to a decrease in animal growth performance (Hassan et al., 2019; Imbabi et al., 2024).
The main identified molecules found in date palm pollen of Egyptian palm tree
| Compound name | Molecular formula | 2D structure | Biological activity | Reference |
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 |
| Disaccharide | C12H22O11 |
| Antimicrobial action | Zhang et al. (2014) |
| Kaempferol-3-O-hexoside (2″-sulfate) | C21H19O14S |
| Antioxidant activity | Abdallah et al. (2023) |
| Quercetin-3,4′-di-O-hexoside | C27H29O17 |
| Improve fertility in female rats | Abdallah et al. (2023); Otify et al. (2021) |
| Isorhamnetin-3-O-(2-rhamnosyl) hexoside | C28H31O16 |
| Anti-obesity and anti-inflammatory activity | González-Arceo et al. (2022) |
| Quercetin | C15H9O7 |
| Antioxidant activity | Dai et al. (2024) |
| Isorhamnetin | C16H11O7 |
| Antibacterial and anti-obesity | González-Arceo et al. (2022) |
| Dihydroxy-palmitic acid | C16H31O4 |
| Antioxidant activity | Otify et al. (2021) |
| Dihydroxy-linoleic acid | C18H31O4− |
| Antioxidant activity | Otify et al. (2021) |
| Caffeoyl-palmitic acid derivative | C25H39O5 |
| Antioxidant activity | Joujou et al. (2024) |
| Estrone acetate | C20H23O3 |
| Antioxidant activity | Abdallah et al. (2023) |
| Octadecadienoic acid derivative | C46H69NO5 |
| Antibacterial and antifungal | Hawar et al. (2023) |
| Hydroxy-stearic acid | C18H35O3 |
| Antiproliferative | – |
The current study demonstrates that dietary supplementation with DPP can enhance growth performance and increase body weight in male rabbits compared to AFB1 group, aligning with previous findings (El-Hammady et al., 2017; Shahba and Mansou, 2022). Aflatoxicosis, primarily caused by AFB1, is a significant health concern in rabbits. Outbreaks of AFB1 poisoning have been associated with various clinical signs, including anorexia, lethargy, weight loss, impaired productivity, jaundice, and high mortality rates (Tsiouris et al., 2021). Antioxidants play a crucial role in combating oxidative stress and mitigating the harmful effects of free radicals (Mishra et al., 2019). Therefore, this study aimed to investigate the impact of DPP on blood biochemistry, semen quality, adipokine levels, immune-antioxidant markers, and pyroptosis-related genes in male rabbits fed AFB1-contaminated diets.
In the liver, AFB1 is primarily metabolized into a bioactive metabolite (8,9-epoxide) that can react with cellular constituents, causing oxidative damage and cellular injury (Khlangwiset and Wu, 2010). In general, it disrupts cellular function through various pathways, such as inhibiting protein formation, perturbing cell membrane permeability, and altering DNA (Speijers and Speijers, 2004). The disaccharides in DPP can provide hydroxyl groups to stabilize membranes and bind to AFB1, potentially preventing its cellular uptake (Otify et al., 2021).
In this study, AFB1 led to elevated values of TC, TG, and LDL, creatinine, urea, and TB, reduced the blood protein fractions (TP, ALB, and GLO) and stimulated elevation in liver enzymes like AST, GGT, and LDH. These results are consistent with previous studies by Hassan et al. (2019), and Imbabi et al. (2024), which reported that oral administration of AFB1 to rabbits resulted in decreased total protein, albumin, and globulin concentrations, lowered glucose concentrations, increased urea and creatinine levels. Additionally, studies by Abdelhady et al. (2017), Hamed et al. (2023), and Ismail et al. (2020), found that daily oral exposure of bucks to AFB1 increased the activity of serum liver enzymes such as AST and ALT, and histological changes were observed, including bile duct hyperplasia, diffuse fatty liver degeneration, and focal hepatocellular necrosis. The improvement in selected rabbits' blood parameters observed in this study is attributed to DPP's rich nutrient and antioxidant profile, which enhances feed digestion and absorption (Šarić et al., 2009; Taghian et al., 2017).
