The shortage of conventional energy resources and the continuous use of electricity have led to an increase in energy demand in the world [1,2]. Therefore, many scientists have directed their research to developing efficient devices for energy storage. Supercapacitors and batteries, which are based on electrochemical energy storage (EES) devices, are important and ideal for domestic and industrial applications. For delivering high energy and power density [3,4], the active materials play an important role in developing EES devices to face the existing challenges. The solutions for these challenges can be discovered by extensive research for constructing novel nanostructures and nanocomposites for energy storage applications. The high power density of pseudocapacitive electrodes that are based on two-dimensional (2D) nanomaterials and transition metal oxides or hydroxides [5,6,7,8,9] has attracted the attention for these kinds of nanostructures. Particularly, these nanostructures showed long cycle life with fast charging and discharging in addition to excellent reversibility. Similarly, copper and zinc showed positive effects to act as efficient electrode materials to overcome the current poor properties of the existing electrodes. The main reasons for using zinc as an electrode are due to its high theoretical capacity (823 mA h g−1) and the strong stability of zinc in water [10,11]. Mattarozzi et al. showed that copper is an excellent electrode material for the cathodic reduction of nitrate [12]. In addition, copper has different advantages such as excellent conductivity, nontoxicity, abundance, and low cost [13]. Therefore, several studies focused on copper and its alloys; for example, copper–tin [14], copper–nickel [15], copper–silver [16], and copper–rhodium [17]. Similarly, CuO/ZnO nanocomposites were prepared by Wu et al. [18] to fabricate electrodes with a specific capacitance of 579 F g−1. Also, after 2,000 cycles, at a current density of 5 A g−1, it showed capacitance retention of 83%. In the case of using silicon oxide, similar results were observed by Kandhasamy et al. [19] in the SiO2/CuO electrodes. With a specific capacitance of 423 F g−1, they achieved a capacitance retention of 86% after 3,000 continuous cycles at a current density of 4 mA g−1. Through doping with zinc sulfide, the Cu–ZnS electrodes were synthesized by Hussain et al. [20] to deliver a specific capacitance of 468 F g−1 at 1 A g−1.
In this trend, the devices for energy storage can be developed by increasing the efficiency of the Cu-based materials. Several structures of copper exist in nature with different morphologies. For example, tribasic copper chlorides (Cu2(OH)3Cl) are a group of minerals, which attracted attention because they possess several polymorphs, i.e., clinoatacamite [21], botallackite [22], and atacamite [23]. In addition, the metastable paratacamite structure was included in this group by the study of Sahoo et al. [24]. Sahoo and his colleagues have used atacamite, which is a stable form of paratacamite, for bio-imaging and photothermal applications through silica encapsulation. Also, Haro et al. [23] demonstrated that the atacamite system with solar radiation yielded efficient bacterial inactivation. Similarly, the structures of paratacamite Cu2(OH)3Cl were stabilized by doping with appropriate quantities of Ni or Zn [25–29]. Ni-doping could well stabilize the Cu2(OH)3Cl paratacamite phase at ambient temperature by Liu et al. [29] to increase its photocatalytic activity. In this study, for the first time, the combination with titanium was used to stabilize the metastable paratacamite phase.
In addition, copper, zinc, and titanium were employed to design three different nanostructures and morphologies. Copper was used to produce a mono-metal layered structure, Cu-HDS. For the first time, titanium was used to create a dual-metal structure based on the Cu–Ti paratacamite phase. This combination caused stabilization of the metastable paratacamite phase. In the third structure, Zn and Ti were used to substitute copper in the Cu2(OH)3Cl lattice for engineering triple-metal structures based on the paratacamite and Zn–Ti LDHs. These different structures were characterized by different techniques. These structures introduce a novel, reliable, and economical route for fabricating supercapacitor electrodes. To the best of our knowledge, the construction of paratacamite containing Cu–Zn–Ti triple-metal nanoparticles as electrodes for supercapacitors has not yet been reported. The current study highlights a facile method to rationally integrate the advantages of three individual metals to deliver enhanced performance through synergistic effects for the enhanced utilization of tri-metallic materials in energy storage devices.
The mono-metal nanolayered structure was prepared by reacting two species of copper salts to produce copper hydroxyl double salts (Cu-HDS). One mole of copper monoxide was combined with 1.25 mol of copper nitrate in 1 L of water under a continuous flow of argon gas. This combination was carried out at 80°C for 24 h. After filtration and washing, the product was kept at 25°C under vacuum to obtain fine particles of greenish-blue powder.
Copper and titanium salts were precipitated during the thermal decomposition of urea to construct dual-metal nanostructures. Typically, an aqueous solution of copper(
Triple-metal structures based on copper, zinc, and titanium were produced by urea hydrolysis. Typically, three aqueous solutions of copper(
By imaging the prepared samples, JEOL-JSM-6330F scanning electron microscopy (SEM) was used for determining their morphologies. The samples were analyzed by the X-ray diffraction (XRD) technique using Rigaku, RINT 2200. The chemical structure of the prepared samples was characterized by energy dispersive X-ray spectroscopy with an electron probe micro-analyzer JEOL-JED 2300. The thermogravimetric analysis and differential thermal analysis were used to analyze the thermal behavior of the samples using a Seiko SSC 5200 apparatus.
