Bacterial vaginosis (BV) is a complex, multifactorial inflammatory condition of the vagina. This disorder is associated with a shift in the vaginal epithelial microbiota from a state dominated by commensal lactic acid bacteria (LAB) to one dominated by facultative and anaerobic bacteria (Bradshaw et al. 2025). Under normal conditions, LAB counteract vaginal infection by producing immunomodulatory and antibacterial factors such as L- and D-lactic acid, hydrogen peroxide, bacteriocins, and arginine deaminase (Perez-Ibave et al. 2025). An additional protective function of the uterus and vagina is performed by a layer of mucus produced by the epithelial cells of the uterus and vagina, rich in mucins (glycoproteins with a high content of O-glycan sugar structures and a small amount of N-glycans) and immunologically active components. It acts as a natural vaginal lubricant, a barrier against pathogens and the development of infections, and a protector of cells from physical and chemical damage. In addition, mucus is secreted as a liquid fraction by the epithelial cells of the uterus and vagina, providing another type of protection. The secreted fluid is a heterogeneous mixture of glycoproteins, antibacterial peptides, secreted IgA (sIgA) antibodies, cathepsin G, lysozyme, lactoferrin, leukocytes, and epithelial cells (Lacroix et al. 2020).
It is believed that the relatively recently discovered species of bacteria in the genus Lactobacillus, L. iners, is a transitional species between eubiosis and dysbiosis and promotes the development of BV. L. iners, because of its limited ability to produce D-lactic acid with antibacterial properties, exhibits little or no protective capacity against pathogens (Bradshaw et al. 2025). Pathogens responsible for the development of BV include facultative anaerobic and anaerobic microorganisms such as Gardnerella vaginalis, Prevotella spp., Atopobium vaginae, Bacteroides spp., Mobiluncus spp., and mycoplasmas (Fig. 1). BV can promote further infections, including sexually transmitted infections (STIs), which may be caused by Trichomonas vaginalis, Chlamydia trachomatis, Neisseria gonorrhoeae, HIV (human immunodeficiency