Fig. 1
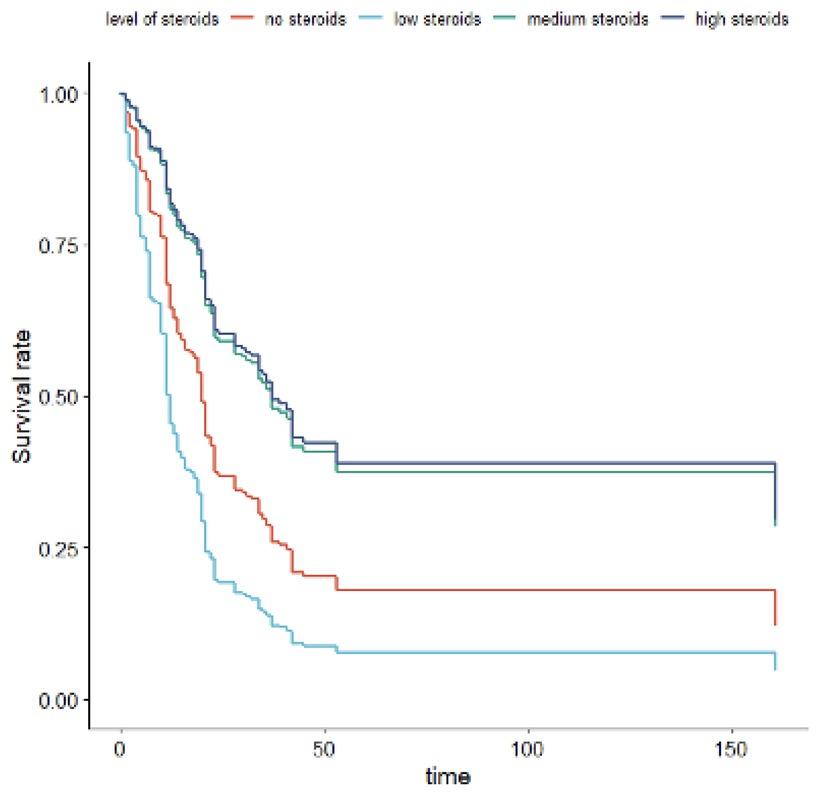
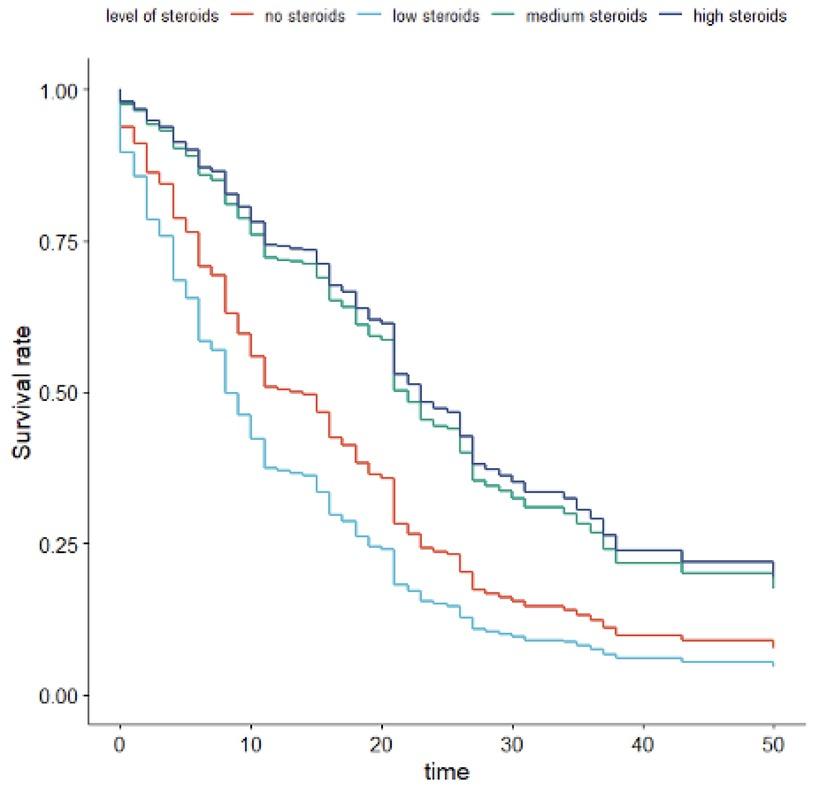
Fig. 2
Sample Characteristics Continuous Variables
| Total N=235 | No steroids N=23 | Low dose N=42 | Medium dose N=23 | High dose N=151 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Continuous variables | Median | IQR | Median | IQR | Median | IQR | Median | IQR | Median | IQR |
| Demographic recorded | ||||||||||
| Age(years) | 49 | 42-57 | 47 | 39-60 | 50 | 45-58 | 50 | 47-52 | 49 | 42-56 |
| BMI (Kg/) | 27 | 23-30 | 26 | 24-32 | 26 | 21-31 | 24 | 21-31 | 28 | 24-32 |
| Days turn test negative | 5 | 0-16 | 2 | 0-10 | 0 | 0-9 | 8 | 3-23 | 7 | 0-18 |
| Inpatient clinical data on admission | ||||||||||
| Oxygen (ml) | 10 | 0-15 | 4 | 0-10 | 15 | 4-15 | 15 | 8.5-15 | 15 | 0.05-10 |
| Laboratory parameters | ||||||||||
| Ferritin (ng/ml) | 1230 | 483-1895 | 468 | 120-1520 | 620 | 214-1804 | 1204 | 532-750 | 1385 | 750-1986 |
| D-Dimer (ng/ml) | 1.11 | 0.52-3.80 | 0.61 | 0.29-3.02 | 1.17 | 0.58-2.2 | 3.9 | 1.01-8.99 | 1.11 | 0.58-3.43 |
| Procalcitonin (ng/ml) | 0.35 | 0.14-1.168 | 0.27 | 0.1-2.26 | 0.21 | 0.1-1.23 | 0.31 | 0.15-0.98 | 0.37 | 0.16-1.10 |
| CRP (mg/L) | 129.5 | 75.2-215.88 | 104 | 64.8-216 | 133 | 73.4-161 | 98 | 59.6-156 | 137 | 84.5-225 |
| Creatinine (mg/dl) | 0.9 | 0.8-1.2 | 0.9 | 0.7-1.28 | 0.8 | 0.7-1.6 | 0.9 | 0.78-1.3 | 1 | 0.8-1.2 |
| CPK (units/L) | 196.5 | 69.25-626.50 | 120 | 17.8-439 | 164 | 52-410 | 310 | 141-609 | 218 | 86-689 |
| ABG PH | 7.37 | 7.25-7.43 | 7.35 | 0-7.43 | 7.3 | 7.19-7.38 | 7.40 | 7.35-7.44 | 7.37 | 7.28-7.43 |
