Figure 1.
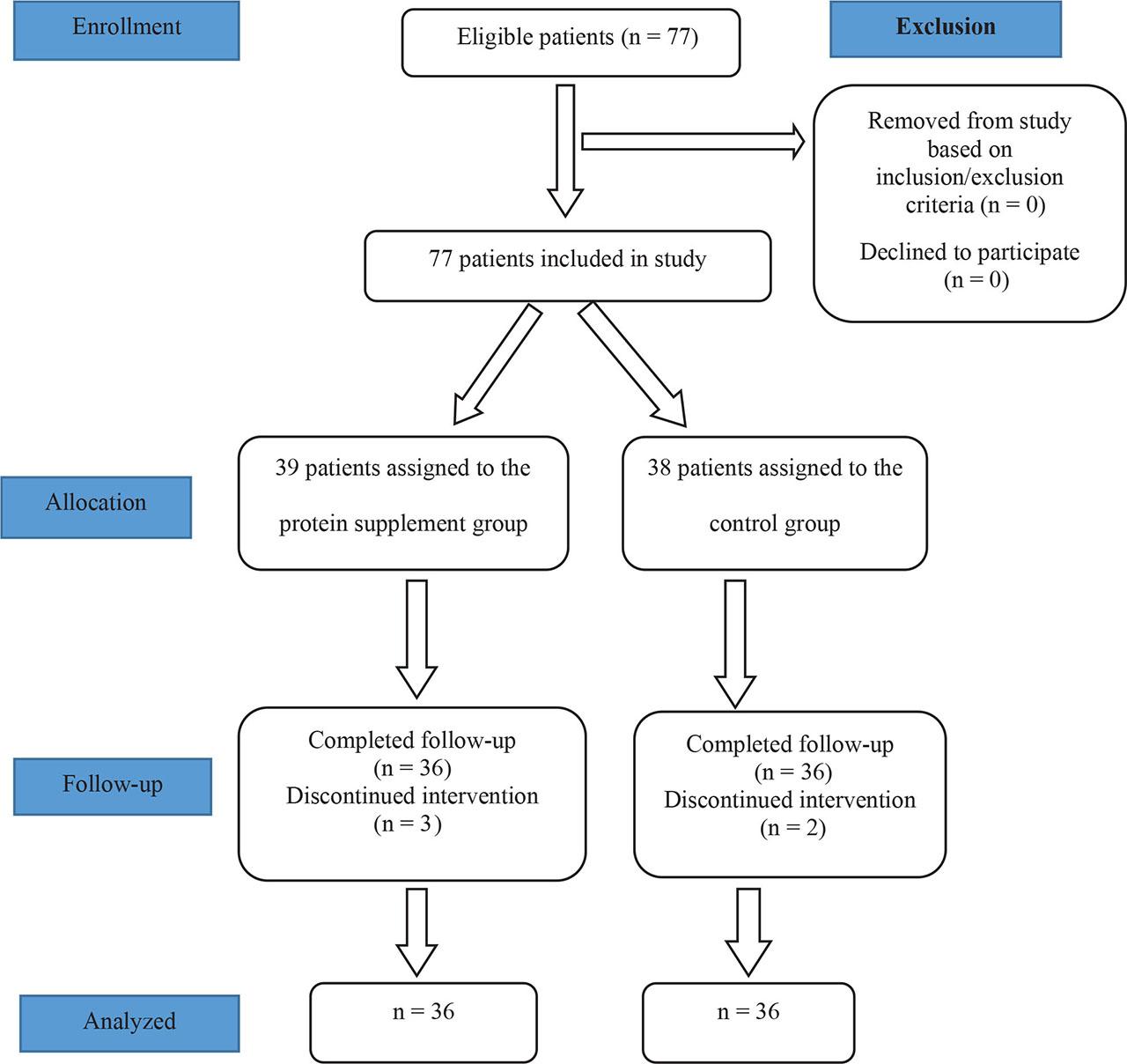
The comparison of laboratory data of neonates in the protein supplement and control groups during trial
| Test | Week | Groups | P-value* | |
|---|---|---|---|---|
| Protein Supplement | Control group | |||
| BUN (mg/dl), mean ± SD | Baseline | 6.06 ± 1.39 | 6.71 ± 2.80 | 0.216 |
| 1 | 5.56 ± 1.24 | 6.39 ± 2.39 | 0.069 | |
| 2 | 5.97 ± 1.21 | 5.84 ± 1.97 | 0.739 | |
| Albumin (g/dL), mean ± SD | Baseline | 3.18 ± 0.33 | 3.13 ± 0.41 | 0.570 |
| 1 | 3.41 ± 0.40 | 3.21 ± 0.37 | 0.031 | |
| 2 | 3.59 ± 0.37 | 3.24 ± 0.40 | 0.000 | |
| PH, mean ± SD | Baseline | 7.34 ± 0.05 | 7.33 ± 0.06 | 0.044 |
| 1 | 7.33 ± 0.04 | 7.36 ± 0.05 | 0.006 | |
| 2 | 7.33 ± 0.06 | 7.36 ± 0.04 | 0.015 | |
| PCO2 (mmHg), mean ± SD | Baseline | 35.12 ± 6.54 | 35.97 ± 5.10 | 0.544 |
| 1 | 39.20 ± 5.93 | 35.41 ± 5.95 | 0.008 | |
| 2 | 41.53 ± 6.68 | 34.63 ± 7.82 | 0.000 | |
| HCO3 (mmol/L), mean ± SD | Baseline | 18.19 ± 4.33 | 18.39 ± 4.87 | 0.854 |
| 1 | 20.47 ± 5.03 | 19.63 ± 4.72 | 0.008 | |
| 2 | 21.06 ± 5.25 | 19.58 ± 4.67 | 0.004 | |
The comparison of protein intake, duration of trial, and anthropometric parameters (outcomes) in the intervention and control groups
| Variable | Groups | P-value* | |
|---|---|---|---|
| Protein supplement | Control | ||
| Protein Intake(gr/kg/day), mean ± SD | 4.55 ± 0.18 | 3.66 ± 0.22 | <0.001 |
| Duration of trial (days), mean ± SD | 10.39 ± 4.42 | 12.50 ± 5.71 | 0.097 |
| Weight (gram), mean ± SD | |||
| Before intervention (gr) | 1276.52 ± 127.23 | 1282.06 ± 118.30 | 0.854 |
| After intervention (gr) (Discharge time) | 1518.79 ± 59.78 | 1455.88 ± 50.38 | <0.001 |