virus), and HPV (human papillomavirus) (Wessman et al. 2017, Bradshaw et al. 2025). In addition, BV increases the risk of premature birth, inflammation of the fetal membranes and placenta, inflammation of the lower pelvic organs, and inflammation of the endometrium. During the course of the infection, uniform, creamy, gray-colored vaginal discharge with a characteristic odor is observed. It is one of the key clinical diagnostic features in patients, according to Amsel’s criteria. However, BV can be asymptomatic in up to 50% of infected women (Amsel et al. 1983).
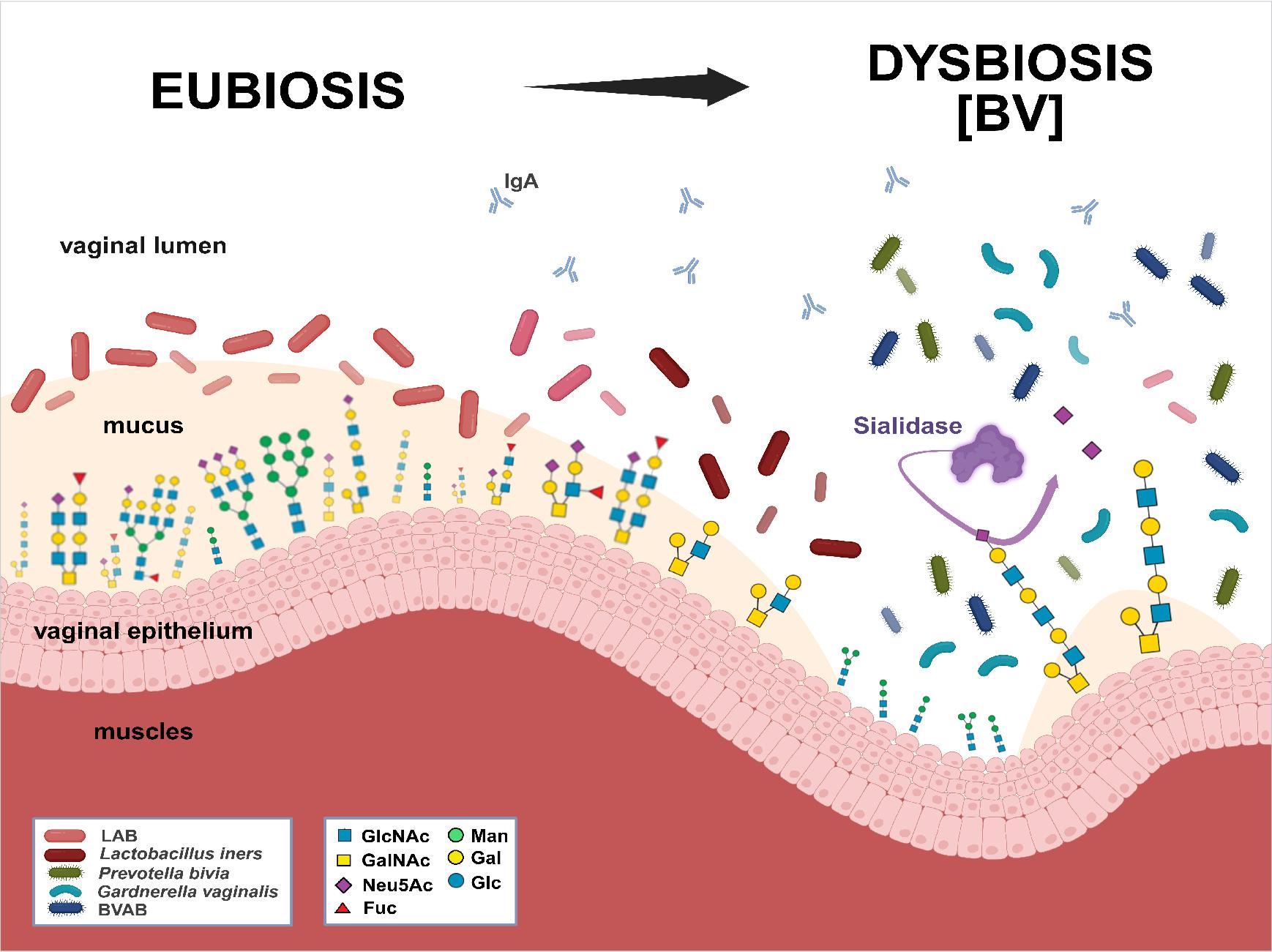
Bacterial shift from eubiosis to dysbiosis in bacterial vaginosis (BV) development. Own graphic design according to Biorender software (https://BioRender.com/ulmg6r6).
Legend: LAB – Lactic Acid Bacteria; BVAB – BV-associated bacteria; sugar residues forming the structures of N- and O-glycans: GlcNAc – N-acetylglucosamine; GalNAc – N-acetylgalactosamine; Man – Mannose; Gal – Galactose; Neu5Ac – N-acetylneuraminic acid; Glc – Glucose; Fuc – Fucose.
Sialidases (commonly referred to as neuraminidases) are hydrolytic enzymes produced, among others, by bacteria associated with BV, such as G. vaginalis, Prevotella spp., and Mobiluncus. Their primary function is to cleave terminal sialic acid residues from glycoconjugates present, among others, in cervicovaginal fluid and on the vaginal epithelial surface. Desialylation alters the structure and function of glycans, facilitating degradation of the mucosal barrier, promoting bacterial adhesion, and enabling biofilm formation. Furthermore, the released sugars serve as an energy source for pathogens. Bacterial sialidase activity is used as a diagnostic marker for BV (e.g., in the BVBlue test). It represents a potential therapeutic target in strategies to limit colonization and biofilm (Myziuk et al. 2003, Arroyo-Moreno et al. 2022, Chen et al. 2024).
This paper aims to assess the role of sialidases in the pathogenesis of bacterial vaginosis, with particular emphasis on their effects on mucosal barrier degradation and biofilm formation, as well as their potential diagnostic and therapeutic significance.
