Familial adenomatous polyposis (FAP) is an autosomal dominant inherited disease caused by pathogenic variants in the adenomatous polyposis coli (APC) gene1 with reported incidence of one in 8,000 to 12,000 live births.2 The main hallmark of the disease is the presence of multiple colorectal adenomas, leading to a 100% lifetime risk of developing cancer if the colon remains in situ.3 To prevent the development of cancer, prophylactic colectomy or proctocolectomy is performed when the adenoma burden cannot be managed endoscopically or at the age of 18–25 years old. The following types of surgery are available4: total colectomy with ileorectal anastomosis (IRA) or ileosigmoid anastomosis (ISA); proctocolectomy with/without mucosectomy and stapled ileal pouch-anal anastomosis (IPAA) or hand-sewn IPAA; and total proctocolectomy with end ileostomy. Until restorative proctocolectomy with IPAA and pouch reconstruction was described in the 1970s, colectomy with IRA or end ileostomy was the only surgical prophylactic procedure available and was associated to a considerable high CRC incidence and mortality.5 After this, proctocolectomy with pouch reconstruction (IPAA) was the technique of choice in patients with a high adenoma burden and was sought to eliminate the risk of CRC in FAP patients. However, since the first report of pouch cancer in 19946, there has been a substantial increase in published literature reporting rates of adenoma and cancer development after primary IPAA. The development of adenomas along life in remnant rectal mucosa is a natural phenomenon in this population. Long live periodical surveillance with rectoscopies is widely recommended in international guidelines as shown in Table 1.4,7,8,9,10 As there are no randomised trials comparing endoscopic surveillance and management strategies for FAP patients with IRA and IPAA, we aimed to systematically evaluate adenoma and cancer development after prophylactic surgery, define potential risk factors and to summarise endoscopic practices from published series.
Summary of recommendations from the international guidelines
| First author and publication date (ref.) | Endoscopic surveillance – patients with IRA | Indications for secondary proctectomy patients with IRA | Endoscopic surveillance – patients with IPAA |
|---|---|---|---|
| Vasen et al., 20087 | Every 3 to 6 months | Multiple large adenomas (> 5 mm) Adenomas with dysplasia | Every 6 to 12 months |
| Balmaña et al., 2013, ESMO8 | Every 12 months | No recommendations | Every 12 months |
| Stoffel et al., 2015, ASCO9 | Every 6 to 12 months | No recommendations | Every 6 months to 5 years (Intervals should be determined on a case-by-case basis and may be even shorter than 1 year for some individuals) |
| Sygnal et al., 2015, ACG10 | Every 12 months | No recommendations | Every 12 months |
| Herzig et al., 2017, ASCRS4 | Every 12 months | No recommendations | Every 12 months |
| Van Leerdam ME et al., 2019, ESGE53 | Every 12 to 24 months | No recommendations | Every 12 to 24 months |
| Yang J et al., 2020, ASGE54 | 6 months after surgery with 6 to 12 months further surveillance interval | 12 months after surgery with 12 to 24 months further surveillance interval. 6 months if advance adenoma |
ACG = American College of Gastroenterology; ASCO = American Society of Clinical Oncology; ASCRS = American Society of Colon and Rectal Surgeons; ASGE = American Society for Gastrointestinal Endoscopy; ESGE = European Society of Gastrointestinal Endoscopy; ESMO = European Society for Medical Oncology; IPAA = ileal pouch anal anastomosis; IRA = ileorectal anastomosis
Our review is reported according to the PRISMA guidelines.11
We searched PUBMED from inception to June 2023 to identify studies evaluating long-term adenoma and cancer development in patients with FAP after prophylactic surgery. Deduplication was performed using Zotero software.12 Reference lists of included studies were hand-searched for additional relevant studies. The search was limited to studies, published in English. We used the following keywords: “FAP”, “IRA”, “IPAA”, “familial adenomatous polyposis” and “proctocolectomy”.
We included single-or multicentre retrospective cohort studies, prospective cohort studies and retrospective analyses of polyposis registries. Due to the rarity of the events, we only considered case reports for inclusion when summarising reports on cancers after primary IPAA. Only the most recent series from the same institution or polyposis registry were included in the analysis, as some research groups regularly publish retrospective analyses of their cohorts or polyposis registries. Full-text screening and data extraction were performed by a single researcher (AG). Manuscripts of three case reports could not be obtained, data were summarised from the two review articles.13,14
Of 97 full-text articles screened for eligibility (Figure 1), 46 met our inclusion criteria. A further 8 articles were identified by hand searching the reference lists of the included studies (6 case reports, 1 retrospective cohort, 1 polyposis registry analysis). We included 22 retrospective analyses, 14 case reports (carcinoma development after primary IPAA), 15 retrospective analyses of prospectively maintained polyposis registries and 3 prospective cohort studies. Only 5 studies were multicentre and 1 was bi-centre. The studies were published between 1994 and 2023. The studies included between 1 and 925 patients. A total of 5010 patients were included in the review. Summary characteristics of the included studies are shown in Table 2.
Flowchart of the systematic review according to the Preferred Reporting Items for Systematic Reviews (PRISMA) schema.