| Gain (gr/kg/day) | 19.12 ± 7.22 | 11.14 ± 5.29 | <0.001 |
| Weight Percentile, n (%) | 0.040 | ||
| After intervention | |||
| ≤ 3 | 6 (16.7) | 16 (44.4) | |
| 3 – 10 | 16 (44.4) | 11 (30.6) | |
| > 10 | 14 (38.9) | 9 (25.0) | |
| Increased head circumference rate (cm/week), mean ± SD | 0.872 ± 0.262 | 0.525 ± 0.210 | <0.001 |
| Increased length rate (cm/week), mean ± SD | 1.00 ± 0.35 | 0.62 ± 0.20 | <0.001 |
Comparison of baseline characteristics of the neonates in the intervention and control groups before the clinical trial
| Variable | Groups | P-value* | |
|---|---|---|---|
| Protein supplement | Control | ||
| Gender, n (%) | 0.345 | ||
| Male | 17 (47.2) | 21 (58.3) | |
| Female | 19 (52.8) | 15 (41.7) | |
| Birth weight (gr), mean ± SD | 1210.3 ± 189.53 | 1229.7 ± 169.6 | 0.660 |
| Birth weight percentile, n (%) | 0.798 | ||
| ≤ 3 | 3 (8.3) | 1 (2.8) | |
| 3 – 10 | 4 (11.1) | 5 (13.9) | |
| > 10 | 29 (80.6) | 30 (83.3) | |
| Gestational age (weeks), mean ± SD | 30.50 ± 1.83 | 30.58 ± 1.70 | 0.850 |
| Gestational age groups (weeks), n (%) | 0.735 | ||
| < 29 | 12 (33.3) | 9 (25.0) | |
| 29 – 31 | 11 (30.6) | 12 (33.3) | |
| 31 – 33 | 13 (36.1) | 15 (41.7) | |
| Birth head circumference (cm), mean ± SD | 26.6 ± 1.7 | 27.0 ± 1.5 | 0.600 |
| Birth length(cm), mean ± SD | 37.5 ± 17.63 | 37.3 ± 15.75 | 0.665 |
| Age (days), mean ± SD | 13.7 ± 9.0 | 11.6 ± 7.1 | 0.291 |
| Type of milk, n (%) | 1.000 | ||
| Formula | 19 (52.8) | 19 (52.8) | |
| FBM | 17 (47.2) | 17 (47.2) | |
| Type of formula, n (%) | 1.000 | ||
| Pre BeBeLac | 8 (47.1) | 7 (38.9) | |
| Pre Aptamil | 4 (23.5) | 5 (27.8) | |
| Pre NAN | 4 (23.5) | 5 (27.8) | |
| BeBeLac AR | 1 (5.9) | 1 (5.6) | |