BV is diagnosed primarily using the Nugent scale, Amsel criteria, and vaginal pH (pH > 4.5). Although these methods were described about 30 years ago, the Nugent scale, based on Gram staining, remains the gold standard for BV diagnosis. BV is characterized by clue cells, i. e., epithelial cells coated with bacteria such as G. vaginalis, in microscopic preparations of vaginal smears. The Nugent scale evaluates the relative abundance of lactobacilli, small Gram- negative or variable rods (Gardnerella, Bacteroides), and curved rods (Mobiluncus). Clinical diagnosis of BV using Amsel criteria requires at least three of four findings: homogeneous milky discharge, clue cells on microscopy, vaginal pH > 4.5, and a fishy odor before or after adding 10% KOH (Amsel et al. 1983, Nugent et al. 1991). Currently, BV diagnosis can also be based on culture techniques or molecular biology tests. Molecular diagnostic techniques have significantly enhanced the accuracy and sensitivity of BV detection compared to traditional methods such as Amsel criteria or Nugent’ s scoring. These advanced approaches, including nucleic acid amplification tests (NAATs) or 16 S rRNA gene sequencing, enable the identification and quantification of key bacterial species associated with BV (Danby et al. 2021, Ferreira et al. 2022). Unlike conventional microscopy, molecular methods can detect fastidious or uncultivable organisms and provide insights into the complex composition of the vaginal microbiota. Moreover, they allow differentiation between Lactobacillus species, which is crucial for understanding the microbial shifts underlying BV (Perez-Ibave et al. 2025). Fluorescence in situ hybridization (FISH) has also emerged as a valuable tool, enabling the visualization of bacterial biofilms directly on vaginal epithelial cells and confirming the presence of BV polymicrobial communities. These tools are particularly valuable in research and clinical settings for diagnosing recurrent or asymptomatic cases and for monitoring treatment efficacy and recurrence risk (Bradshaw et al. 2025).
Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry (MALDI-MS) is a method used to diagnose bacterial vaginosis. This technique enables rapid, high-resolution identification of microorganisms isolated from clinical samples, a crucial step in diagnosing bacterial infections. MALDI-MS replaces traditional methods for identifying microorganisms in clinical laboratories. Bacterial biotyping is based on the analysis of mass spectra of protein profiles characteristic of a given microorganism species. The main advantages of this method include short analysis time, high specificity, and lower operating costs than molecular methods. Limitations include difficulty differentiating closely related strains and dependence of result accuracy on the quality and completeness of the reference database. Nevertheless, MALDI MS may become the future gold standard in modern microbiological diagnostics because of its efficiency and reproducibility (Rychert 2019).
A promising disposable paper-based microfluidic test has been developed for rapid point-of-care diagnosis of BV. The device detects a BV-specific biomarker using an innovative immunosensing approach and has been successfully validated in clinical samples (Avila-Huerta et al. 2023). Other studies suggest the potential use of fluorescent carbon dot-based methods to detect neuraminidase activity and image sialic acid for BV diagnosis (Liu et al. 2023). Another approach to diagnosing BV is to assess sialidase activity. Tests that enable rapid diagnosis of bacterial vaginosis (BV) detect bacterial sialidase activity in vaginal secretions. These assays are classified as point-of-care tests (POCT) and may complement classical diagnostic criteria, such as the Amsel or Nugent criteria. An example of this type of test is the BVBlue test, a commercially available chromogenic POCT designed to detect elevated vaginal sialidase activity produced by BV pathogens (Myziuk et al. 2003, Bradshaw et al. 2025). The methods outlined above are depicted in Figure 2.