Characteristics of included studies
| First author and publication date (ref.) | No. of patients | Country | Setting | Study design | Surgery performed (period) | Study population |
|---|---|---|---|---|---|---|
| Aelvoet et al., 202355 | 144 (111 IPAA, 33 ileostomy) | The Netherlands | Single | Cohort/Retrospective | / | IPAA, ileostomy |
| Tatsuta et al., 202356 | 65 (22 IRA, 20 IPAA) | Japan | Single | Cohort/Retrospective | 1976–2022 | IRA, IPAA |
| Anele et al., 202257 | 199 (199 IRA) | United Kingdom | Single | Cohort/Retrospective | 1990–2017 | IRA |
| Colletti et al., 202258 | 715 (715 IRA) | Italy | Multicentre | Retrospective analysis of the Registry | 1977–2021 | IRA |
| Pasquer et al., 202159 | 289 (197 IRA, 92 IPAA) | France | Multicentre | Retrospective analysis of the Registry | 1965–2015 | IRA, IPAA |
| Ardoino et al., 202060 | 925 (585 IRA, 340 IPAA) | Italy | Multicenter | Retrospective analysis of the Registry | 1947–2015 | IRA, IPAA |
| Tajika et al., 201916 | 47 (14 IRA, 25 IPAA, 8 ileostomy) | Japan | Single | Cohort/Retrospective | 1965–2017 | IRA, IPAA and ileostomy |
| Ganschow et al., 201861 | 192 | Germany | Singe | Cohort/Prospective and retrospective analysis of Polyposis Registry | Endoscopy data collected during 2010–2013 | IPAA |
| Kariv et al., 201762 | 45 | Israel | Single | Cohort/Retrospective | 1986–2013 | IPAA |
| Patel et al., 201642 | 21 (6 IRA, 5 IPAA, 10 intact colon) | Indianapolis, USA | Single | Cohort/Retrospective | Endoscopies performed between 2004–2016 | IRA, IPAA and intact colon |
| Walsh et al., 201663 | 1 | Ireland | Single | Case report | 1987 | IPAA - cancer |
| Maehata et al., 201520 | 27 | Japan | Single | Cohort/Retrospective | 1990–2004 | IRA |
| Ganschow et al., 201550 | 100; 50 hand-sewn and 50 stapled anastomoses | Germany | Single | Cohort/Prospective | ? | Hand-sewn vs. stapled anastomosis |
| Goldstein et al., 201563 | 59 | Israel | Single | Cohort/Retrospective | 1986–2013 | IPAA |
| Zahid et al., 201564 | 27 | Australia | Single | Cohort/Retrospective | 1984–2011 | IPAA |
| Kennedy et al., 201465 | 95; 85 hand-sewn and 1 stapled anastomosis | Rochester, Mayo Clinic, USA | Single | Cohort/Retrospective | 1987–2011 | IPAA |
| Koskenvuo et al., 201322 | 140 | Finland | Single | Cohort/Retrospective | 1963–2012 | IRA |
| Pommaret et al., 201335 | 118 | France | Single | Cohort/Retrospective | / | IPAA and IRA |
| Boostrom et al., 201366 | 117 | Rochester, Mayo Clinic, USA | Single | Cohort/Retrospective | 1972–2007 | IPAA |
| Ozdemir et al., 201337 | 260; 86 hand-sewn and 175 stapled anastomoses | Cleveland, USA | Single | Analysis of polyposis registry | 1983–2010 | Hand-sewn vs. stapled anastomosis |
| Wasmuth et al., 201367 | 61; 39 hand-sewn with mucosectomy and 22 without of which 15 were stapled and 7 hand-sewn anastomoses | Norway | Multicenter | Analysis of polyposis registry | 1986–2008 | IPAA (mucosectomy vs. no-mucosectomy) |
| Yan et al., 201268 | 42 (33 IPAA; 6 IRA ?) | China | Single | Cohort/Retrospective | 1988–2008 | IPAA and IRA |
| Makni et al., 201269 | 1 | Tunisia | Single | Case report | 1996 | IPAA - cancer |
| Tonelli et al., 201251 | 69 | Italy | Single | Cohort/Prospective data collection | 1984–2008 | IPAA |
| von Roon et al., 201170 | 140; 44 hand-sewn and 76 stapled anastomoses | UK | Single | Retrospective analysis of St. Mark’s Hospital Polyposis Registry | 1978–2007 | Hand-sewn vs. stapled anastomosis |
| Banasiewicz et al., 201132 | 165 | Poland | Bicenter | Bicenter/Retrospective analysis | 1985–2009 operated, Clinical data from endoscopy FUP between 2004–2009 | IPAA |
| Booij et al., 201018 | 43 (34 IRA) | The Netherlands | Single | Cohort/Retrospective | 1977–2005 | IRA and IPAA |
| Sinha et al., 201026 | 427 | UK | Single | Retrospective analysis of St. Mark’s Hospital Polyposis Registry | 1990–2008 | IRA |
| Ault et al., 200971 | 2 | Los Angeles, USA | Single | Case series | 1990, 1993 | IPAA - cancer |
| Nieuwenhuis et al., 200927 | 475 | Denmark, Finland, Sweden, Netherlands | Multicenter | Analysis of polyposis registry | / | IRA |
| Yamaguchi et al., 200925 | 59 | Japan | Single | Cohort/Retrospective | 1962–2007 | IRA |
| Friederich et al., 200831 | 212; 71 hand-sewn with mucosectomy and 115 stapled anastomoses | The Netherlands | Single | Analysis of National Polyposis Registry | 1985–2005 | IPAA |
| Campos et al., 200819 | 36 | Brasil | Single | Cohort/Retrospective | 1977–2006 | IRA and IPAA |
| Bullow et al., 200824 | 776; 576 operated in pre-pouch period and 200 in pouch period starting in 1990 | Denmark, Finland, Sweden, Netherlands | Multicenter | Analysis of polyposis registry | 1950–2006 | IRA |
| Gleeson et al., 200830 | 16 | Rochester, Mayo Clinic, USA | Single | Cohort/Retrospective analysis | 1964–2003(Analysis of endoscopies between 1992–2006) | IPAA and IRA |
| Lee et al., 200872 | 1 | Korea | Single | Case report | 1998 | IPAA - cancer |
| Linehan et al., 200773 | 1 | Ireland | Single | Case report | 1997 | IPAA - cancer |
| Valanzano et al., 200728 | 25 | Italy | Single | Cohort/Prospective | 1986–2004 | IRA |
| Moussata et al., 200717 | 21 | France | Single | Cohort/Retrospective | / | IPAA and IRA |
| Ulas et al., 200674 | 1 | Turkey | Single | Case report | 1993 | IPAA - cancer |
| Campos et al., 200519 | 1 | Brazil | Single | Case report | / | IPAA - cancer |
| Groves et al., 200534 | 60 | UK | Single | Retrospective analysis of St. Mark’s Hospital Polyposis Registry | / | IPAA |
| Vroueraets et al., 200475 | 2 | The Netherlands | Single | Case report | 1990, 1991 | IPAA – cancer |
| Ooi et al., 200336 | 2 | Cleveland, USA | Single | Case report | / | IPAA – cancer |
| Church et al., 200338 | 197; 62 operated in pre-pouch period and 135 in pouch period starting in 1983 | Cleveland, USA | Single | Analysis of polyposis registry | 1950–1999 | IRA |
| Cherki et al., 200376 | 1 | France | Single | Case report | / | IPAA - cancer |
| Thompson-Fawcett et al., 200177 | 33 | Canada | Single | Cohort/Prospective | / | IPAA |
| Church et al., 200115 | 213 (165 IRA) | Cleveland, USA | Single | Analysis of polyposis registry | / | IRA and IPAA |
| Brown et al., 200178 | 1 | Singapore | Single | Case report | / | IPAA - cancer |
| Bertario et al., 200023 | 371 | Italy | Multicenter | Retrospective analysis of Hereditary tumor registry | 1955–1997 | IRA |
| Vuilleumier et al., 200079 | 1 | UK | Single | Case report | 1990 | IPAA - cancer |
| Jenner et al., 199821 | 55 | Australia | Single | Analysis of polyposis registry | ?–1994 | IRA |
| Bassuini et al., 199680 | 1 | UK | Single | Case report | 1991 | IPAA - cancer |
| Hoehner et al., 19946 | 1 | Iowa, USA | Single | Case report | / | IPAA - cancer |
FUP = follow up; IPAA = ileal pouch anal anastomosis; IRA = ileorectal anastomosis
Five studies described the rate of adenoma development in the residual rectum (Supplementary Table 1). In 8 studies that analysed the frequency of secondary proctectomy due to endoscopically unmanageable polyposis, the rate of proctectomy ranged from 3.7% to 35%.15 Five studies described adenoma evaluated in the neoterminal ileum (Table 3), with a high variance in reported rates from 0%16 to 47.6% in patients followed-up for median of > 20 years17 in one study including a paediatric cohort18, 2 patients required resection of the terminal ileum and construction of a new IRA, one due to low grade dysplasia (LGD) and one due to high grade dysplasia (HGD) adenoma.