| PCo2 (Torr) | 35.8 | 29.93-45.58 | 30 | 0-36.4 | 37.1 | 32.6-48.6 | 35.2 | 31.7-42.9 | 38 | 31.5-48.3 |
| PO2 (Torr) | 61.3 | 44.30-84.95 | 48.7 | 0-88.7 | 64 | 47-73 | 61.8 | 45.5-75 | 62 | 47.4-85.7 |
| Lactate | 1.7 | 1.2-2.5 | 1.35 | 0.23-2.28 | 2.4 | 1.4-6.3 | 1.9 | 1.37-3.47 | 1.7 | 1.2-2.3 |
| Bicarbonate (mEq/L) | 21.55 | 18.8-24 | 20 | 14.2-22.5 | 20.7 | 16.3-23.9 | 21.4 | 19.7-23.6 | 22.1 | 19.5-24.4 |
| Magnesium (mg/dl) | 2.03 | 1.84-2.24 | 2.01 | 1.72-2.16 | 1.94 | 1.7-2.24 | 2.09 | 1.87-2.39 | 2.04 | 1.90-2.26 |
| Platelets (/microliter) | 196.5 | 150.25-260.5 | 206 | 158-252 | 214 | 141-299 | 164 | 126-253 | 197 | 152-262 |
| WBC (/microliter) | 7.9 | 6.03-10.7 | 7.9 | 5.9-10.8 | 8.1 | 6.3-13 | 7.15 | 6.15-9.85 | 7.9 | 6-10.7 |
| Severity of illness | ||||||||||
| APACHE 2 score | 16 | 12-20 | 15.5 | 2.25-24.5 | 16 | 13-22 | 18.5 | 13-24.2 | 16 | 12-19 |
| Variable of interest | ||||||||||
| Days on Mech. Vent. | 10 | 4-20 | 4 | 0-8.75 | 2 | 1-9 | 15 | 7.75-23 | 14 | 8-23 |
| LOSICU (days) | 12 | 4-22.75 | 4 | 0-8.75 | 2 | 1-9 | 17 | 8.75-23.8 | 18 | 10-30.5 |
| LOS in hospital (days) | 18 | 8-32 | 10 | 6.25-18.8 | 5 | 2-12 | 28 | 13.8-49.5 | 22 | 12.5-35.5 |
Sample characteristics (categorical variables)
| Categorical variables | Total Sample N 235 | No steroids N 23 | Low dose N 42 | Medium dose N 23 | High dose N 151 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1= variable present | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 |
| Demographic recorded | ||||||||||