The electrochemical attributes of the electrodes were evaluated using a three-electrode setup, comprising a platinum sheet and an Ag/AgCl electrode serving as the counter and reference electrodes, respectively. In the process of constructing the working electrode, a mixture was created by blending 80% Cu-HDS, CT-1, and CZT-3, 10% activated carbon (AC), and 10% polyvinylidene fluoride with anhydrous 1-methyl-2-pyrrolidinone. This blend was thoroughly mixed at room temperature using a magnetic stirrer. Subsequently, this homogeneous slurry was applied to a nickel foam substrate, which had been previously cleaned, covering an area of 1 × 1 cm with a uniform mass loading of approximately 1 mg. The coated substrate was then dried in an oven set at 90°C for 12 h. To assess the electrodes’ electrochemical behavior, all tests were performed in a 2 M KOH aqueous electrolyte, utilizing both cyclic voltammetry (CV) and chargedischarge (CD) techniques for comprehensive analysis.
Powder XRD was used to identify the crystalline structure of the prepared sample Cu-HDS, as shown in Figure 1a. The XRD pattern of C-HDS showed the characteristic peaks of copper hydroxyl nitrate (Cu4(OH)6(NO3)2), agreeing with JCPDS 77-0148. Also, Figure 1a displays symmetrical and strong peaks at low 2θ angles of 12.87° and 25.86°. At the same time, asymmetrical and weak peaks were observed at high 2θ angles of 33.7°, 35.74°, 36.57°, 40.0°, 42.08°, 43.75°, and 49.36°. These peaks were in agreement with the d-spacings of 0.69, 0.344, 0.27, 0.25, 0.225, 0.215, 0.207, and 0.15 nm, respectively. In addition, these peaks are attributed to the reflections of the planes [001], [002], [120], [201], [121], [003], [013], [103], and [300], respectively. The arrangement of the reflections of the planes [001], [002], and [003] indicated that copper hydroxyl nitrate has a layered structure with an interlayered spacing of 0.69 nm, where the d 001 value (0.69 nm) equals the double value of d 002 (2 × 0.344 nm) and is equivalent to the triple value of d 003 (3 × 0.225 nm). This indicates that the copper hydroxyl nitrate crystals consist of nanolayers of copper linked by hydrogen bonding between the NO3 group and hydroxyl groups.
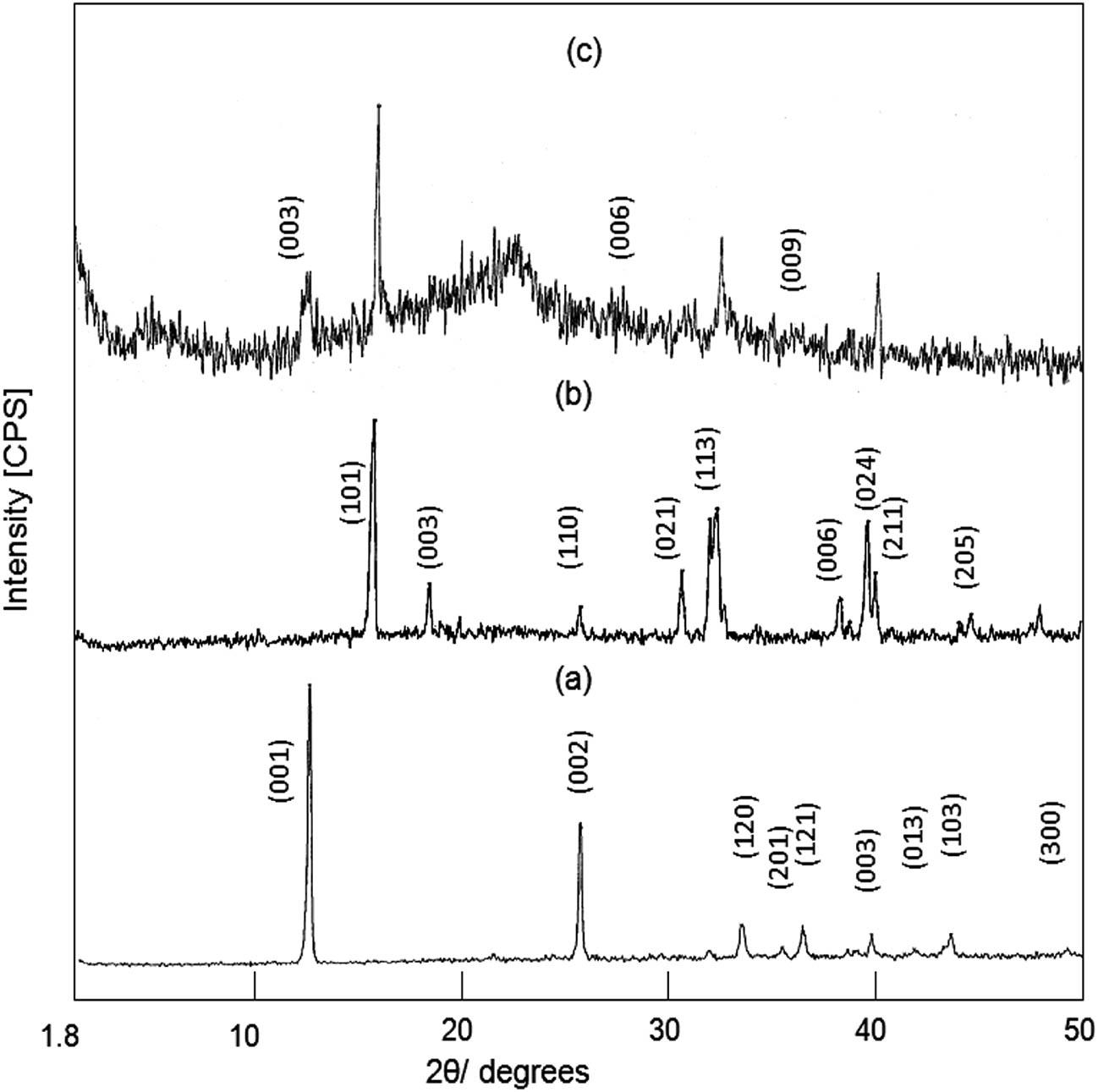
XRD patterns of (a) Cu-HDS, (b) CT-1, and (c) CZT-3.