The present study data indicates that rabbits exposed to DPP have higher total protein and albumin concentrations. These findings are consistent with those reported by El-Hammady et al. (2017), who observed that bucks fed a diet containing DPP showed improved total blood protein and albumin levels due to the acceleration of the digestion process, leading to increased amino acids in circulation. These findings demonstrated that supplementation with DPP reduced lipid formation, triglyceride, LDL, and VLDL levels, while increasing HDL concentration due to the high flavonoid compounds present in DPP, which prevent the accumulation of lipid peroxidation products (Abdel-Shaheed et al., 2021; Saleh et al., 2021). The hypo-lipidemic effect of DPP may be attributed to the presence of polyunsaturated fatty acids and phytosterols, which interfere with cholesterol absorption by enterocytes (Al-Abbasi and Al-Wazeer, 2023). Furthermore, DPP supplementation reduced AST and ALT levels, indicating an improvement in liver health. The high flavonoid content in date palm pollen helps protect unsaturated fatty acids, strengthening cellular membranes and protecting them from oxidation, thereby maintaining liver integrity and function (Abdel-Shaheed et al., 2021). Results revealed that aflatoxin caused tissue damage and impaired the immune system due to oxidative stress, affecting systemic inflammation with an increase in the levels of tumor necrosis factor-α and anti-inflammatory cytokines (Riahi et al., 2021; Abdelnour et al., 2024). DPP supplementation in our study significantly decreased oxidative stress biomarkers, concurrently increasing antioxidant capacity. This is further supported by the observed reduction in MDA levels, an indirect indicator of free radical formation (Abdelnour et al., 2024). DPP can enhance the expression of SOD2, NRF2, GPX4, and CAT genes, leading to improved semen quality in infertile men (Fallahi et al., 2021). These results corroborate the robust antioxidant properties of DPP, as evidenced by extensive literature (Otify et al., 2021).
Pollen has been shown to be a crucial component such as disaccharide, kaempferol-3-O-hexoside and quercetin in boosting immunity and antioxidative status by improving intestinal absorption of nutrients and activating digestive enzymes in large animals, poultry, and rabbits (Attia et al., 2011; Al-Samarrai et al., 2017; Baagar et al., 2022; Al-Abbasi and Al-Wazeer, 2023). Similarly, bucks consuming a DPP diet in the present study showed an increased antioxidant capacity of seminal plasma. Propolis has been found to activate antioxidant enzymes such as catalase and superoxide dismutase against free radicals (Baagar et al., 2022). Additionally, supplementation of DPP extract to sperm extender during cryopreservation improved semen quality and viability in human males (Al-Dujaily et al., 2012), rabbit (Laghouati et al., 2021), buffalo (El-Sheshtawy et al., 2016), bulls and stallions (El-Sisy et al., 2018) .
Reproductive efficiency is an essential factor in animal production. Healthy mature testes with good quality semen production are necessary to maintain this efficiency. The process of spermatogenesis occurs in the seminiferous tubules, and Sertoli cells stimulate the production and maturation of spermatozoa (Sun et al., 2018). Although spermatogonial cells are protected by the blood-testis barrier, AFB1 can pass through this natural barrier and impair the process of spermatogenesis (Zhang et al., 2024 a). Our results revealed that diets contaminated with AFB1 consumed by bucks led to lower sperm concentration, increased abnormality percentages, and reduced testosterone levels. This is consistent with Ataman and Ndiokwere (2016), who reported that oral exposure to AFB1 resulted in decreased testes weight and serum testosterone concentration, as well as significantly decreased ejaculate volume, sperm concentration and motility, and semen fructose concentration. A study by Sun et al. (2018) mentioned that AFB1 impaired reproductive efficiency with increased sperm abnormality and decreased acrosome integrity. Consistent with previous research, the improved reproductive efficiency of treated bucks in our study mirrors the findings of Laghouati et al. (2021). Their work demonstrated that different concentrations (20, 40, and 80 mg/mL) of DPP aqueous extract boosted semen viability, spermatogenesis, and acrosomal reaction, likely due to its antioxidant properties.
AFB1 decreases testosterone production by affecting its biosynthetic pathway in Leydig cells (Adedara et al., 2014). The low level of testosterone secretion observed in this experiment may lead to changes in epididymis function and consequently, alterations in sperm morphology (Hajb et al., 2023). Moreover, the impaired synthesis of the testosterone hormone may result in a decrease in buck's testes weight due to testicular degeneration (Abdelnour et al., 2021; AlGaradi et al., 2023). The testosterone hormone plays an important role in sperm production and the integrity of the testes. The present study showed a high testosterone production as a result of DPP supplementation. This increase in concentration agrees with previous studies by Al-Samarrai et al. (2017), who found that supplementation of bucks with different concentrations of DPP daily for one month significantly elevated blood testosterone levels due to the effect of DPP on the function of the testes of treated bucks. Additionally, DPP supplementation resulted in an increase in both testosterone production and circulating LH levels (Iftikhar et al., 2011).