| Gender (F=0, M=1) (%) | 13 | 87 | 3.8 | 17 | 2.9 | 7.6 | 1.3 | 7.1 | 4.6 | 55.5 |
| Presence of symptoms | ||||||||||
| cough | 20 | 80 | 6.7 | 14.3 | 2.1 | 8.4 | 2.5 | 5.9 | 8.8 | 51.3 |
| fever | 9 | 91 | 2.5 | 18.5 | 1.3 | 9.2 | 0.4 | 8 | 5 | 55 |
| dyspnea | 20 | 80 | 7.6 | 13.4 | 1.3 | 9.2 | 1.7 | 6.7 | 9.7 | 50.4 |
| Gastric complaints | 88 | 12 | 18.5 | 2.5 | 8 | 2.5 | 7.1 | 1.3 | 54.6 | 5.5 |
| Comorbidities | ||||||||||
| diabetes | 57 | 43 | 10.1 | 10.9 | 7.1 | 3.4 | 5.5 | 3 | 34.5 | 25.6 |
| hypertension | 75 | 25 | 15.5 | 5.5 | 6.3 | 4.2 | 5 | 3.4 | 48.3 | 11.8 |
| coronary disease | 93 | 7 | 18.5 | 2.5 | 8.8 | 1.7 | 8 | 0.4 | 58 | 2.1 |
| renal failure | 88 | 12 | 18.1 | 2.9 | 9.7 | 0.8 | 6.7 | 1.7 | 53.4 | 6.7 |
| dialysis | 93 | 7 | 19.7 | 1.2 | 9.7 | 0.8 | 7.6 | 0.8 | 56.3 | 3.8 |
| Inpatient clinical data on admission | ||||||||||
| Immunosuppressed | 96 | 4 | 20.6 | 0.4 | 10.1 | 0.4 | 7.1 | 1.3 | 58.4 | 1.7 |
| Fever | 14 | 86 | 4.2 | 16.8 | 1.7 | 8.8 | 1.3 | 7.1 | 6.7 | 53.4 |
| Tachycardia (pulse>100) | 21 | 79 | 5.5 | 15.5 | 2.1 | 8.4 | 2.5 | 5.9 | 11.3 | 48.7 |
| Hypotension on admission (MAP<60 mm of Hg) | 50 | 50 | 10.9 | 10.1 | 6.7 | 3.8 | 4.2 | 4.2 | 28.2 | 31.9 |
| Hypoxia on admission | 13 | 87 | 5.9 | 15.1 | 0.8 | 9.7 | 0.8 | 7.6 | 5.9 | 54.2 |
| Mechanical Ventilation | 15 | 85 | 7.1 | 13.9 | 2.5 | 8 | 0.4 | 8 | 4.6 | 55.5 |
| Vasopressors | 21 | 79 | 10.1 | 10.9 | 3.4 | 7.1 | 0.8 | 7.6 | 6.7 | 53.4 |
| Dialysis on admission | 70 | 30 | 16.4 | 4.6 | 8.4 | 2.1 | 5 | 3.4 | 39.9 | 20.2 |
| Clinical parameters | ||||||||||