By substituting titanium inside the crystals of copper, all the peaks of HDS disappeared, and new peaks were observed in the XRD pattern of the sample CT-1. Figure 1b exhibits peaks at 2Θ = 16.3°, 19.1°, 26.07°, 30.85°, 32.52°, 40.11°, and 44.85°. These peaks are due to the reflections of the planes [101], [003], [110], [021], [113], [006], [024], [211], and [205] and agree with the JCPDS files Nos 50-1560 and 50-1558. This indicates that the sample CT-1 has a new phase of paratacamite. Paratacamite and its congeners are intermediate phases in a solid-solution series extending between Cu4Cl2(OH)6 and Cu3MCl2(OH)6, where M is a cation with an ionic radius similar to that of copper. The ionic radii of titanium and copper are nearly similar (74 pm and 71 pm, respectively). This indicates that titanium combined with copper to create a dual-metal structure based on the paratacamite phase. This combination produced the stabilized paratacamite phase, which usually exists in a metastable form.
To create the triple-metal structure, a new metal (zinc) was combined with copper in addition to titanium through the structure of paratacamite. The peaks of the paratacamite phase did not disappear, and a new structure started to grow, as shown in Figure 1c. The XRD pattern of the sample CZT-3 is shown in Figure 1c. Figure 1c shows weak peaks at 2θ = 13.3°, 27.1°, and 39.3° in agreement with d-spacings of 0.67, 0.33, and 0.23 nm. These peaks are assigned to the characteristic reflections of the planes [003], [006], and [009] of Zn-Ti layered double hydroxide (Zn-Ti-carbonate LDH) [30]. The peaks of CZT-3 exhibit some common features of nanolayered materials, yielding 0.67 nm = 2 × 0.33 nm = 3 × 0.23 nm. In addition, Figure 1c shows the peaks of the paratacamite structure, which indicates that the sample CZT-3 contains a network consisting of two structures: paratacamite and LDHs. The XRD results confirmed the formation of the triple-metal nanostructure.
The different metals and elements in the prepared sample CZT-3 were identified and analyzed using energy-dispersive X-ray spectrometry (EDX) to give clear information on its chemical composition. During the EDX measurements, the corresponding peaks of the main elements of paratacamite are observed, as shown in Figure 2.
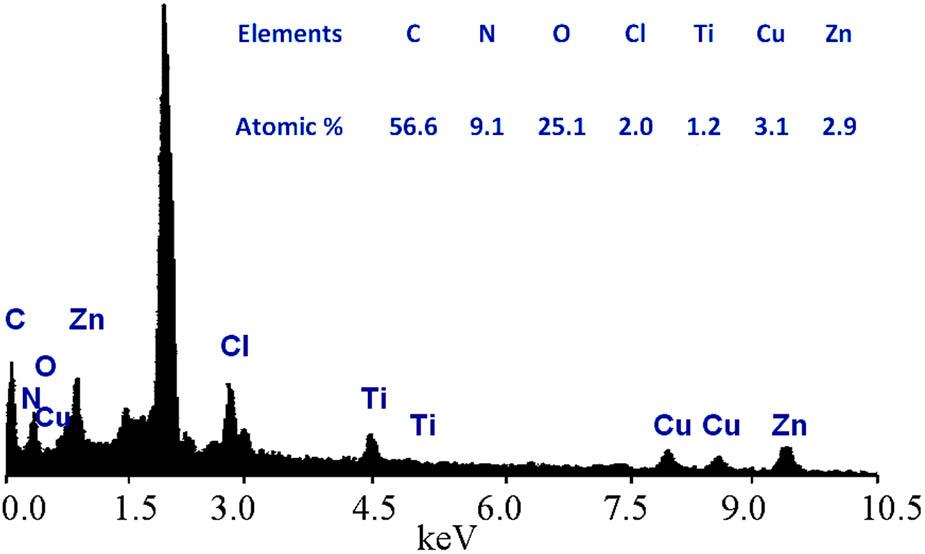
Chemical composition and the EDX spectrum of CZT-3.
The atomic percentages of copper, zinc, chloride, and oxygen were 3.1, 2.9, 2.0, and 25.1%, respectively. In addition, titanium, carbon, and nitrogen in addition to zinc, are detected in the spectrum of CZT-3, confirming the presence of Zn–Ti–carbonate LDH, as shown in Figure 2. The strong and sharp peak in the spectrum was attributed to platinum, which was used for coating the sample for EDX measurements.
The thermal characteristics of the prepared samples Cu-HDS, CT-1, and CZT-3 were determined by TG and DTA, as shown in Figures 3 and 4. For Cu-HDS, the thermogravimetric curve presented in Figure 3a shows one sharp weight loss, indicating the dehydroxylation of the hydroxide layers and decomposition of nitrate anions. This occurs within a very close temperature range (225–265°C). The total weight loss was 33.7%, agreeing with the weight losses of nitrate anions and hydroxyl groups. This suggested that the two processes are essentially inseparable under these conditions. This was confirmed by only one endothermic peak at 244°C, as shown in the DTA curve in Figure 3a.