AFB1 significantly increases oxidative stress, leading to tissue damage, including the testicular tissues. The main findings of adverse effects of AFB1 include damage to spermatogonia and Leydig cells, reduced testosterone levels, and the development of sperm abnormalities and testicular dysfunction (Asadi et al., 2017). DPP has a powerful antioxidant capacity and can attenuate oxidative stress in cellular studies (Zhang et al., 2024 b). Other natural compounds, such as thymoquinone (AlGaradi et al., 2023), bergamot oil nano-emulsion (Saghir et al., 2023), and vitamin C (Ataman and Ndiokwere, 2016), have been shown to protect the testicular tissues in bucks and rats against heat stress and cisplatin.
Adipose tissue, an endocrine gland, synthesizes various adipokines, including visfatin and leptin. Research indicates that adipokines have a crucial role in improving sperm parameters and functionality, making them potential biomarkers for male fertility (Elfassy et al., 2018). However, there are insufficient studies on the effects of AFB on adipokines. Circulating adipokines, such as adiponectin, were significantly reduced due to exposure to zearalenone in pigs (Nagl et al., 2021). This effect was also observed in our study after exposure to AFB in male rabbits. Several studies have shown that DPP can improve fertility in rabbits and other mammals, and authors suggest that this improvement may be attributed to the ability of DPP to promote adipogenesis (El-Sheshtawy et al., 2016; Al-Samarrai et al., 2017; Taghian et al., 2017; Laghouati et al., 2021; Baagar et al., 2022).
Pyroptosis is a proinflammatory response involving programmed cell death and is associated with the enzymatic activity of inflammatory proteases belonging to the caspase family. AFB1 can stimulate apoptosis and induce pyroptosis, leading to an increase in cytokine markers (Zhang et al., 2019; Su et al., 2023). Consistent with these findings, our results show that AFB1 significantly upregulated neuroinflammation mediators such as NLRP3, GSDMD, Caspase-1, and IL-18 genes. DPP can target these genes due to its anti-inflammatory effects (Al-Azhary et al., 2023). Deficiency of Caspase-1 protects organs especially the testes from inflammation and apoptosis, making it a potential therapeutic target (Bergsbaken et al., 2009). The cytosolic signaling complex, NLRP3 activates many inflammatory mediators that are related to the pathogenesis of various diseases (Mangan et al., 2018). In this study, the DPP mitigated the upregulation of NLRP3 induced by AFB1. Curcumin relieved AFB1-induced hepatic pyroptosis by adjusting the action of NLRP3 in bucks (Cui et al., 2023). This mechanism was also identified in our study through DPP therapy in tissue samples. IL-18 is another pyroptosis marker and an inflammatory cytokine. This cytokine has been verified to be connected with several disorders, cancer, HBV infection, (Bao et al., 2015), and toxicity (Cui et al., 2023). GSDMD, Gasdermin D is a crucial mediator of the process of pyroptosis that appears after inflammasome activation as part of the innate immune response. DPP is a potential agent for protecting testicular tissues from the toxicity of AFB via downregulation of GSDMD, NLRP3, IL-18 and Caspase-1. Further clarification is needed on the therapeutic protective activity of DPP to alleviate pyroptosis pathways, which may be linked to their antioxidative potential, especially during the fertility period.
This study highlights the detrimental impact of aflatoxin B1 (AFB1) at 0.3 mg/kg diet on male rabbit reproductive health, serving as a relevant model for other mammalian species. AFB1 exposure was shown to disrupt blood biochemistry, induce oxidative stress, damage testicular tissue, imbalance hormones, dysregulate adipokines, and promote pyroptosis in the testes, ultimately causing infertility. Crucially, dietary supplementation with 300 mg/kg of DPP, a natural anti-pyroptotic agent, effectively mitigated this AFB1-induced testicular toxicity. This protective strategy could offer significant economic advantages to rabbit breeders by preserving reproductive function. Future research should delve into the specific mechanisms of action of DPP's bioactive compounds to further enhance the productivity of domestic animals. A multidisciplinary approach is vital for fully understanding and leveraging the potential of these natural compounds.