| Lymphopenia (≤ 1100 cells/μL) | 44 | 56 | 12.2 | 8.8 | 7.1 | 3.4 | 2.9 | 5.5 | 21.8 | 38.2 |
| Bacterial infection | 49 | 51 | 16 | 5 | 9.2 | 1.3 | 3.4 | 5 | 20.2 | 39.9 |
| Bacteremia | 60 | 40 | 8.1 | 2.9 | 9.7 | 0.8 | 5.5 | 2.9 | 26.9 | 33.2 |
| Positive cultures | 45 | 55 | 15.1 | 5.9 | 8.8 | 1.7 | 2.5 | 5.9 | 18.1 | 42 |
| Arrhythmia | 15 | 22 | 15.5 | 5.5 | 8.8 | 1.7 | 6.7 | 1.7 | 47.1 | 13 |
| Associated treatment | ||||||||||
| Anticoagulants | 4 | 96 | 2.9 | 18.1 | 0 | 10.5 | 0 | 8.4 | 0.8 | 59.2 |
| Gastro-intestinal prophylaxis (Proton pump inhibitors) | 4 | 96 | 2.1 | 18.9 | 0.8 | 9.7 | 0 | 8.4 | 1.3 | 58.8 |
| Therapeutic agent | ||||||||||
| Lopinavir/Ritonavir | 6 | 94 | 3.4 | 17.6 | 1.3 | 9.2 | 0.4 | 8 | 1.3 | 58.8 |
| Chloroquine | 12 | 88 | 4.2 | 16.8 | 1.7 | 8.8 | 1.3 | 7.1 | 5 | 55 |
| Lopinavir/Ritonavir | 64 | 36 | 16.8 | 4.2 | 6.3 | 4.2 | 4.6 | 3.8 | 36.1 | 23.9 |
| Favipiravir | 20 | 80 | 10.5 | 10.5 | 2.5 | 8 | 1.3 | 7.1 | 5.9 | 54.2 |
| Tociluzimab | 84 | 16 | 20.2 | 0.8 | 9.7 | 0.8 | 6.7 | 1.7 | 47.5 | 12.6 |
| Plasmatherapy | 83 | 17 | 21 | 0 | 9.7 | 0.8 | 6.7 | 1.7 | 45.8 | 14.3 |
| Tracheostomy | 87 | 17 | 20.2 | 0.8 | 10.5 | 0 | 7.6 | 0.8 | 48.7 | 11.3 |
| ECMO | 95 | 5 | 21 | 0 | 10.5 | 0 | 7.6 | 0.8 | 55.5 | 4.6 |
| Surgeries | 96 | 4 | 20.6 | 0.4 | 10.5 | 0 | 8 | 0.4 | 56.7 | 3.4 |
| Sedatives | 11 | 89 | 6.7 | 14.3 | 1.7 | 8.8 | 0.4 | 8 | 2.5 | 57.6 |
| Narcotics | 24 | 76 | 9.7 | 11.3 | 1.7 | 8.8 | 2.1 | 6.3 | 10.5 | 49.6 |
| Neuromuscular blocking agents | 15 | 85 | 8.8 | 12.2 | 2.5 | 8 | 0.8 | 7.6 | 2.9 | 57.1 |