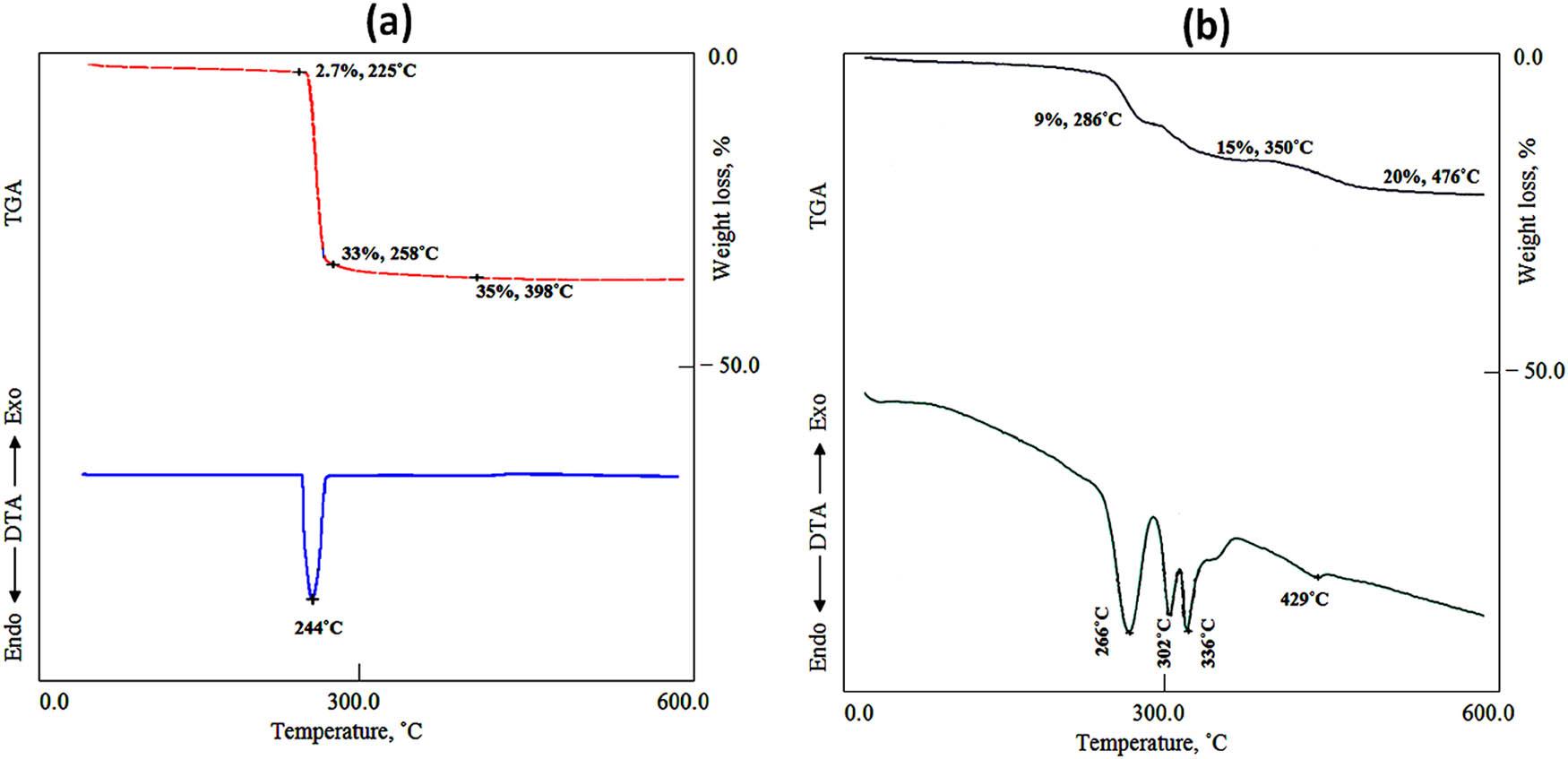
TGA and DTA of (a) Cu-HDS and (b) CT-1.
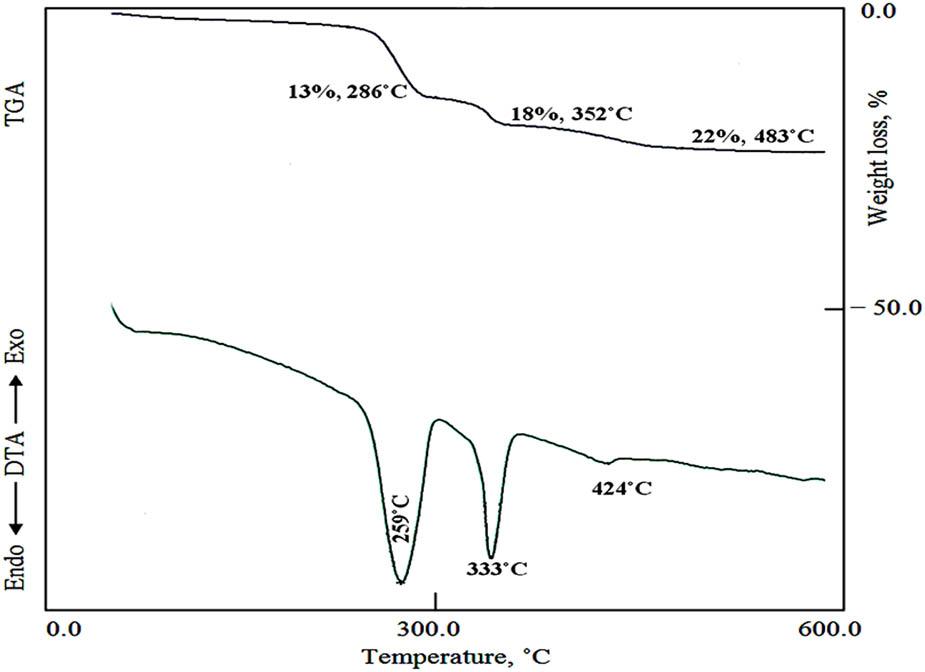
TGA and DTA of CZT-3.