Rate of adenoma development in the neoterminal ileum in patients after ileorectal anastomosis (IRA) and ileal pouch anal anastomosis (IPAA)
| First author and publication date (ref.) | Adenomas in the neoterminal ileum – after primary IPAA; n (%) | Cumulative risk for development of neoterminal adenomas | Years since surgery | Risk factor for adenomas in neoterminal ileum | Rate of adenomas in the neoterminal ileum – after primary IRA; n (%) | Years since surgery |
|---|---|---|---|---|---|---|
| Tajika et al., 201916 | 4/24 (16.7) | 4.4% at 20 years and 36% at 30 years after primary surgery | 23.1 ± 5.8 | 0/14 (0.0) | ||
| Boostrom et al., 201366 | 4/33 polyps (12.0) | |||||
| Pommaretet et al., 201335 | 9/118 (6.5) | Presence of pouch adenomas (OR, 2.16, P = 0.007) | ||||
| Booij et al., 201018 | 5/34 (14.7) 2 patients had resection of neo-terminal ileum, one due to LGD and other due to HGD adenoma. | |||||
| Gleeson et al., 200830 | 3/13 (23.1) | Median 6.5 (0–15) | 4/16 (25.0) | Median 12 (1–29) | ||
| Moussata et al., 200717 | Mean 17.6 +-7.8(6–35) Mean from colectomy to diagnosis: 16.4+-8.5 (5–30) | 10/21 (47.6) of which 2 were advanced adenomas. | ||||
| Groves et al., 200534 | 2/20 (10.0) | 6 (1–14) | 1/47 (2.0%) | 12 (0–39) | ||
| Thompson-Fawcett et al., 200177 | 1/24 (4.2) | Median 7 (1–19) |
HGD = high grade dysplasia; LGD = low grade dysplasia
The reported rate of cancer in the rectal remnant (Table 4) after primary IRA is 8.8%18 to 16.7%19 with a median follow-up from surgery19 of 91.1 months (3–557 months). However, studies from Japan report higher rates of up to 37%20, but this is due to the inclusion of in situ carcinoma in the cancer definition. The same study had the longest median follow-up of 21.1 years (3–35). On the other hand, a small cohort of 21 patients from France reported zero cases of cancer during a median follow-up of 8.4 years. Jenner et al.21 only included patients with a confirmed mutation. Five studies reported a cumulative incidence of rectal cancer ranging from 3%22 to 17.2%19 at 5 years, 7.7%23 to 24.1%19 at 10 years, 11%22 to 23%23 at 20 years, and 24%22 at 30 years after the primary IRA. In one of the largest studies24, which analysed data from 4 national registries and 776 patients, the 10-year cumulative risk of residual rectal cancer was 4.4% (95% CI, 2.6–6.2) for patients who underwent surgery before 1990 and only 2.5% (0–5.5) after the 1990. Only one study reported the time from surgery to cancer diagnosis (median 102 months [1–26 years])23; other studies reported follow-up time from surgery, but did not clearly define when follow-up started nor the surveillance regime. Five studies reported mortality ranging from 1.6%23 to 11.1%20 in which 3 out of 27 patients died from cancer in the rectal remnant. Only one of two studies that examined long-term survival after diagnosis of residual rectal cancer reported a 5-year survival rate of 55%.22 In a study from Japan, 5-year survival was 94%25, but the excellent survival was explained by the inclusion of carcinoma in situ despite the exact proportion of these was not given.
Patient characteristics and rate of rectal remnant cancer rate in patients after ileorectal anastomosis (IRA)
| First author and publication date | Proportion of man; n / (%) | APC mutation Underwent n/(%); Positive in; n/(%) | Follow-up (years/months) since surgery | Years since surgery to cancer diagnosis | Age at surgery | Age at cancer diagnosis | Rectal remnant cancer rate; n/(%) | Cumulative risk for rectal cancer | Rectal cancer mortality |
|---|---|---|---|---|---|---|---|---|---|
| Colletti et al., 202258 | 57.4% | 93.6% / | / | Median of 13 years | / | / | 47 / 715 (6.57) | / | 14/47 (29.8%) at median follow up of 13 years. |
| Pasquer et al., 202159 | 95 (48.2) | / | / | / | / | / | 12 / (6.1); 1 was metastatic, 2 were resected endoscopically, 10 surgically | / | / |
| Maehata et al., 201520 | 16 (59.3) |
| 21.1 (3–35) | / | Median 27 years (9–66) | / | 10/27 (37.0); 6/10 cancers were TisN0M0 | 8% at 10 years; 19% at 20 years; 57% at 30 years | 3/27 (11.1) |
| Koskenvuo et al., 201322 | 59 (42.1) | / | Median 15 years (0–44) | / | Mean 36 years (18–71) | Cumulative risk 2% at 40 years age; 7% at 50; 13% at 60 years age and 16 % at 70 years age. | 18/140 (13%) | 3% at 5 years; 4% at 10 years; 11% at 20 years; 24% at 30 years after IRA | 10/140 (7%); 5-year survival 55%. Cumulative risk for death due to rectal cancer after IRA: 2% at 5 years, 3% at 10 years and 9% at 30 years. |
| Booij et al., 201018 | 19 (44.2) | / | / | / | Median 16 (7–25) | / | 3/34 (8.8) | / | 2/34 (5.8) |
| Sinha et al., 201026 | 232 (54.3) |
| Median 15 years (7–25) | / | Median 21 years (11–67) | / | 48/427 (11.2%) | / | / |
| Yamaguchi et al., 200925 | 35 (59.3) | / | Median 8.9 years | / | Median 30 years (13–65) | / | 17/59 (30%) | / | 5-year survival 94%; 10-year survival 94%. |
| Nieuwenhuis et al., 200927 | / | / | / | / | / | / | / | 3.7% for group 1; 9.3% for group 2; 8.3% for group 3.% | / |
| Campos et al., 200819 | / | / | 91.1 (3–557) | / | Mean 45.8 years | Mean 50.6 years | 6/36 (16.7) | 17.2% at 5 years; 24.1% at 10 years; 43.1% after 15 years | / |