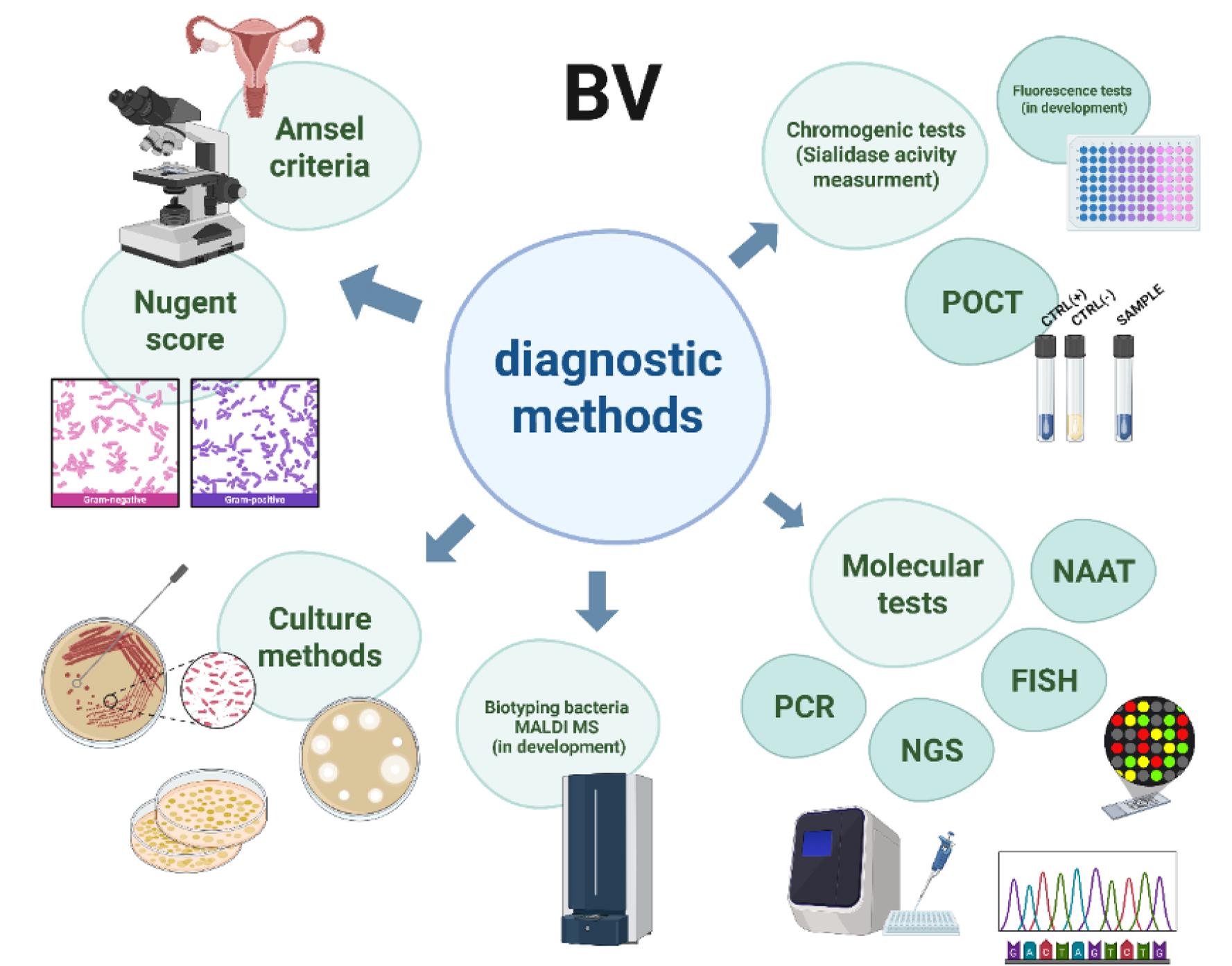
Overview of bacterial vaginosis diagnosis. Own graphic design using Biorender software (https://BioRender.com/entt8c0).
Legend: BV – Bacterial Vaginosis; POCT – Point-Of-Care Tests; NAAT – Nucleic Acid Amplification Test; FISH – Fluorescence In Situ Hybridization; NGS – Next-Generation Sequencing; PCR – Polymerase Chain Reaction; MALDI MS – Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry.
Bacterial overgrowth in BV is associated with the production of many virulence factors, including glycosidases (e.g., sialidases), proteases, and sulfatases. Bacterial enzymes modify host molecules and alter the local environment to support microbial survival and interaction with host tissues. Through these activities, they contribute to processes such as nutrient acquisition, tissue remodeling, and modulation of immune responses. Glycosidase activity alters the sugar composition of glycoproteins. As a result, in dysbiosis, the glycan composition of the vaginal mucosa and secreted fluid differs from that in eubiosis (Bonnardel et al. 2021).
The Glyco-Evasion Hypothesis, proposed by Kreisman and Cobb (2012), suggests that microbes can evade host immune responses not only by mimicking host-like surface glycans but also by actively modulating host glycosylation pathways. In the vaginal environment, pathogens may alter epithelial glycosylation to suppress immune detection or inflammation, thereby promoting persistent colonization or infection (Fig. 1). Sialidases, hydrolytic enzymes that degrade sialylated glycans, exhibit optimal activity at the slightly acidic vaginal pH. The loss of sialic acid residues leaves glycans naked, devoid of negative charge and susceptible to further hydrolysis by other enzymes. The loss of the negative charge imparted by sialic acid in both glycans and the mucosal layer disrupts the mucous layer’s physiological functions (Chen et al. 2024) (Fig. 3).