In the case of CT-1, the thermal properties were observed (Figure 3b). The TG curve showed three weight losses, indicating three decomposition processes for the crystalline structure of CT-1. Figure 3b shows that the first weight loss up to 286°C is 9%, agreeing with the removal of chloride from the paratacamite structure. The second weight loss is 6% after heating to 350°C, which is due to the removal of hydroxyl groups from the paratacamite structure. The continuous heating up to 476°C caused the third weight loss. This weight loss was 5%, indicating the complete removal of the hydroxyl groups from the crystalline structure of CT-1.
The DTA diagram shows that the first weight loss is accompanied by one endothermic peak at 266°C for the removal of chloride from the paratacamite structure. However, two endothermic peaks were observed at 302 and 336°C for the second weight loss, confirming the removal of hydroxyl groups from the paratacamite structure. In addition, there is a weak endothermic peak at 429°C, agreeing with the complete removal of the hydroxyl groups.
By substituting zinc and reducing the percentage of copper, the TG curve of the sample CZT-3 showed that the first weight loss increased to 13% at 286°C, as shown in Figure 4. This high loss is due to the removal of chloride from the paratacamite structure, in addition to the decomposition of the interlayered anions of Zn–Ti–cyanate LDH. This finding confirmed the insertion of zinc inside the structure of Cu–Ti paratacamite and created the new structure of Zn-Ti-cyanate LDH, agreeing with the results of XRD.
In addition, the TG curve of the sample CZT-3 indicated that the second and third weight losses decreased to 5 and 4%, respectively. This indicates that the presence of zinc consumed titanium to create new LDHs, and the reduction of copper content led to decreasing the structure of Cu paratacamite. The TG results were confirmed by DTA data, as shown in Figure 4. The DTA curve showed that the endothermic peak attributed to the first weight loss became stronger. At the same time, the two endothermic peaks of the second weight loss were reduced to only one endothermic peak at 333°C. Also, the weak endothermic peak was observed at 429°C, indicating the complete removal of the hydroxyl groups.
SEM is a powerful tool for imaging of nanomaterials. To increase the resolution of the images, the samples are coated with platinum to perform SEM. Consequently, the powder samples were coated with thin films of platinum.
Figure 5 shows the SEM images of CT-1. A polyhedral structure was observed for the sample CT-1, agreeing with the paratacamite structure, as shown in Figure 5a. After magnification, Figure 5b shows a clear octahedral structure, agreeing with the results of Sciberras et al. [31]. Through further magnification, Figure 5c shows that the sample CT-1 consists of nanoparticles. These nanoparticles were aggregated to form the polyhedral structure of the paratacamite phase, confirming the results of XRD.

SEM images of CT-1: (a) at 10 micrometer, (b) at 1.0 micrometer and (c) at 100 nanometer.
Figure 6 shows the morphology of the sample CZT-3. By inserting zinc with titanium inside the copper structure, SEM images showed new morphology in addition to the nanoparticles of the paratacamite phase, as shown in Figure 6a. Figure 6a shows a network consisting of the nanoparticles of paratacamite and the nanolayers of LDHs. By magnification to the nanoscale, a clear plate-like structure is observed, as shown in Figure 6b, confirming the presence of Zn-Ti LDH and agreeing with the literature on LDH structures. In the same location, there are particles with the size of 20 nm, representing the paratacamite structure. This indicates that zinc consumed titanium to create the new phase of the LDH structure in addition to the paratacamite phase, as confirmed by the XRD and EDX analyses.

SEM images of CZT-3: (a) at 10 micrometer and (b) at 100 nanometer.
During the evaluation of the electrochemical capabilities of the Cu-HDS, CT-1, and CZT-3 electrodes, a trio-electrode cell configuration was employed, incorporating both CV and galvanostatic charge–discharge (GCD) methods within an aqueous solution environment (Figures 7–10). Initial assessments displayed each electrode’s redox potential in the 0.0–0.5 V range at a scanning velocity of 10 mV/s, illustrating their Faradaic characteristics. These electrodes were crafted under specific experimental conditions that optimized the balance between oxidative and reductive qualities. The dual-metal composition of copper and zinc played a pivotal role in enhancing the capacitive properties of the electrodes by leveraging their redox activity in an alkaline setting. Of the tested electrodes, CZT-3 stood out by presenting a notably larger area under the CV curve, indicative of superior electrochemical efficiency. This heightened activity is attributed to a synergistic effect arising from the unique morphology and the electrochemical dynamics of the Cu–Zn.