| Gleeson et al., 200830 | / | / | FUP initiated median 12 (1–29) years after surgery | / | / | 40 and 59 years. | 2/16 (12.5) | / | / |
| Bullow et al., 200824 | 401 (51.7) | / | Median 7 years (0–13). Patients were operated between 1950–2006 | Median 27 (7–75) | / | 60/776 (7.7%) (56/576; 10% and 4/200; 2%) | 10-year cumulative risk 4.4% [95% CI 2.6–6.2] in pre-pouch era; 10-year cumulative risk 2.5% [95% CI 0–5.5] in pouch era; | / | |
| Moussata et al., 200717 They only watched ileal muocas above the IRA | 10 (47.6) |
| Mean 8.4 years ± 5 since colectomy | / | / | / | 0/21 (0.0) | / | / |
| Church et al., 200338 | 92 / (46.7) | / | Pre-pouch era: 212 months (IQR 148 months); Pouch era: 60 months (IQR 80 months) | / | Median age 23 years (IQR 15.5 years pre-pouch and 17 years pouch) | / | 8 (12.9%) in the pre-pouch era and 0 in pouch era. | / | / |
| Bertario et al., 200023 | 206/371 (55.5) |
| Median 81 months | Median 102 months (1–26 years) | Mean 32 years | / | 27/371 (7.3) |
| 6/371 (1.6) |
| Jenner et al., 199821 | 25/55 (45.0) | 55/(100.0) | Median 10 (1–31) | / | Mean age 30 (13–62) | Median 41 | 7/55 (12.7) | / | / |
Colonic phenotype divided in 3 groups: (Group 1 - <100 polyps and mutation in codons 1–157, 312–412 and 1596–2843; Group 2 Hundred of polyps and mutation in codons 158–311, 413–1249 and 1465–1595; Group 3 Thousand of polyps and mutation in codon 125
APC = adenomatous polyposis coli; FUP = follow up
Eleven studies reported nine risk factors predictive of the progressive rectal residual phenotype (Supplementary Table 2). Four studies analysed the genotype-phenotype relationship; The presence of a pathogenic variant between codons 1250–1464 was an independent risk factor for subsequent cancer development (HR 4.4 [1.3–15.0]23 and for the secondary proctectomy26,27 (HR 3.91 [1.45–10.51], P = 0.007). In a small study of 25 patients, all patients (n = 3) with carpeting rectal remnant polyposis had a pathogenic variant in codon 1309, but this was only descriptive data.28 An aggressive colonic phenotype with at least 500 polyps at time for surgery was identified as a risk factor in three studies (Supplementary Table 2). Two studies15,25 have identified > 20 rectal remnant polyps at the time of surgery or during the endoscopic surveillance26 as an independent risk factor for secondary proctectomy (HR 30.99 [9.57–100.32] P < 0.001), while in one study a cut-off of > 10 rectal adenomas28 was associated with a more aggressive phenotype, as these patients developed a mean of 9.29 rectal residual adenomas per patient per year compared with 0.67 adenomas per patient per year if they had < 5 rectal polyps at the time of surgery. Other potential risk factors included patient age at diagnosis of rectal residual cancer, time since surgery, presence of congenital hypertrophy of the retinal pigment epithelium, and presence of colon cancer at the time of primary surgery. APC site mutation, preoperative colon phenotype, presence of duodenal adenomas and rectal remnant phenotype on surveillance were not identified as risk factors for progressive rectal remnant disease phenotype only in one study.20
Seventeen studies (Table 5) reported on the development of adenomas after IPAA, of which eight studies differentiated between the pouch body and the anastomosis, one study only reported the anastomotic adenoma rate, while in the remaining seven studies the authors did not precisely define the anatomical location of the adenomas. The median age of patients at the time of surgery ranged from 15.4 to 34.6 years, with a median follow-up from surgery of 5.4 years to a median of 21.6 years. The reported rate of adenoma in the pouch body ranged from 9.4%29 to 76.9%.30 The proportion of HGD histology among adenomas at the polyp level ranged from 5.9%17 to 53.2%.31 In one study, the proportion of advanced adenomas on a per-patient basis was 11.2%.31 The cumulative risk of adenoma development after primary IPAA was 12% and 58% at 5 and 20 years after the surgery respectively.16 According to the analysis from Poland32, 50% of all patients would develop LGD 15 years after the surgery, while HGD is estimated to be present in half of the patients 17.5 years after the surgery. Six studies analysed the rate of adenoma development in the neo terminal ileum, the proportion of patients with histologically confirmed adenoma varied from 4.2%33 to 23.1%30 with at a median follow-up from surgery of 6.534 to 23.1 years.16 The cumulative risk of developing an adenoma in the neo terminal ileum was 4.4% at 20 years and increased to 36% at 30 years after the surgery as reported in the same study. The presence of pouch body adenomas was the only independent risk factor for the neo terminal ileum adenomas (OR, 2.16, P = 0.007).35
Patient characteristics and rate of adenomas in patients after primary ileal pouch anal anastomosis (IPAA)