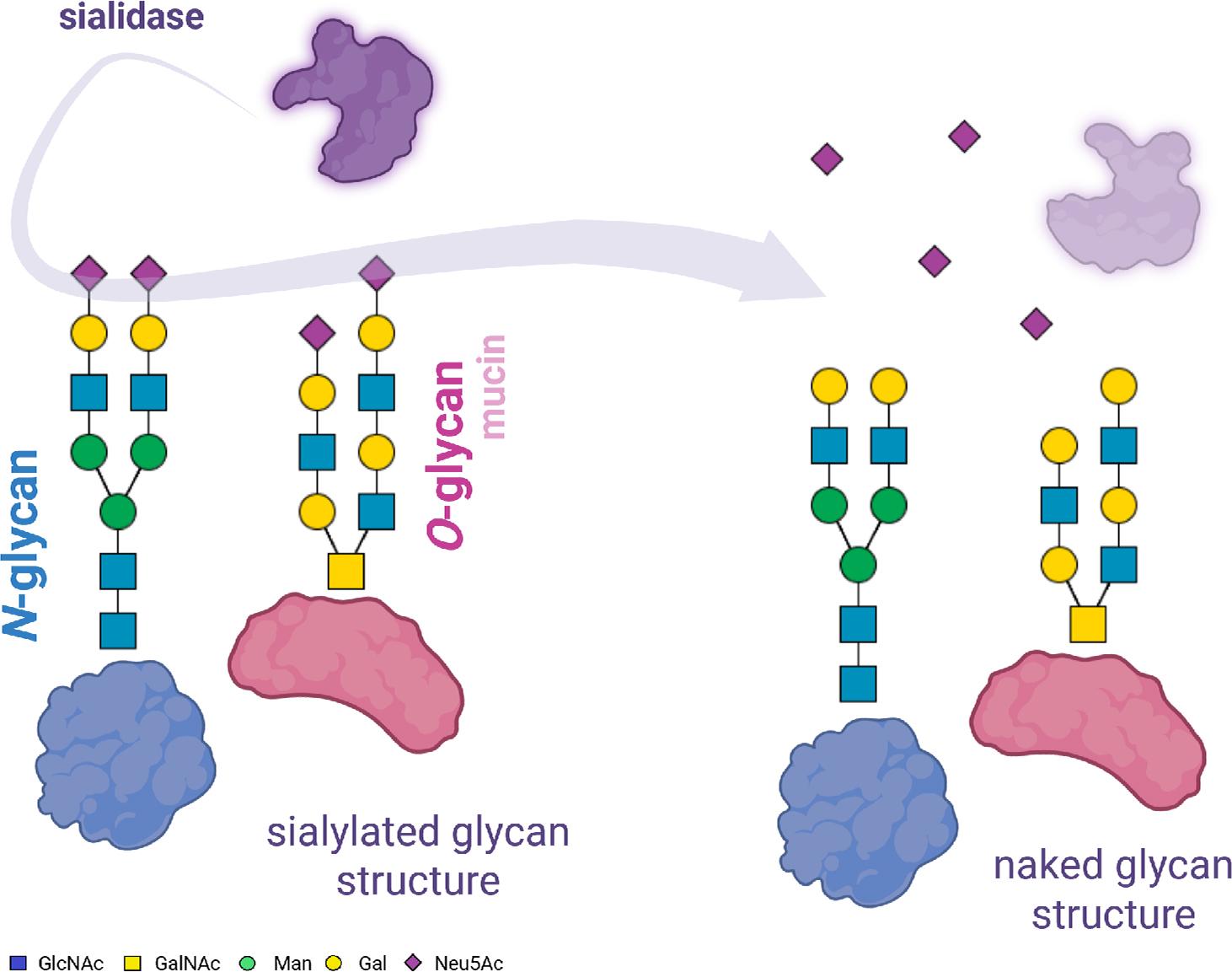
Sialidase mechanism of action. Own graphic design according to Biorender software (https://biorender.com/97qvka8).