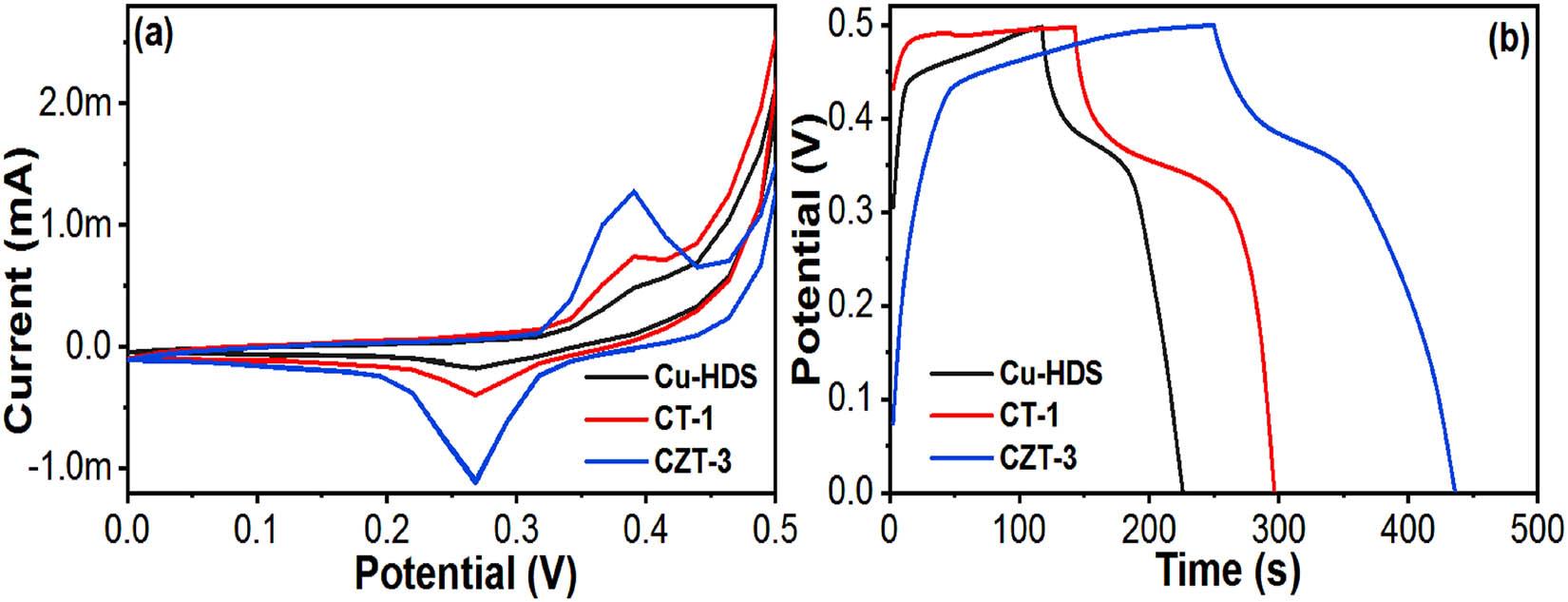
Comparative (a) CV and (b) CD of the Cu-HDS, CT-1, and CZT-3 electrodes at different current densities.
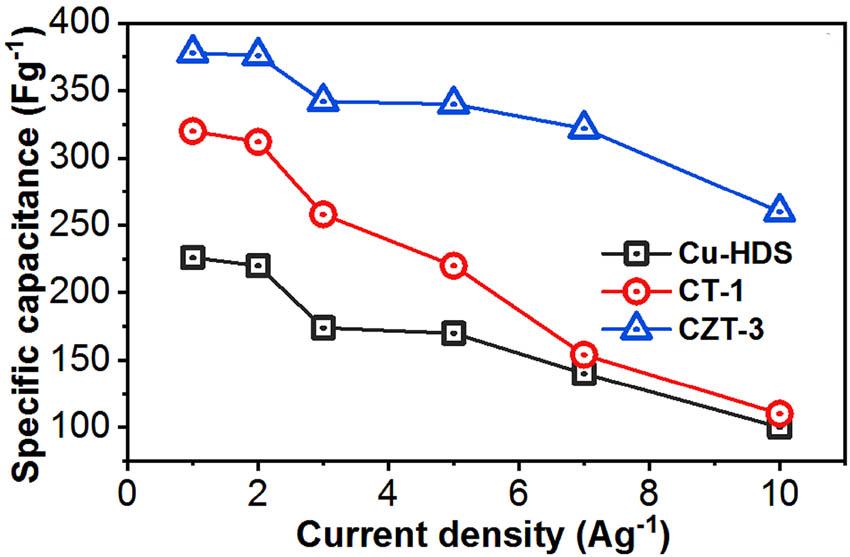
Comparative specific capacitances of the Cu-HDS, CT-1, and CZT-3 electrodes at different current densities.
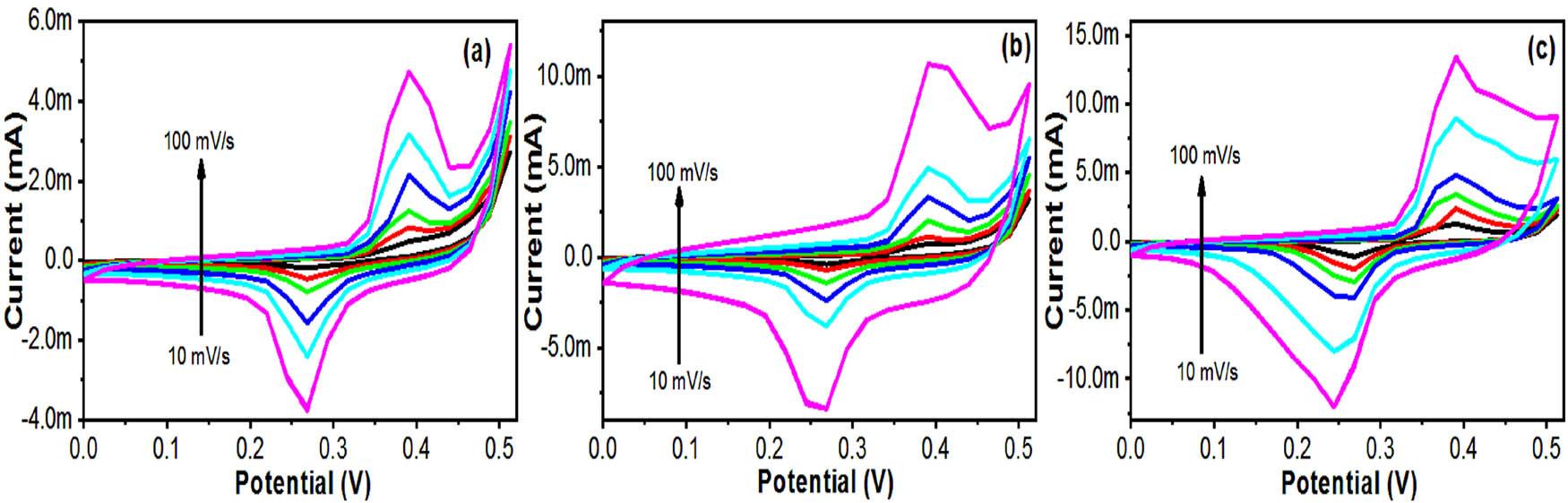
CV of the (a) Cu-HDS, (b) CT-1, and (c) CZT-3 electrodes at different scan rates.