| First author and publication date | Sex (man); n (%) | APC mutation Underwent; n (%); Positive in; n (%) | Distinguish between pouch body and rectal cuff | Follow-up (months/years) | Time from surgery to first adenomas (years) | Age at surgery (years) | Rate of adenomas (≥ 1 polyp) | Size of adenomas, mm | Histology of adenomas; n (%) | Number of Adenomas |
|---|---|---|---|---|---|---|---|---|---|---|
| Aelvoet AS et al., 202355 | 81 (56) |
| Yes | Median 152 (77–240) | 15% at 5 years; 48% at 10 years; 85% at 20 years. | Median 24 (18–32) | Median 5 (3–15) | Tubular adenomas 31 (28%), Tubulovillous 26 (23%), Villous 5 (5%) | Prepouch ileum 4(2–13), Pouch body 20 (5–50), rectal cuff 6 (3–10) | |
| Tajika et al., 201926 | 16 (47.1) | / | Yes | Median 21.6 (3.7–8.8) | 32 (35.9) of patients showed progression of pouch adenomas during FUP | Median 34.6 (17–52) | 24/34 (70.6) | 2–40 mm | 6 advanced adenomas (25.0) | 1–300 |
| Ganschow et al., 201861 | 100 (52.1) |
| No | Median 12.8 (9–17) for patients with pouch adenomas and (2.5–12.2) for patients without pouch adenomas; | 32 (35.9) of patients showed progression of pouch adenomas during FUP | 27.5 years (10.2–58.5) | 90/192 (46.9) at a median of 8.5 years (0.9–25.1) after IPAA. 5 years after IPAA 84.9% patients free of adenoma; 15 years after 40.4% and 20 years after 21.9% patients were free of adenomas. | 53/192 (58.9) ≤ 4 mm; 24/192 (26.7) 5 – 10 mm; 13/192 (14.4) ≥ 10 mm | Tubular adenomas in 69/192 (76.7); tubulovillous adenomas in 16/192 (17.8); villous in 5/192 (5.6) | 46/192 (51.1) had < 4; 14/192 (15.6) 5–10; 30/192 (33.3) > 10 adenomas |
| Goldstein et al., 201563 | 24 (41.0) | Yes | Mean 11.6 years +-14.6 years | Median adenoma free time interval since surgery; Cuff 10.8 years Pouch 16.9 years | Mean 30.8 years +-10.8 years | 35/59 (59.0);
| / | All LGD | / | |
| Zahid et al., 201519 | 14 (51.8) | No | Mean 9.2 years | Median; 72 months (18–249) | Median 31 years (14–65) | 12/27 (44.0) | / | Only 1 polyp HGD (< 99%) | / | |
| Kennedy et al., 201466 | 43 (45.0) | Watched only anastomosis | Mean 7.6 (0 – 24) | Mean 15.4 (4–20) | 9/95 (9.4) | |||||
| Pommaretet et al., 201336 |
| / | Median 15 years | 25 years (9−61 years) | 57/118 (48.3) | > 10 mm:12 | 94% LGD; 6% HGD |
| ||
| Boostrom et al., 201366 | 52 (44.5) | Yes | 125 months (25–423 months) | 12.4 years (15–405 months) | 26 years (4–60 years) | 30/117 (25.6) | 5.9 mm (2 mm to 20 mm) | 22 LGD, 8 tubulovillous | / | |
| Wasmuth et al., 201367 | 34 (55.7) | / | Yes (body and anastomosis) | Cumulative rate of adenomas at 28 years 17% for mucosectomy group and 75% at 15 years in a group without mucosectomy (P < 0.0001) | 20 (10–49) |
| ||||
| Tonelli et al., 201251 | / | 45 (65%) | No | Median 133 months (12–288 months) | Mean 7 years (1–15 years) | 33 years (17–63 years) | 25 (36.0) | Mean 3 mm (1–40) | Adenomas, dysplasia not specified | Mean 8 (1–47) |
| Yan et al., 201268 | 30 (71.5) | / | Yes | Median 7.2 (2.2–20) | 29 (16–65) | At the anastomosis 6/33 (18.2) | / | / | / | |
| Banasiewicz et al., 201133 | 79 (47.9) | / | / | Endoscopies performed 2–19 years since surgery. |
| 21/165 (12.7) | LGD - 21/32 (65.6); HGD - 11/32 (34.4) | |||
| Gleeson et al., 200831 | / | / | Yes | / | FUP began median 6.5 (0–15) after surgery | / | 13/13 (100): 10/13 pouch body; 2/13 anastomosis; 3/13 ileum above anastomosis | < 5 mm | / | 5–30 |
| Friederich et al., 200832 | 119 (56.0) | / | / | Mean 7.9 (0.4–20.3 years) |
| Mean 30.0 years (10–62.6 years) | 47/212 (35%) | / | / | / |
| Campos et al., 200817 | / | / | No | 50.8 (5–228) | 3/26 (11.5) | |||||
| Moussata et al., 200725 | 12 (57.1) |
| Yes (only polyps in the ileal mucosa of the pouch body are described) | Mean 5.4 +- 2.6 (1–11) | Mean 4.7+-3.3 years (1–14) | 17/23 (74.0) | Mean size 5.2 mm +-3.4 mm; 3 polyps were > 10 mm. | LGD 16/17 (94.1); HGD 1/17 (5.9) | / | |
| Groves et al., 200535 | 35 (58.3) | / | Between pouch and above anastomosis ileum | 6 years (1–17 years) | / | 32.5 years (13–66 years) | 34/60 (57%) of which 5 were > 10 mm / 11 were advance adenomas | Mean size 5 mm (1–40 mm) | / | Median number 4 |
| Thompson-Fawcett et al., 200177 | / |
| Only pouch body | / | / | / | 20/33 (60.0) adenomas | 1–3 mm | / | Median 10 (1–100) Also lymphoid hyperplasia included |
APC = adenomatous polyposis coli; FUP = follow up; HGD = high grade dysplasia; IRA = ileorectal anastomosis; LGD = low grade dysplasia
Since the first case report of cancer arising in the ileal pouch of a FAP patient in 19946, we have identified 45 (Table 6) cancers that have developed in FAP patients after primary IPAA. Of these, 30 were located in the pouch body and 15 in the anastomosis/rectal cuff. The time from surgery to cancer diagnosis was reported for 22 patients and ranged from 2.336 to 33 years.37 The information about the interval since last follow-up was reported for only 15 patients. The shortest interval between normal endoscopic surveillance and cancer diagnosis was 9 months.16 Of the studies that reported the final outcome, 13 (28.9%) patients were alive at the last follow-up (range 8 months to 6 years) after surgical therapy and 9 patients died of disseminated cancer (1 month to 4 years after diagnosis), most despite an initial R0 resection.
