Fig. 1.
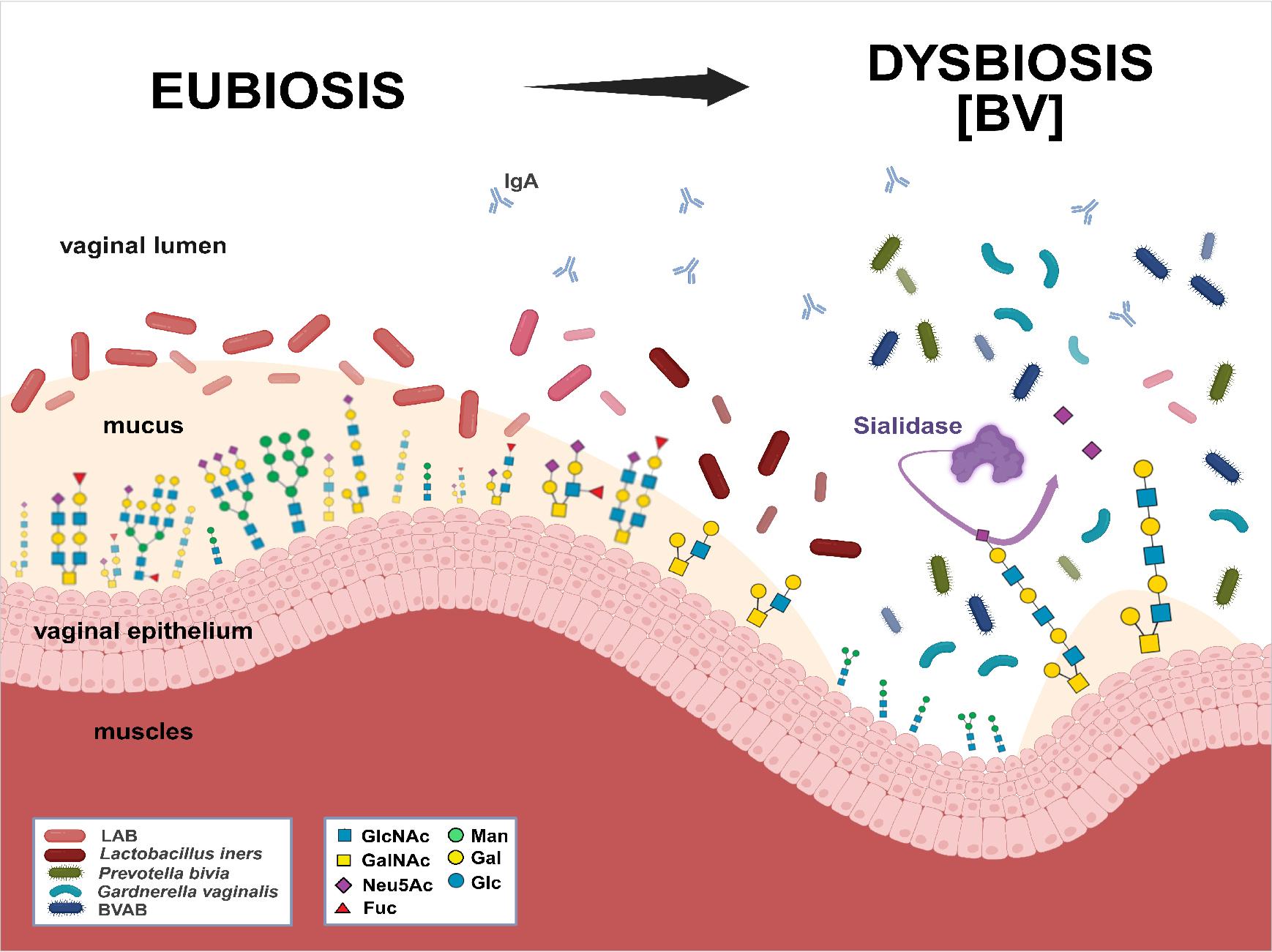
Fig. 2.