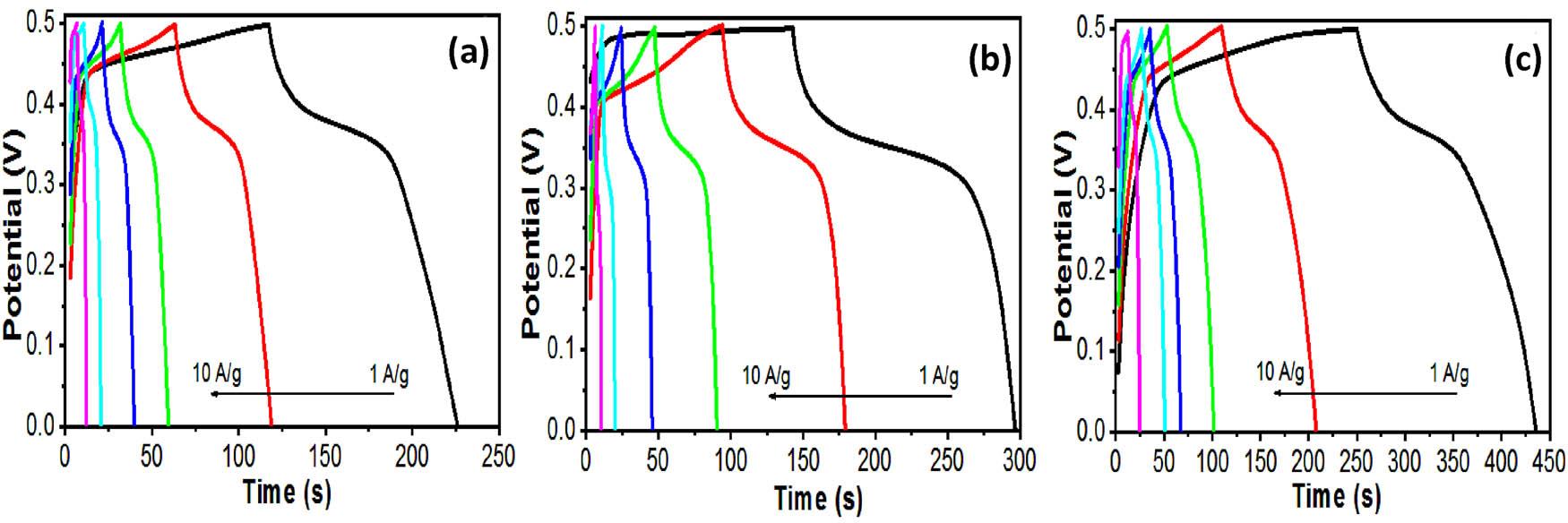
GCD curves of the (a) Cu-HDS, (b) CT-1, and (c) CZT-3 electrodes at different current densities.
Additionally, variations in the dopant ratios contributed to the modulation of the electrodes’ structure and size, particularly in the layered double hydroxides, further amplifying the CZT-3 electrode’s performance. To gain insights into the electrochemical efficiency and capacitance properties of the Cu-HDS, CT-1, and CZT-3 electrodes, their GCD profiles were analyzed at various current intensities. Figure 8 shows these profiles, indicating the dynamic and swift electrochemical responses of each electrode, thus reinforcing their Faradaic properties.
The specific capacitance of these electrodes was evaluated by analyzing the discharge durations, offering a quantitative measure of their energy storage capabilities. This approach not only confirmed the distinct capacitive behaviors of the Cu-HDS, CT-1, and CZT-3 electrodes but also highlighted their potentials for advanced electrochemical applications.
The electrochemical behavior of the Cu-HDS, CT-1, and CZT-3 electrodes was explored under varying current densities, providing key insights into their potential for supercapacitor applications. The GCD analysis (Figures 9 and 10), revealing nuanced performance metrics, confirms the excellent capacitive performance of the CZT-3 electrode across a broad spectrum of current loads. This is particularly notable when considering the electrodes’ specific capacitance, where the CZT-3 electrode consistently surpasses its counterparts, affirming its exceptional electrochemical storage capability.
The superior specific capacitance values observed for the CZT-3 electrode are attributed to its innovative layered double hydroxide structure. This unique configuration not only facilitates an enhanced ion exchange but also optimizes the electrode’s surface area, making it more receptive to electrochemical reactions. Such structural benefits are instrumental in promoting efficient ion mobility and ensuring a more stable CD cycle over extended periods. The CZT-3 electrode delivered the highest specific capacitances of 378, 376, 342, 340, 322, and 260 F/g at current loads of 1, 2, 3, 4, 5, 7, and 10 A/g (Figure 9), respectively. The CT-1 electrode delivered the highest specific capacitances of 320, 312, 258, 220, 154, and 110 F/g at current loads of 1, 2, 3, 4, 5, 7, and 10 A/g, respectively (Figure 10c). The Cu-HDS electrode delivered the highest specific capacitances of 226, 220, 174, 170, 140, and 100 F/g at current loads of 1, 2, 3, 4, 5, 7, and 10 A/g, respectively (Figure 10a).