Legend: Sugar residues forming the structures of N- and O-glycans: GlcNAc – N-acetylglucosamine; GalNAc – N-acetylgalactosamine; Man – Mannose; Gal – Galactose; Neu5Ac – N-acetylneuraminic acid.
The destruction of the mucous membrane lining the vagina promotes the adhesion of BV pathogens. In addition, glycans on IgA secretory antibodies and cytokines are destroyed, disrupting the host’s immune response to infection (Lewis et al. 2012). The expression of sialidases varies among bacterial strains and may influence their invasive potential. Sialidases are produced by various bacterial genera, not only those associated with infections of the female genital tract. Although bacterial sialidases are the primary contributors to desialylation in BV, human sialidases may also play a role in maintaining mucosal homeostasis or contributing to dysbiosis under certain conditions. It is suggested that vaginal epithelial cells may express endogenous human sialidases NEU1, NEU3, and NEU4, which regulate mucosal immunity and glycan remodeling. These sialidases differ in their subcellular localization, substrate preferences, and consequently in their biological functions. NEU1 is a ubiquitously expressed enzyme located in lysosomes and on the cell membrane, acting on oligosaccharides, glycopeptides, and glycoproteins. NEU3 primarily degrades gangliosides at the outer or inner leaflet of the plasma membrane. NEU4, found on mitochondria and possibly in lysosomes or the endoplasmic reticulum, exhibits broad substrate specificity and participates in ganglioside catabolism (Chen et al. 2024) (Table 1).
Comparison of bacterial and human sialidases.
| Organism | Sialidase type | Substrate | Result of action | References |
|---|---|---|---|---|
| Gardnarella vaginalis | GvNanH1 | α-2,3 and α-2,6-linked Neu5Ac or Neu5Gc on N- and O-glycans in the vaginal and cervical environment | major virulence factor to BV; diagnostic marker (GvNanH3); mucin and glycoprotein degradation; GvNanH1 and GvNanH2 are stronger obligations than GvNanH3 | Novak et al. 2023 |
| Prevotella timonensis | PtNanH1 | α-2,3 and α-2,6-linked Neu5Ac or Neu5Gc on O-glycans (mucin type) in the vaginal and cervical environment and IgA or IgG glycans | strong mucin degradation; contributes to BV and epithelial glycocalyx disruption; PtNanH2 are stronger obligations than PtNanH1 | Pelayo et al. 2024; Segui-Perez et al. 2024 |
| Streptococcus pneumoniae | NanA | α-2,3; α-2,6- and α-2,8-linked Neu5Ac on glycans in the respiratory environment and the blood-brain barrier | involved in colonization and pathogenesis of respiratory infections; NanA is exo- or hydrolytic sialidase, hydrolyzing its substrates to release Neu5Ac; NanB is intramolecular trans-sialidase | Manco et al. 2006; Tseng et al. 2021 |
| NanB | α-2,3 linked Neu5Ac on glycans in the respiratory environment | |||
| Clostridium perfringens | NanH | α-2,3; α-2,6- and α-2,8-linked sialid acid in glycoproteins, glycolipids, colominic acid | tissue degradation; virulence factor | Wang 2020 |
| Homo sapiens | NEU1 | α-2,3 and α-2,6-linked Neu5Ac on N- and O-glycans in the every cell organism | NEU1 is a lysosomal sialidase, NEU3 is a plasma membrane-associated sialidase, NEU4 is found in lysosomes, mitochondria, and the endoplasmic reticulum; glycan remodeling, potential role in homeostasis and dysbiosis | Zhou et al. 2020, Chen et al. 2024 |
Interestingly, the pore-forming toxin vaginolysin, produced by G. vaginalis, through its cytolytic activity, provides epithelial components for N. gonorrhoeae, facilitating its colonization. In addition, N. gonorrhoeae uses sialic acid residues in the surrounding vaginal environment to evade immune cells. This is known as antigenic mimicry (Morrill et al. 2023). Although G. vaginalis is considered the primary pathogen of BV, scientific studies have shown that this bacterium requires ammonia and sialidases produced by P. bivia for epithelial colonization. The presence of sialidase-producing P. bivia strains may enhance mucin degradation and facilitate the colonization and persistence of G. vaginalis and other BV-associated bacteria (BVAB) through synergistic interactions (Segui-Perez et al. 2024).