Cancer rate after primary ileal pouch anal anastomosis (IPAA)
| First author and publication date (ref.) | No of patients | Age at cancer diagnosis (years) | Time to cancer (years) | Interval since last surveillance endoscopy and findings | Endoscopic findings at diagnosis | Location | Staging of cancer and status |
|---|---|---|---|---|---|---|---|
| Aelvoet et al., 202355 | 3/111 (2.7%) | / | / | / | / | / | Pouch excision |
| Pasquer et al., 202158 | 1/92 (1.1) | 30 | / | 1 month | Pouch body | Endoscopic resection | |
| Ganschow et al., 201861 | 1 | / | 27 | / | / | Pouch body | Resection and reconstruction of a new pouch - alive |
| Walsh et al., 201663 | 1 | 54 | / | Regular annual surveillance | New endoscopy due to anemia and rectal blood loos | Anastomosis | T3N2Mx, resection and ileostomy, alive during last FUP. |
| Wasmuth et al., 201367 | 1 | / | 11 | / | / | Rectal cuff | Resection and ileostomy - alive |
| Boostrom et al., 201366 | 1 | / | 23.7 | / | / | Pouch body | Transanal resection - alive |
| Ozdemir et al., 201338 | 4 | / |
| Regular annual surveillance | / | All ATZ |
|
| Makni et al., 201269 | 1 | 26 | 10 | 8 months | Polyps, LGD? | Pouch body? | Pouch excision – died 12 months later dissemination |
| Tonelli et al., 201251 | 2 |
| 10 |
|
|
| Excision with ileostomy, T3N0M0, died 6 months later dissemination/Excision with ileostomy, T2N0M0, alive after 56 month FUP |
| voon Roon et al., 201170 | 1 | / | 13 | / | / | Pouch body | Excision of a pouch – died 2 years of disseminated disease |
| Banasiewicz et al., 201133 | 5 | / | / | / | / | Pouch body | / |
| Ault et al., 200971 | 2 |
|
|
| Pain and blood per rectum, 3 cm mass/Sacral pain, bleeding ulcer | Pouch body / Pouch body | T2N1Mx, died of AMI prior treatment / Metastatic disease, chemotherapy |
| Tajika et al., 200983 | 2 |
|
|
| 30×25 mm cancer / Polyposis and 25 × 25 mm polyp | Pouch body/Kock’s pouch body |
|
| Lee et al., 200871 | 1 | / | 7 | / | Ulcerating tumor | Pouch body | T4N1M0, APR ileostomy. Developed metastases 2 years later. |
| Friederich et al., 200832 | 4 |
|
|
| / | All pouch body |
|
| Linehan et al., 200772 | 1 | 40 | 10 | / | Pelvic pain, discharge | Pouch body (patient had ileostomy but pouch was left in situ) | Excision. At last FUP patient was well. |
| Ulas et al., 200674 | 1 | / | 9 | / | / | Anastomosis | Dukes B, APR, metachronous cancer after 1 year |
| Campos et al., 200519 | 1 | / | 12 | No FUP | Presented with rectal bleeding | Pouch body | T2N0Mx, APR and ileostomy, patient well at 6 years FUP. |
| Vroueraets et al., 200475 | 2 |
|
| 5 years normal, then 2 and 1 years (both multiple LGD adenomas refused surgery) / Regular FUP every 2 years | Presented after 1 year with rectal bleeding /Normal. Routine biopsies at subsequent FUP revealed adenoca. |
| T2N0M0, APR, alive 1 year later / T4N0M0, APR, alive 8 months later |
| Cherki et al., 200376 | 1 | 35 | 3.5 | 1.5 years | / | Pouch body | T3N1M1, resection with ileostomy, died 1 month later |
| Ooi et al., 200336 | 2 |
|
|
|
|
| T3NOMO, APR, ileostomy, died 2.5 years later dissemination / T2N0M0, transanal excision with ileostomy (refused APR), died 4 years later, dissemination |
| Brown et al., 200178 | 1 | 44 | 7 years 4 months | Under FUP every 6 months | / | Anastomosis | / |
| Vuilleumier et al., 200079 | 1 | 38 | 7 | No FUP | / | Anastomosis | Resection with ileostomy – died 12 months later dissemination |
| Palkar et al., 199715 | 1 | 39 | 4.7 | 3 months | ? | Pouch body | T4NOM? - alive |
| Kim et al., 199715 | 1 | / | / | / | / | Pouch body? | / |
| Bassuini et al., 199680 | 1 | 31 | 3 | No FUP | / | Pouch body | / |
| Von Herbay et al., 199614 | 1 | 33 | 8 | Anastomosis | T1N0M0 | ||
| Hoehner et al., 19947 | 1 | 34 | 20 | / | / | Anastomosis | / |
The data from these cases has been drawn from reviews by Tajika and Smith as full-text of the papers were not accessible.
FUP = follow up; HGD = high grade dysplasia; LGD = low grade dysplasia
Six studies (Supplementary Table 3) compared the rates of adenoma development at the anastomosis between hand-sewn and stapled techniques. The incidence of adenoma was lower for hand-sewn anastomosis, ranging from 0 to 33%, and for stapled anastomosis, ranging from 33.9 to 57%. The 10-year cumulative risk of adenoma development is 20–22.6% for hand-sewn anastomosis and 51.1–64% for stapled anastomosis.
Nine studies analysed risk factors for adenoma development (Supplementary Table 4). None of the seven studies found a genotype-phenotype association. There was no association between colon adenoma burden at the time of surgery and subsequent development of pouch adenomas in three out of four studies. In the only positive study, none of the patients with < 200 colon polyps developed pouch adenomas, whereas almost half of the patients with > 1000 colon polyps later developed later pouch adenomas. Three studies have identified age of the pouch as a risk factor, while three others found no association between time since surgery and the rate of pouch adenomas. An association between the Spigelman score and the development of pouch adenomas was not confirmed. One study identified the presence of gastric adenomas as an independent risk factor for the development of pouch adenomas.
Using a systematic approach, we identified a wide range of reported adenoma and cancer rates in the rectal remnant, pouch body, at IPAA and in the neoterminal ileum. The wide range in adenoma rates is probably partly due to the wide range of included studies in terms of year of publication. The equipment and quality of optical diagnosis has improved considerably in recent years, allowing better detection of adenomas and more precise examination of the pouch and rectal remnants. In addition, the risk stratification of patients at the time of surgery has also improved, allowing patients with a more aggressive phenotype to undergo primary restorative proctocolectomy while primary IRA can still be offered to patients with an attenuated phenotype or low rectal disease burden. Indeed, in the largest study of four European national polyposis registries, the cumulative risk of cancer in the rectal remnant (CRR) was 10% in patients operated in the ‘pre-pouch’ period and only 2% in those who were operated in the ‘pouch period.24 Similar findings have been reported from the USA38 where 8 patients operated before 1983 (12.9%) were diagnosed with CRR compared to none of those operated after 1983 when pouch surgery was introduced at the Cleveland Clinic. Recently published data from two Japanese studies reporting an overall CRR rate of 30%25 – 37%20 must be interpreted with caution as carcinoma in situ was also included in the definition of cancer in their cohorts. The risk of metachronous cancer after IRA has been recognised early and these patients have been advised to undergo regular surveillance of the rectal remnant. Traditionally, surveillance was recommended every 3 to 12 months. This recommendation has been maintained ever since and can be also found in the recently published international guidelines (Table 1). The French national guidelines published in 200539 are the only ones to include the genotype information, as they recommend more frequent surveillance if the pathogenic variant is located between codons 1250–1500. However, they were published in 2005.