Analyzing the specific capacitances across varying current loads, the CZT-3 electrode exhibited significant performance compared to that of both the CT-1 and Cu-HDS electrodes, showcasing capacitance levels ranging from 378 F/g at 1 A/g to 260 F/g at 10 A/g. In comparison, the CT-1 electrode’s performance peaked at 320 F/g at 1 A/g, gradually decreasing to 110 F/g at 10 A/g, while the Cu-HDS electrode’s capacitance started at 226 F/g at 1 A/g and dropped to 100 F/g at 10 A/g. The significant performance of the CZT-3 electrode is largely attributed to its layered double hydroxide structure, which significantly enhances ion diffusion and increases active site availability. This structural characteristic ensures that the CZT-3 electrode not only maintains a high level of specific capacitance but also demonstrates extended durability in CD cycles, reinforcing its suitability for advanced electrochemical applications.
In addition to specific capacitance, the cyclic durability of these electrodes emerges as a critical parameter. The CZT-3 electrode, in particular, exhibits remarkable resilience, maintaining a high percentage of its initial capacitance after thousands of cycles (Figure 11). This endurance is pivotal for the practical application of supercapacitors in energy storage, where longevity and reliability are as crucial as capacity.
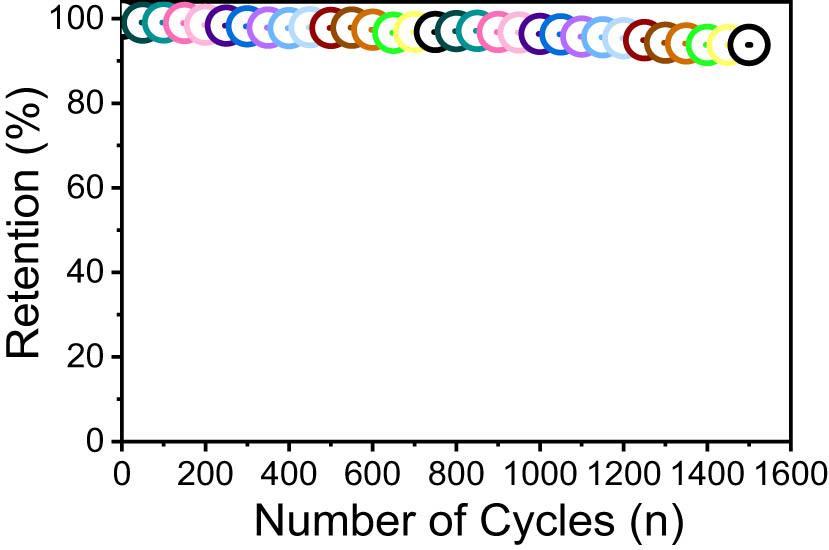
Cyclic stability graph of the CZT-3 electrode.
The amalgamation of high surface area, favorable metal–oxygen interactions, and efficient ion pathways within the CZT-3 electrode underscores its potential as a leading material for next-generation supercapacitors. These attributes contribute to its elevated performance metrics, positioning it as a viable option for enhancing energy storage technologies. The findings from this study not only validate the CZT-3 electrode’s superior capacitive behavior but also highlight its significance in advancing the development of high-performance, durable, and cost-effective supercapacitive devices.
The current study indicated that engineering Cu-based materials plays an important role in the fabrication of high-efficiency electrodes in supercapacitor technologies. In this track, mono, dual, and triple-metal nanostructures were designed to produce three different phases and morphologies. The mono-metal structure contained nanolayers of Cu-HDS with an interlayered spacing of 0.69 nm. The second morphology was produced by the dual-metal structure of copper and titanium (CT-1). This dual-metal structure contained nanoparticles of the paratacamite phase. The third morphology was formed by combining copper and titanium with zinc to build the triple-metal structure (CZT-3). A new morphology was observed for this triple-metal structure. This morphology was a network consisting of nanolayers and nanoparticles, which was suitable as a leading material for high-performance supercapacitors. This finding was ascertained by demonstrating that among the Cu-HDS, CT-1, and CZT-3 electrodes analyzed for supercapacitor applications, the CZT-3 electrode exhibited superior electrochemical performance. Its exceptional specific capacitance and remarkable cyclic stability, attributed to the combination of the nanolayered structures of LDHs and nanoparticles of paratacamite, highlight its potential as a leading material for high-performance supercapacitors. This research underscores the critical role of material composition and structural optimization in advancing energy storage technologies, positioning the CZT-3 electrode as a pivotal development for cost-effective, efficient, and durable supercapacitive devices.
Many studies, which have used ternary systems for storing energy, depended on the combination of stable materials such as K/graphene/LiCl gel [6], Cr2O3/graphene oxide/conducting polymers [7], and MgH2/halloysite nanotube/polyaniline [8]. In the current study, for the first time, titanium and zinc were inserted inside the copper nanostructures to stabilize the metastable Cu-paratacamite. At the same time, the Cu–Zn–Ti paratacamite was combined with Zn–Al nanolayers to form a network consisting of nanoparticles and nanolayers that is considered a promising candidate for low-cost, efficient, and durable supercapacitive devices, paving the way for further applications.
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [Grant No. KFU253702].
Conceptualization, O.S. and S.A.A; methodology, O.S. and S.A.A; software, M.O.; validation, N.P. and A.O.; formal analysis, O.S. and S.A.A; investigation, O.S., N.P.; and M.O.; resources, M.O., A.O., and N.P.; data curation, A.O., M.O., and N.P.; writing – original draft preparation, O.S., N.P., M.O., A.O,. and S.A.A; writing – review and editing, O.S. and S.A.A; visualization, O.S.; supervision, O.S. and S.A.A; project administration, O.S.; funding acquisition, O.S.
Authors state no conflict of interest.
Data will be provided by the authors upon request.