G. vaginalis expresses multiple sialidase isoforms with distinct localizations and functions. G. vaginalis possesses three genes: nanH1, nanH2, and nanH3, which encode GvNanH1, GvNanH2 and GvNanH3 sialidases. However, the nanH2 and nanH3 genes are present only in certain strains. G. vaginalis sialidases hydrolyze α2,3- and α2,6-linked sialic acid residues on glycoproteins and mucins. GvNanH1 sialidase isoforms are thought to be intracellular enzymes with limited activity, whereas GvNanH2 and GvNanH3 isoforms are highly active extracellular sialidases that degrade mucins, promote biofilm formation, and compromise the integrity of the epithelial barrier. The GvNanH3 sialidase is the most active sialidase among clinical isolates of G. vaginalis, correlates with the presence of BV, and may serve as a biomarker for this condition (Novak et al. 2023). Notably, the nanH3 gene has been specifically linked to persistent high-risk HPV infections and is considered a potential virulence marker in certain G. vaginalis strains. These isoforms enable G. vaginalis to adapt to different host environments and may influence the severity and persistence of BV (Novak et al. 2023). Additionally, recent research has shown that GvNanH2 can significantly affect sperm function by altering the sperm glycocalyx (Dohadwala et al. 2025).
P. bivia exhibits lower sialidase activity than G. vaginalis, but it also contributes to the sialidase pool in the vaginal microbiome. Recent studies have identified the sialidase nanH gene in P. bivia. The product of this gene shows activity against mucin-type substrates, although at lower levels than in other Prevotella species, such as P. timonensis (Arroyo-Moreno et al. 2022, Novak et al. 2023). P. timonensis encodes two sialidases: PtNanH1 and PtNanH2. Both enzymes degrade mucins in vaginal mucus and exhibit activity against α2,3- and α2,6-linked sialic acid residues. Studies have shown that P. timonensis is one of the major producers of sialidases in the vaginal microbiota, which may contribute to dysbiosis and increased susceptibility to other infections (Pelayo et al. 2024; Segui-Perez et al. 2024). In summary, the bacteria involved in the development of bacterial vaginosis that produce sialidases include G. vaginalis, P. timonensis, P. bivia, and BV-associated bacteria such as Mobiluncus spp. and Bacteroides spp. (Morrill et al. 2023).
Bacterial sialidases are believed to serve as markers of BV, and the activity of these enzymes in mucus secreted by the vaginal epithelium may be a diagnostic target. In routine tests for sialidase activity, mainly used in scientific research, sialic acids conjugated to a protochromophore, chemiluminescent, or fluorophore label are used as substrates. Depending on the substrate used, sialidases favor the α-linkage of the O-glycosidic bond, such as α2,3- or α2,6-linked sialic acid. As a result of sialidase activity, the labels are released from sialic acid, and the labels exhibit luminescence depending on the type of label. Subsequently, a measurement is performed according to the label used: spectrophotometric, chemiluminescent, or fluorescent. An example is the biochemiluminescence test, in which sialic acid is conjugated with luciferin. In the presence of sialidases in the test sample, luciferase is released and subsequently oxidized by firefly luciferin, generating a light signal that is detected by appropriate equipment. The test described is proposed for diagnosing BV. It is a quantitative test with high sensitivity and specificity. Other advanced methods for assessing sialidases, such as HPLC or LC-MS, are also proposed. Due to the high cost of substrates and their properties, there remains a need to refine methods for biological samples (Wu et al. 2019, Yuan et al. 2020). In the study by Wu et al. (2019), patients diagnosed with bacterial vaginosis via the Amsel criteria were further stratified based on sialidase activity measured by a chemiluminescent assay: approximately 27.6% of BV-positive individuals exhibited high sialidase levels (signal-to-cutoff ratio ≥10), enabling differentiation of patients by biomarker activity for risk assessment and monitoring purposes (Wu et al. 2019).