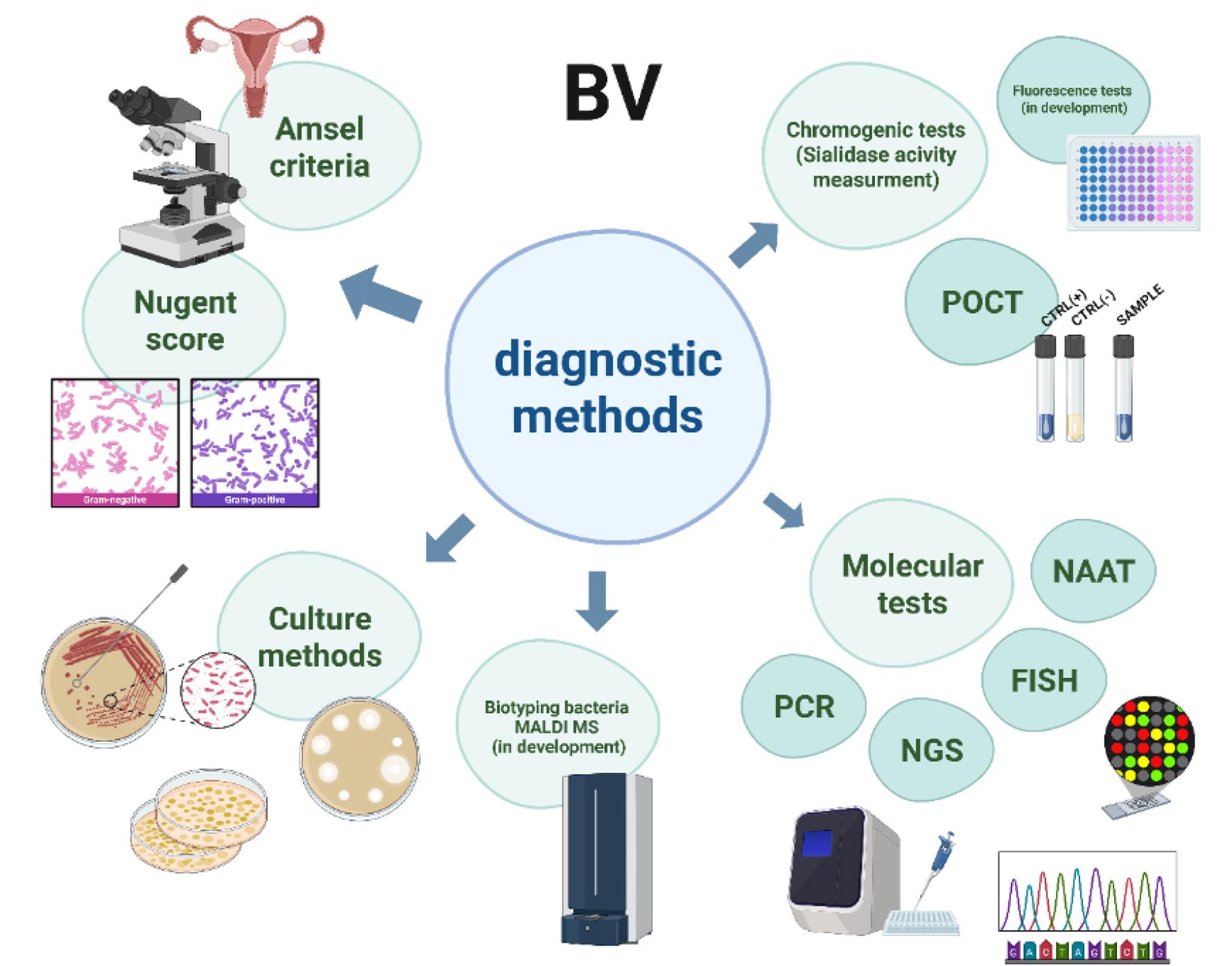
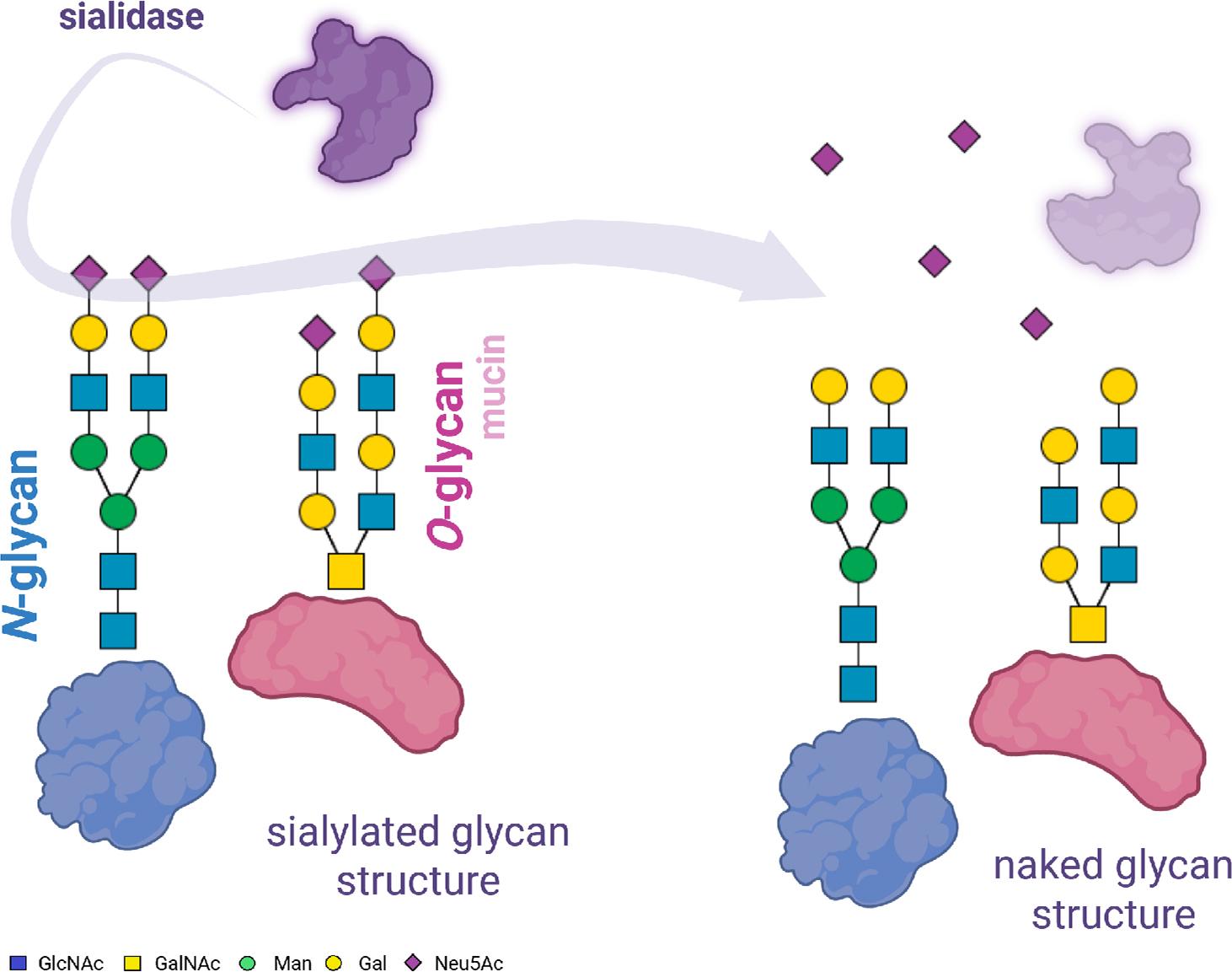
Fig. 3.
Comparison of bacterial and human sialidases_
| Organism | Sialidase type | Substrate | Result of action | References |
|---|---|---|---|---|
| Gardnarella vaginalis | GvNanH1 | α-2,3 and α-2,6-linked Neu5Ac or Neu5Gc on N- and O-glycans in the vaginal and cervical environment | major virulence factor to BV; diagnostic marker (GvNanH3); mucin and glycoprotein degradation; GvNanH1 and GvNanH2 are stronger obligations than GvNanH3 | Novak et al. 2023 |
| Prevotella timonensis | PtNanH1 | α-2,3 and α-2,6-linked Neu5Ac or Neu5Gc on O-glycans (mucin type) in the vaginal and cervical environment and IgA or IgG glycans | strong mucin degradation; contributes to BV and epithelial glycocalyx disruption; PtNanH2 are stronger obligations than PtNanH1 | Pelayo et al. 2024; Segui-Perez et al. 2024 |
| Streptococcus pneumoniae | NanA | α-2,3; α-2,6- and α-2,8-linked Neu5Ac on glycans in the respiratory environment and the blood-brain barrier | involved in colonization and pathogenesis of respiratory infections; NanA is exo- or hydrolytic sialidase, hydrolyzing its substrates to release Neu5Ac; NanB is intramolecular trans-sialidase | Manco et al. 2006; Tseng et al. 2021 |
| NanB | α-2,3 linked Neu5Ac on glycans in the respiratory environment | |||
| Clostridium perfringens | NanH | α-2,3; α-2,6- and α-2,8-linked sialid acid in glycoproteins, glycolipids, colominic acid | tissue degradation; virulence factor | Wang 2020 |
| Homo sapiens | NEU1 | α-2,3 and α-2,6-linked Neu5Ac on N- and O-glycans in the every cell organism | NEU1 is a lysosomal sialidase, NEU3 is a plasma membrane-associated sialidase, NEU4 is found in lysosomes, mitochondria, and the endoplasmic reticulum; glycan remodeling, potential role in homeostasis and dysbiosis | Zhou et al. 2020, Chen et al. 2024 |