The main obstacle to refining recommendations for endoscopic surveillance is the lack of high-quality, prospective data. Unfortunately, we have not found a single randomised trial that has compared different surveillance strategies or aimed to identify factors that would allow risk stratification. Members of the International Society for Gastrointestinal Hereditary Tumors (InSiGHT)40 proposed a staging system41 and stage-specific interventions for patients with intact colon and those with IRA, but unfortunately no effort has been made to validate this staging system. Data on endoscopic treatment modalities are even more descriptive. In fact, in five international recommendations (Table 1), only Vasen et al.7 recommended endoscopic removal of all polyps with dysplasia or those larger than 5 mm. Endoscopic management of these patients has therefore been influenced by expert groups. Unfortunately, preferred methods of endoscopic management were rarely described in the reviewed studies. Maehata et al.20 recommend removal of all polyps larger than 8 mm. A descriptive study with a small sample size (n = 6)42 showed that large-scale cold snare polypectomy can effectively reduce the polyp burden in the rectal remnant even in cases of very high polyp numbers. The mean number of polyps removed was 78.5 (30–155). During the follow-up (mean 10.7 months), none of the patients developed rectal cancer and there were no complications related to polypectomy. This is in contrast to another study from the USA30, which advocates the use of ablative therapy with argon plasma coagulation. A similar practice was supported by a study published in France in 2007.17 National French guidelines published in 2005 recommend ablation with APC for small polyps (a few millimetres) and mucosectomy for larger polyps.39
Improvements in endoscopic resection techniques have also been applied to the treatment of large lesions in the rectal remnant. Recently two reports, both from Japan43,44, have been published of successful endoscopic submucosal dissection (ESD) of 75 mm Is + IIa adenoma and residual adenoma at the IRA. In our endoscopy unit (Hospital Clinic, Barcelona) we also perform advanced endoscopic resection techniques. Figure 2 (A and B) shows a recent endoscopic mucosal resection (EMR) of an 18mm laterally spreading tumour granular type (LST-G) in the rectal remnant of a patient with FAP.
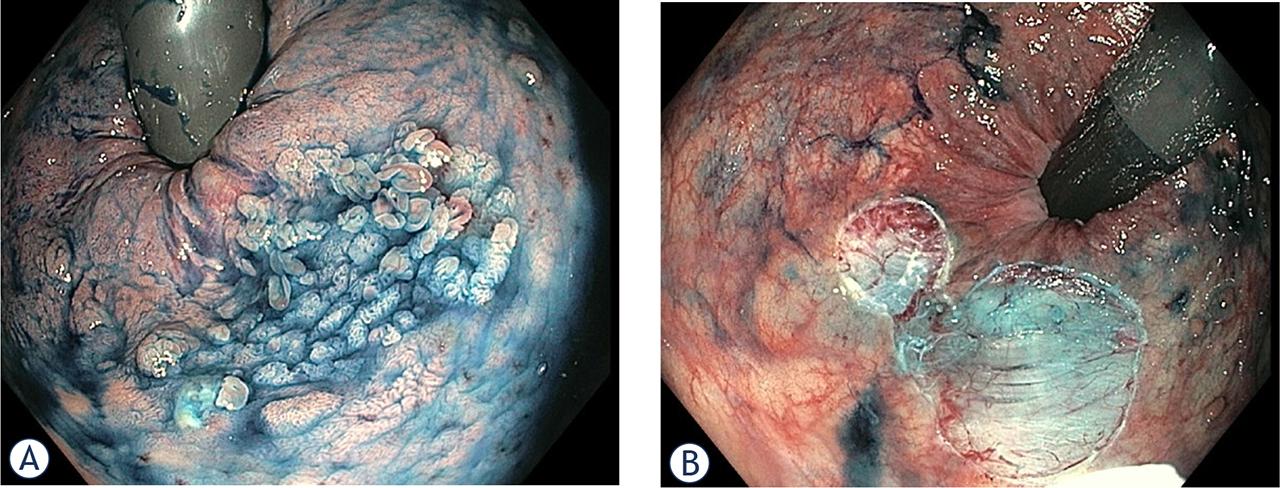
Surveillance endoscopy in a 48-year old patient with FAP after colectomy with IRA revealed 18 m LST-G (A). After submucosal injection with gelofusine, indigo carmine and adrenaline, piecemeal endoscopic mucosal resection (pEMR) (B) was performed.
There is little data on the use of advanced imaging techniques. The study from St. Mark’s hospital in London45 showed no benefit of dye-based chromoendoscopy to detect additional adenomas in the rectal remnant. The European Society of Gastrointestinal Endoscopy (ESGE) guidelines46 published in 2014 did not recommend the use of advanced endoscopic imaging in patients with FAP, but did not specifically differentiate between the patients with intact colon and those after surgery. On the other hand, the French Society of Endoscopy39 recommended the use of dye-based chromoendoscopy with indigo carmine. We believe that use of dye-based chromoendoscopy in these patients does not increase the detection of clinically relevant lesions and it is not routinely performed in our unit. Considering the data on a cumulative risk of 57% for CRR 30 years after surgery20 and the fact that adenoma development in the rectal remnant is an inevitable event16, regular endoscopic surveillance is mandatory. Our recommendations are in line with other guidelines and our patients are recommended annual endoscopic surveillance, despite alarming data from an early study published in 20015 from four European registries in which 75% of patients with CRR had a negative rectoscopy within 12 months and 35% within 6 months prior to diagnosis of CRR. There was no information on the endoscopy equipment used for surveillance. We believe that the high rates of negative rectoscopies prior to cancer diagnosis may – to some extent - be influenced by the quality of endoscopy, which has been limited by the technical aspects of the equipment used in the past. This problem needs to be addressed again in the light of developments in endoscopic equipment.
When restorative proctocolectomy with IPAA was first described in 197847, it was believed that this operation would eliminate the risk of colorectal cancer in patients with FAP. However, a few years later, as the first pouches began to age, case reports of cancers arising in the pouch began to appear in the literature.6 Since then, reports have become more frequent and we have identified 45 cases of cancer after primary IPAA, of which 26 arose in the ileal mucosa of the pouch body and 15 at the anastomosis. Furthermore, we now know that cancer can develop even after mucosectomy down to the dentate line48, because even after removal of all visible rectal mucosa, some microscopic rectal columnar epithelium remains at the ATZ.49 In the study from the Heidelberg Polyposis Registry with 100 patients50, rectal residual mucosa (defined as visible mucosa or detected by histology from blinded biopsies) was found in 42 (84%) cases after stapled and in 21 (42%) cases after hand-sewn anastomosis.