However, qualitative chromogenic tests (BVBlue test) can detect elevated sialidase activity in vaginal fluid samples in a gynecologist’s practice or a diagnostic laboratory. These tests are classified as POCT (point-of-care tests). The BVBlue test detects elevated vaginal sialidase activity (≥7.8 U) produced by BV pathogens. One unit of sialidase activity is defined as the amount of enzyme that releases 1 nmol of substrate per milliliter per minute at 37 °C. These tests are relatively quick compared to the Amsel or Nugent criteria. They provide results within minutes, enabling prompt diagnosis and treatment. The principles of these tests are similar to those of the chromogenic methods described above. In the presence of sialidases from a vaginal swab or vaginal fluid, the substrate in the reagent is broken down, and the addition of a specific reagent causes a color change in the reaction suspension. The result is read visually. Depending on the test type, the severity of the infection can be assessed by the intensity of the color. These tests are specific, sensitive, and accurate, as well as quick, and do not require additional equipment, such as an incubator. These tests are beneficial when BV is suspected and standard diagnostic methods, such as microscopy, are unavailable, provided that other infections have been ruled out. Additionally, similar to the Amsel criteria, they do not assess the severity of BV symptoms. For accurate results, the test should not be used within 72 hours of douching, vaginal intercourse, or use of intravaginal products (Muzny et al. 2023, Myziuk et al. 2003). The BVBlue test is considered an accurate and rapid diagnostic tool for bacterial vaginosis, with a specificity of 91–100% and a sensitivity of 38–91.7%, comparable to that of diagnostics based on the Nugent scale or Amsel criteria. However, its reliability depends on the local vaginal micro-biota epidemiology, so population-specific studies are recommended before routine use. In the study evaluating the BVBlue test, patients were stratified according to Amsel criteria and the Nugent score, demonstrating that positive BVBlue results strongly correlated with elevated vaginal pH, a positive amine test, the presence of clue cells, and a prior history of BV treatment, highlighting its diagnostic reliability (Myziuk 2003).
In recent years, sialidases have attracted attention as potential therapeutic targets. Research has shown that inhibiting their activity may reduce bacterial invasiveness and improve treatment outcomes, particularly in recurrent cases. The use of influenza medications offers a novel approach to BV treatment (Chen et al. 2024). For example, the zanamivir has demonstrated partial inhibition of sialidase GvNanH2 activity. Zanamivir reduced G. vaginalis sialidase activity by 30% and decreased its ability to invade host cells by 50% (Govinden et al. 2018).
Additionally, deoxy-dehydro-sialic acid (DDSia), a synthetic sialidase inhibitor, prevents the desialylation of secretory IgA during incubation with BV samples (Lewis et al. 2012). However, sensitivity to sialidase inhibitors varies among microbial and viral enzymes. Although no drugs targeting sialidases are currently explicitly approved for BV, ongoing studies suggest they could be used as adjuncts or alternatives to antibiotics, particularly given increasing resistance and high recurrence rates (Wong et al. 2018). Surprisingly, Motamedi et al. presented a novel approach to combating bacterial vaginosis through a multi-epitope vaccine targeting G. vaginalis. Designed using bioinformatics and immunoinformatics tools, the construct demonstrated high antigenicity, non-allergenicity, and structural stability, with molecular docking and dynamics confirming strong binding interactions. Immune simulation showed elevated immunoglobulin levels and cytokine responses, suggesting potential for long-term immunity, pending experimental validation (Motamedi et al. 2025).
Sialidase has emerged as a key biomarker for bacterial vaginosis, with significant potential to improve diagnostic accuracy and enable rapid point-of-care testing (POCT). Such tests are crucial for timely diagnosis, especially in resource-limited settings, because they reduce reliance on specialized laboratories and trained personnel. Given their essential role in BV pathogenesis, sialidases may be explored not only as diagnostic markers but also as therapeutic targets for drugs with properties beyond traditional antibiotics. Recent studies indicate that inhibiting sialidase activity can reduce bacterial invasiveness and improve treatment outcomes, particularly in recurrent BV. Moreover, diagnostic tests that detect sialidase presence and activity are valuable for assessing BV risk, confirming diagnosis, and monitoring the effectiveness of antibiotic therapy. Understanding sialidase’s immunogenic properties further opens the door to preventive strategies, including vaccine development, underscoring its relevance for future interventions in women’s health.