Researchers from Japan16 found a 70% incidence of adenomas in the pouch body with one of the longest follow-up periods reported to date (> 20 years). Similarly, in a study from France, 74% of patients had at least one adenoma in the pouch, but with a mean follow-up of only 5.4 years. In contrast, one study found that isolated rectal cuff adenomas were more common than isolated pouch adenomas (49.1% vs. 6.8%), while 18.7% of patients had both pouch and rectal cuff adenomas. Cumulative 5-year, 10-year and 20-year risks for pouch adenomas were 32%, 52% and 68% in the Japanese study16, a slightly lower 5-year cumulative risk but a similarly high 10-year risk was observed in a Dutch study31; 16% and 42%, but the authors of this paper did not specifically define the exact location of the adenomas. The authors also reported a 10-year cumulative risk of developing precancerous adenomas of 12.8%.
On the other hand, the adenoma rates – at least in the stapled group - seem to be higher in the studies that only looked at the anastomosis and compared hand-sewn with stapled: 0–33% vs. 33.9–57%. In view of these figures, it is essential that patients with primary IPAA also undergo regular endoscopic surveillance. Particular attention should be paid to the rectal cuff and anastomosis, and the pouch should be examined in both forward and retroflexed position.
International guidelines most commonly recommend annual endoscopy examination, whereas ASCO guidelines9 advocate ‘case-by-case’ interval allocation. In 11 of only 12 studies that described a surveillance protocol, an interval of 12 months was recommended except in Brazil where endoscopy of the pouch was recommended every 2 years.
Interestingly, in the Netherlands pouch endoscopy was recommended every 1 to 3 years in the late 1990s but in 2001 the protocol was changed to annual endoscopic surveillance regardless of the anastomotic technique (hand-sewn or stapled).
One of the main concerns is the short interval (< 1 year) between the last normal endoscopy and the cancer diagnosis and the aggressive course of the disease despite an initial R0 resection (Supplementary Table 4). It is not entirely clear whether the adenoma-carcinoma sequence is faster in the ileal mucosa compared with the colon and rectum, or whether “negative” endoscopies prior to cancer diagnosis could be explained by the poor quality of pouch endoscopy. Chromoendoscopy improves the detection of diminutive adenomas31 and lymphoid hyperplastic nodules45, but its use is discouraged33,35 for the same reasons as in the examination of rectal remnants – increased of detection of clinically irrelevant polyps. Endoscopy should be performed with a gastroscope or paediatric colonoscope, as stricture can occur at the anastomosis, especially after hand suturing.
There are no official recommendations for endoscopic management of FAP patients after IPAA. We have found considerable heterogeneity in local practice. Italian authors recommend resection of all adenomas > 3 mm.51 On the contrary, ablation with argon plasma coagulation is the preferred resection technique in a French study.17 Ablative techniques were also supported by the study from the Mayo Clinic.30 In a small descriptive cohort of only 5 patients42, large-scale cold snare polypectomy with a mean of 110.6 (30–342) resected polyps demonstrated the efficacy of cold snare in controlling large polyp burden (> 30 polyps) with no reported polypectomy related complication. In our unit we do not use nor encourage use of argon plasma coagulation. We recommend resection of all polyps > 3 mm. Advanced resection techniques, when performed in the tertiary centres, may be a viable alternative prior to surgical resection. A case report of successful en bloc ESD of a 15 mm ‘non-lifting’ HGD adenoma in the ileal pouch has recently been published.52 Figure 3 (A and B) shows an EMR of 25 mm LST in a patient with FAP after IPAA. The polyp was located in the rectal cuff and extended from the anastomosis to the dentate line. The procedure was performed at our Endoscopy Unit. It should be emphasised that the wall of the ileum is very thin and special care must be taken when resecting larger lesions.
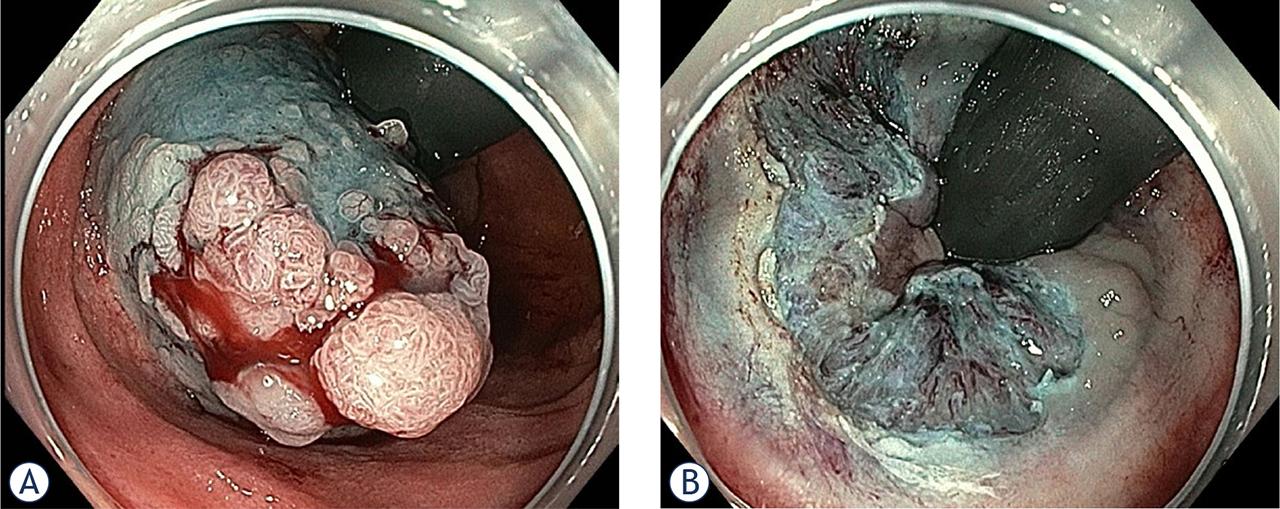
Surveillance endoscopy in a 49-year old patient with FAP after proctocolectomy with IPAA revealed 25 mm LST-G mixed type lesion in the rectal cuff. Lesion was spreading from the anastomosis to the dentate line. Patient had undergone surgery five years earlier and did not show up for endoscopy follow-up since then (A). Lesion was removed with pEMR (B).
Although there is no randomised trial comparing different endoscopic surveillance intervals, it is unlikely that prospective data will be available in the future. The main reason is ethical issue, as these patients are at increased risk of colorectal cancer. However, with the introduction of high quality colonoscopy and improvements in endoscopy technique, a ‘negative’ endoscopy before cancer diagnosis should become highly unlikely if not impossible.